Spectral Medical Appoints Former FDA Director, Dr. David W. Feigal, Jr., to Its Board of Directors
27 Juni 2023 - 2:00PM

Spectral Medical Inc. (“Spectral” or the “Company”) (TSX:
EDT), a late-stage theranostic company advancing
therapeutic options for sepsis and septic shock
,
today announced that it has appointed Dr. David W. Feigal, Jr.,
M.D., M.P.H., to its Board of Directors.
Dr. Feigal brings over four decades of
experience in regulatory affairs and clinical research of medical
devices, biologics, and products in multiple therapeutic areas.
Currently, Dr. Feigal is a Partner at NDA Partners LLC, a ProPharma
Company, which is a life sciences management consulting and
contract development organization (CDO) focused on providing
product development and regulatory services to the pharmaceutical,
biotechnology, and medical device industries worldwide. He has also
worked for major U.S. and international life science corporations
such as Amgen, Inc. serving as Vice President of Global Requlatory
Affairs, and Elan Pharmaceuticals, Inc., serving as Senior Vice
President of Global Regulatory Affairs and Global Safety
Surveillance and Biostatistics. He has an extensive track record of
international regulatory success with the approval/clearance of
medical products in numerous therapeutic areas.
Dr. Feigal also has extensive experience as a
regulator and spent 12 years at the Food and Drug Administration
(FDA), holding senior roles within the Center for Drug Evaluation
and Research (CDER), including Director, Division of Anti-Viral
Drug Products; Acting Director, Division of Anti-Infective Drug
Products; Director, Office of Drug Evaluation-IV; as well as
Medical Deputy Center Director and ultimately Center Director
within the Center for Devices and Radiological Health (CDRH).
Prior to joining the FDA, he worked for a decade
within the academic and hospital settings of the University of
California, San Diego, San Francisco, and Davis. Dr. Feigal has
contributed to global government programs conducted by the World
Health Organization, the European Medicines Evaluation Agency
(EMEA), the National Institutes of Health, and others. He has
served on numerous committees within the U.S. Department of Health
and Human Services, has served on boards of both companies and
funds, is a renowned speaker, and has published over 50 papers. He
holds a BA from the University of Minnesota, an MD from Stanford
University and a Master of Public Health from the University of
California, Berkeley.
“We are extremely proud to have Dr. Feigal join
our Board of Directors,” commented, Anthony Bihl, Chairman of the
Board at Spectral Medical. “Dr. Feigal brings decades of experience
in the medical field and extensive knowledge regarding clinical
development and regulatory processes. Given the status of our Phase
3 trial of PMX and our continued progression toward completion and
potential FDA submission, we believe Dr. Feigal will provide
valuable insights and assist with meeting the complex requirements
of the FDA. With the combined knowledge and expertise of Dr.
Feigal, along with the current leadership team, we are well
positioned to advance Spectral to meaningful milestones and
potential FDA approval of PMX, bringing a personalized therapy to
those suffering from endotoxemic septic shock.”
Chris Seto, Chief Executive Officer of Spectral
Medical, added, “We are approaching an inflection point for
Spectral with respect to clinical development. We have undertaken
efforts to enhance enrollment of Tigris, and continue to witness
data that exceeds our expectations. As we expect to hit significant
milestones relating to PMX, we are excited to have Dr. Feigal on
our Board to provide his expertise and valuable knowledge to assist
our efforts of obtaining FDA approval. We look forward to his
contributions to Spectral and aiding in the advancement of PMX to
market.”
About
Spectral
Spectral is a Phase 3 company seeking U.S. FDA
approval for its unique product for the treatment of patients with
septic shock, Toraymyxin™ (“PMX”). PMX is a therapeutic
hemoperfusion device that removes endotoxin, which can cause
sepsis, from the bloodstream and is guided by the Company’s
Endotoxin Activity Assay (EAA™), the only FDA cleared diagnostic
for the risk of developing sepsis.
PMX is approved for therapeutic use in Japan and
Europe, and has been used safely and effectively on more than
340,000 patients to date. In March 2009, Spectral obtained the
exclusive development and commercial rights in the U.S. for PMX,
and in November 2010, signed an exclusive distribution agreement
for this product in Canada. In July 2022, the U.S. FDA granted
Breakthrough Device Designation for PMX for the treatment of
endotoxemic septic shock. Approximately 330,000 patients are
diagnosed with septic shock in North America each year.
Spectral is listed on the Toronto Stock Exchange
under the symbol EDT. For more information please visit
www.spectraldx.com.
Forward-looking statement
Information in this news release that is not
current or historical factual information may constitute
forward-looking information within the meaning of securities laws.
Implicit in this information, particularly in respect of the future
outlook of Spectral and anticipated events or results, are
assumptions based on beliefs of Spectral's senior management as
well as information currently available to it. While these
assumptions were considered reasonable by Spectral at the time of
preparation, they may prove to be incorrect. Readers are cautioned
that actual results are subject to a number of risks and
uncertainties, including the availability of funds and resources to
pursue R&D projects, the successful and timely completion of
clinical studies, the ability of Spectral to take advantage of
business opportunities in the biomedical industry, the granting of
necessary approvals by regulatory authorities as well as general
economic, market and business conditions, and could differ
materially from what is currently expected.
The TSX has not reviewed and does not accept responsibility for
the adequacy or accuracy of this statement.
For further information, please contact:
| Ali Mahdavi |
David Waldman/Natalya
Rudman |
| Capital Markets & Investor
Relations |
US Investor Relations |
| Spinnaker Capital Markets
Inc. |
Crescendo Communications,
LLC |
| 416-962-3300 |
212-671-1020 |
|
am@spinnakercmi.com |
edt@crescendo-ir.com |
|
|
Blair McInnis |
| |
CFO |
| |
Spectral Medical Inc. |
| |
416-626-3233 |
| |
bmcinnis@spectraldx.com |
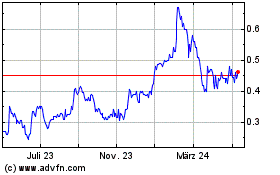
Spectral Medical (TSX:EDT)
Historical Stock Chart
Von Apr 2024 bis Mai 2024
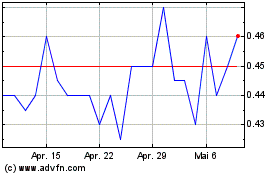
Spectral Medical (TSX:EDT)
Historical Stock Chart
Von Mai 2023 bis Mai 2024