Form 8-K - Current report
08 Januar 2024 - 2:00PM
Edgar (US Regulatory)
false
0001737953
0001737953
2024-01-08
2024-01-08
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event
reported): January 8, 2024
REPLIMUNE GROUP, INC.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-38596 |
|
82-2082553 |
(State or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification Number) |
500 Unicorn Park Drive
Suite 303
Woburn, MA 01801
(Address of principal executive offices, including Zip Code)
Registrant’s telephone number, including
area code: (781) 222-9600
Check the appropriate box below if the Form 8-K filing is
intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ¨ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR
230.425) |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR
240.14a-12) |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR
240.14d-2(b)) |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR
240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
|
Trading
Symbol(s) |
|
Name of each exchange on which registered |
| Common Stock, par value $0.001 per share |
|
REPL |
|
The Nasdaq Stock Market LLC
(Nasdaq Global Select Market) |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933
(§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this
chapter). Emerging growth company x
If an
emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for
complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
| Item 7.01 | Regulation FD Disclosure. |
On
January 8, 2024, Replimune Group, Inc. (the “Company”) announced updated clinical data of RP1 and RP2 during a
presentation at the 42nd Annual J.P. Morgan Healthcare Conference. A copy of the presentation slides
are furnished as Exhibit 99.1 to this Current Report on Form 8-K and a replay of the webcast will be available on the Company’s
website at www.replimune.com under “Investors and Media” for 30 days following the event. The Company undertakes
no obligation to update, supplement or amend the materials attached hereto.
The information contained in this Item 7.01 and in the accompanying
Exhibit 99.1 shall not be incorporated by reference into any filing of the Company, whether made before or after the date hereof,
regardless of any general incorporation language in such filing, unless expressly incorporated by specific reference to such filing. The
information in this Item 7.01 and the accompanying Exhibit 99.1 shall not be deemed to be “filed” for purposes of Section 18
of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section or Sections 11 and 12(a)(2) of
the Securities Act of 1933, as amended.
The Company preliminarily estimates that as of December 31, 2023,
it had approximately $466 million in cash and cash equivalents and short-term investments. The Company believes that its existing cash
and cash equivalents and short-term investments along with its debt commitments will enable it to fund its operating expenses and capital
expenditure requirements into the second half of 2026.
This amount is unaudited and preliminary, and does not present all
information necessary for an understanding of the Company’s financial condition as of December 31, 2023. The review of the
Company’s condensed consolidated financial statements for the three and nine-months ended December 31, 2023 is ongoing and
could result in changes to the preliminary estimates due to the completion of financial closing procedures, final adjustments and other
developments that may arise between now and the time the condensed consolidated financial statements the three and nine-months ended December 31,
2023 are finalized and publicly released. The Company’s independent registered public accounting firm, PricewaterhouseCoopers LLP,
has not audited, reviewed, compiled or performed any procedures with respect to the preliminary financial estimate, and does not express
an opinion or any other form of assurance with respect thereto. The preliminary financial estimate presented above has been prepared by
and is the responsibility of management. Estimates of financial results are inherently uncertain and subject to change, and the Company
undertakes no obligation to update this information. In addition, the estimated balance of cash and cash equivalents and short-term investments
as of December 31, 2023 is not necessarily indicative of future performance or any other period, including the results to be achieved
for the remainder of the fiscal year ending March 31, 2024 or any future period.
| Item 9.01 | Financial Statements and Exhibits. |
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly
authorized.
| |
REPLIMUNE GROUP, INC. |
| |
|
|
| Date: January 8, 2024 |
By: |
/s/ Philip Astley-Sparke |
| |
|
Philip Astley-Sparke |
| |
|
Chief Executive Officer |
Exhibit 99.1
| Safe harbor
Any statements contained herein that are not statements of historical facts may be deemed to be forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended,
including statements regarding the advancement, timing and sufficiency of our clinical trials, patient enrollments in our existing and
planned clinical trials and the timing thereof, the results of our clinical trials, the timing and release of our clinical data, statements
regarding our expectations about our cash runway, our goals to develop and commercialize our product candidates, our expectations
regarding the size of the patient populations for our product candidates if approved for commercial use and other statements identified
by words such as “could,” “expects,” “intends,” “may,” “plans,” “potential,” “should,” “will,” “would,” or similar expressions and the negatives
of those terms. Forward-looking statements are not promises or guarantees of future performance, and are subject to a variety of risks
and uncertainties, many of which are beyond our control, and which could cause actual results to differ materially from those
contemplated in such forward-looking statements. These factors include risks related to our limited operating history, our ability to
generate positive clinical trial results for our product candidates, the costs and timing of operating our in-house manufacturing facility,
the timing and scope of regulatory approvals, changes in laws and regulations to which we are subject, competitive pressures, our ability
to identify additional product candidates, political and global macro factors including the impact of the SARS-COV-2 coronavirus as a
global pandemic and related public health issues, and other risks as may be detailed from time to time in our Annual Reports on Form
10-K, Quarterly Reports on Form 10-Q, and other reports we file with the Securities and Exchange Commission. Our actual results could
differ materially from the results described in or implied by such forward-looking statements. Forward-looking statements speak only as
of the date hereof, and, except as required by law, we undertake no obligation to update or revise these forward-looking statements.
Igniting a Systemic Immune
Response to Cancer
JP Morgan
January 2024 |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 2
Safe harbor
Any statements contained herein that are not statements of historical facts may be deemed to be forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended,
including statements regarding the advancement, timing and sufficiency of our clinical trials, patient enrollments in our existing and
planned clinical trials and the timing thereof, the results of our clinical trials, the timing and release of our clinical data, statements
regarding our expectations about our cash runway, our goals to develop and commercialize our product candidates, our expectations
regarding the size of the patient populations for our product candidates if approved for commercial use and other statements identified
by words such as “could,” “expects,” “intends,” “may,” “plans,” “potential,” “should,” “will,” “would,” or similar expressions and the
negatives of those terms. Forward-looking statements are not promises or guarantees of future performance, and are subject to a
variety of risks and uncertainties, many of which are beyond our control, and which could cause actual results to differ materially from
those contemplated in such forward-looking statements. These factors include risks related to our limited operating history, our ability
to generate positive clinical trial results for our product candidates, the costs and timing of operating our in-house manufacturing
facility, the timing and scope of regulatory approvals, changes in laws and regulations to which we are subject, competitive pressures,
our ability to identify additional product candidates, political and global macro factors including the impact of the SARS-COV-2
coronavirus as a global pandemic and related public health issues, the ongoing military conflict between Russia and Ukraine and the
impact on the global economy and related governmental imposed sanctions, and other risks as may be detailed from time to time in
our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, and other reports we file with the Securities and Exchange
Commission. Our actual results could differ materially from the results described in or implied by such forward-looking statements.
Forward-looking statements speak only as of the date hereof, and, except as required by law, we undertake no obligation to update or
revise these forward-looking statements. |

| © 2022 Replimune Group Inc. 3 3
• RP1 – activity across multiple skin cancers supports broad skin cancer strategy
• 140 patient registrational IGNYTE study in anti-PD1 failed melanoma
• ~ 1 in 3 patients demonstrating durable response
• 100% of responses >6 months with median DOR >24 months
• BLA filing planned 2H 2024
• 211 patient 1L CSCC randomized controlled CERPASS study; primary analysis reported December 2023
• Missed significance at P<0.025 for dual endpoints (ORR/CRR)
• However, clear clinical benefit for RP1+cemiplimab was demonstrated
• CRR vs. cemplimab alone (38.1% vs 25.0%, p=0.0401)
• Duration of response increased
• Strong data in hard-to-treat solid organ transplant patients as monotherapy
• Potential for the portfolio to deliver commercial revenues beginning in late 2025
• RP2 has shown compelling monotherapy and combination activity
• Uveal melanoma RCT study in planning -> potential for a rare cancer franchise
• Strong balance sheet ~ $466m (1) as of 31 December 2023; runway into H2 2026
Replimune; Industry Leader in Oncolytic Immunotherapy
1Per the protocol p<0.025 is required for formal statistical success in CERPASS for CRR or ORR alone. *SOT=solid organ transplant
(1) Unaudited estimate |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 4
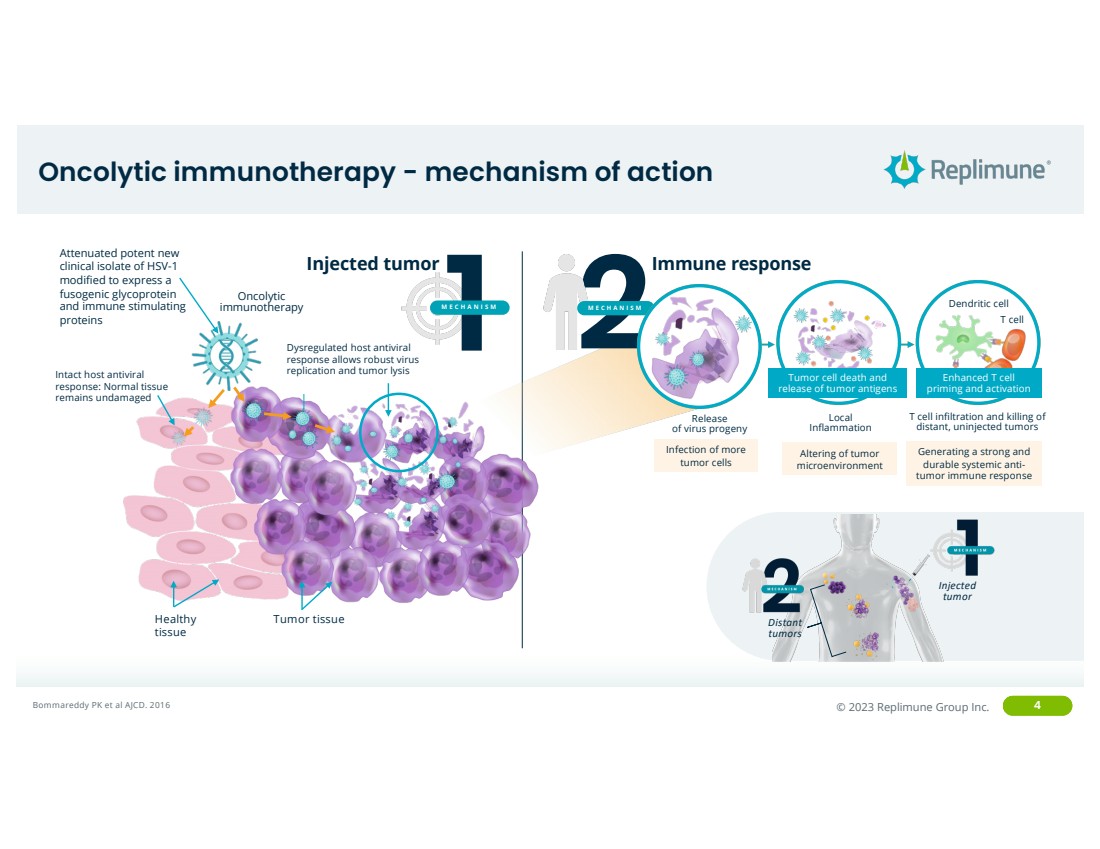
Oncolytic immunotherapy - mechanism of action
Injected
tumor
Distant
tumors
Bommareddy PK et al AJCD. 2016
Healthy
tissue
Tumor tissue
Oncolytic immunotherapy
Intact host antiviral
response: Normal tissue
remains undamaged
Dysregulated host antiviral
response allows robust virus
replication and tumor lysis
Attenuated potent new
clinical isolate of HSV-1
modified to express a
fusogenic glycoprotein
and immune stimulating
proteins
Injected tumor 1
MECHANISM 2
MECHANISM
Altering of tumor
microenvironment
Infection of more
tumor cells
Release
of virus progeny
Local
Inflammation
T cell infiltration and killing of
distant, uninjected tumors
Dendritic cell
T cell
Generating a strong and
durable systemic anti-tumor immune response
Immune response
Tumor cell death and
release of tumor antigens
Enhanced T cell
priming and activation |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 5
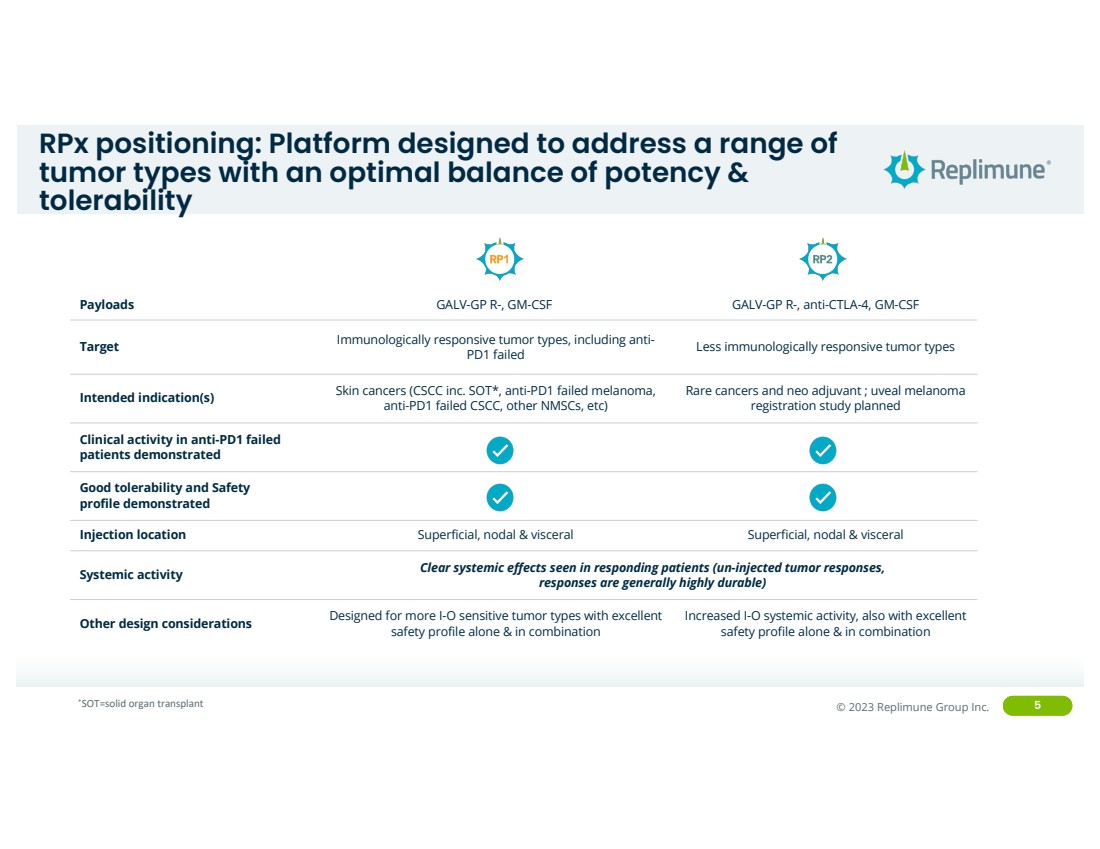
RPx positioning: Platform designed to address a range of
tumor types with an optimal balance of potency &
tolerability
Payloads GALV-GP R-, GM-CSF GALV-GP R-, anti-CTLA-4, GM-CSF
Target Immunologically responsive tumor types, including anti-PD1 failed Less immunologically responsive tumor types
Intended indication(s) Skin cancers (CSCC inc. SOT*, anti-PD1 failed melanoma,
anti-PD1 failed CSCC, other NMSCs, etc)
Rare cancers and neo adjuvant ; uveal melanoma
registration study planned
Clinical activity in anti-PD1 failed
patients demonstrated
Good tolerability and Safety
profile demonstrated
Injection location Superficial, nodal & visceral Superficial, nodal & visceral
Systemic activity Clear systemic effects seen in responding patients (un-injected tumor responses,
responses are generally highly durable)
Other design considerations Designed for more I-O sensitive tumor types with excellent
safety profile alone & in combination
Increased I-O systemic activity, also with excellent
safety profile alone & in combination
*SOT=solid organ transplant |

| © 2022 Replimune Group Inc. 6
AGENDA
© 2023 Replimune Group Inc.
RP1: Establishing a major
skin cancer franchise |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 7
IGNYTE RP1 + nivolumab in anti-PD1 failed melanoma
registrational study data
Consistent ORR benefit across all subgroups
Includes 1 patient with a unconfirmed PR (uPR). There are 5 patients still on study with the opportunity for response.
Response data presented is by investigator assessment; the primary analysis from the study will be by blinded, independent central review. BOR=best overall response
• 1 in 3 patients experienced a response
• 26.4% ORR in hard-to-treat Ipi+Nivo failed patients (approx. 50% of the overall study population)
• 100% of responses lasted >6 months, with median DOR >24 months
BOR
n (%)
All patients (n=156)
Prior
cohort
(n=16)
Anti-PD1
failed cohort
(n=140)
All patients
(n=156)
Prior single
agent anti-PD1
(n=84)
Prior combination
anti–PD-1 & anti–
CTLA-4*
(n=72)
Stage
IIIb/IIIc/IVa
(n=76)
Stage IVb/c/d
(n=80)
Primary
resistance to
anti-PD1
(n=91)
Secondary
resistance to
anti-PD1
(n=63)
CR 2 (12.5) 17 (12.1) 19 (12.2) 14 (16.7) 5 (6.9) 15 (19.7) 4 (5.0) 12 (13.2) 6 (9.5)
PR 4 (25.0) 26 (18.6) 30 (19.2) 16 (19.0) 14 (19.4) 14 (18.4) 16 (20.0) 19 (20.9) 11 (17.5)
SD 2 (12.5) 29 (20.7) 31 (19.9) 21 (25.0) 10 (13.9) 18 (23.7) 13 (16.3) 15 (16.5) 16 (25.4)
PD 8 (50.0) 68 (48.6) 76 (48.7) 33 (39.3) 43 (59.7) 29 (38.2) 47 (58.8) 45 (49.5) 30 (47.6)
ORR 6 (37.5) 43 (30.7) 49 (31.4) 30 (35.7) 19 (26.4) 29 (38.2) 20 (25.0) 31 (34.1) 17 (27.0) |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 8
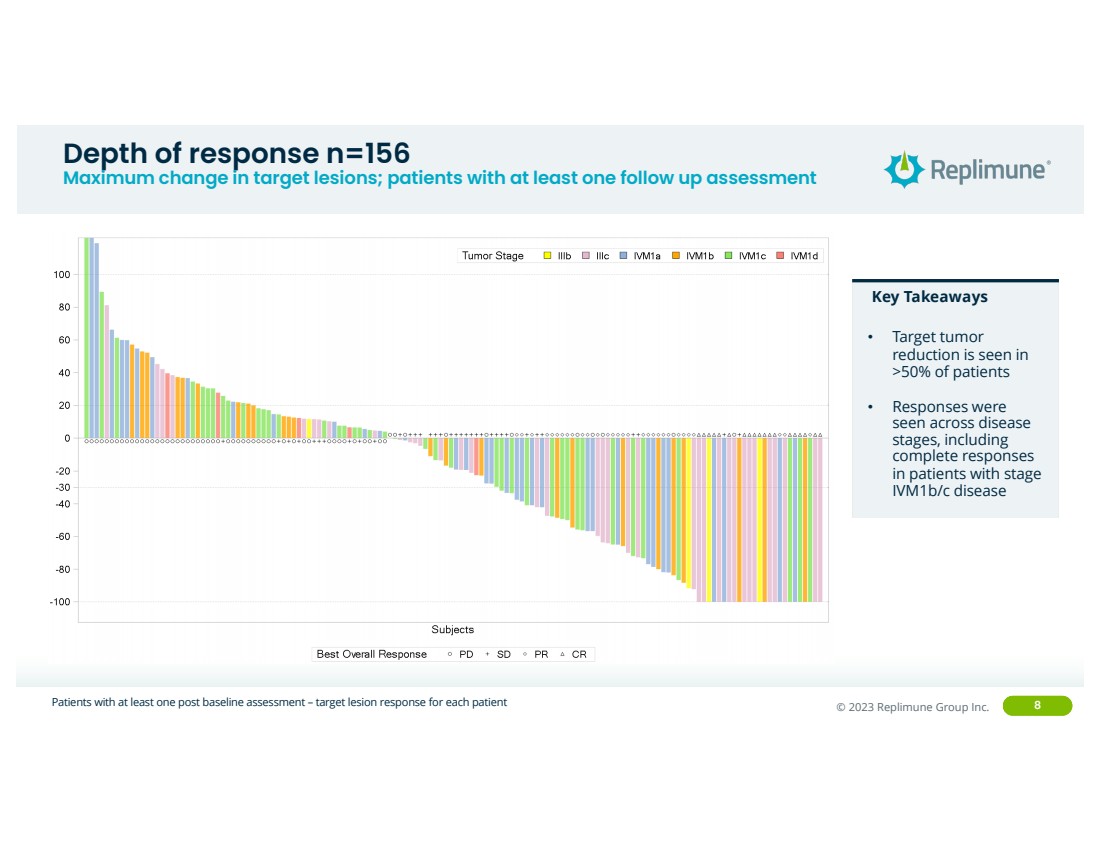
Depth of response n=156
Maximum change in target lesions; patients with at least one follow up assessment
Patients with at least one post baseline assessment – target lesion response for each patient
Key Takeaways
• Target tumor
reduction is seen in
>50% of patients
• Responses were
seen across disease
stages, including
complete responses
in patients with stage
IVM1b/c disease |
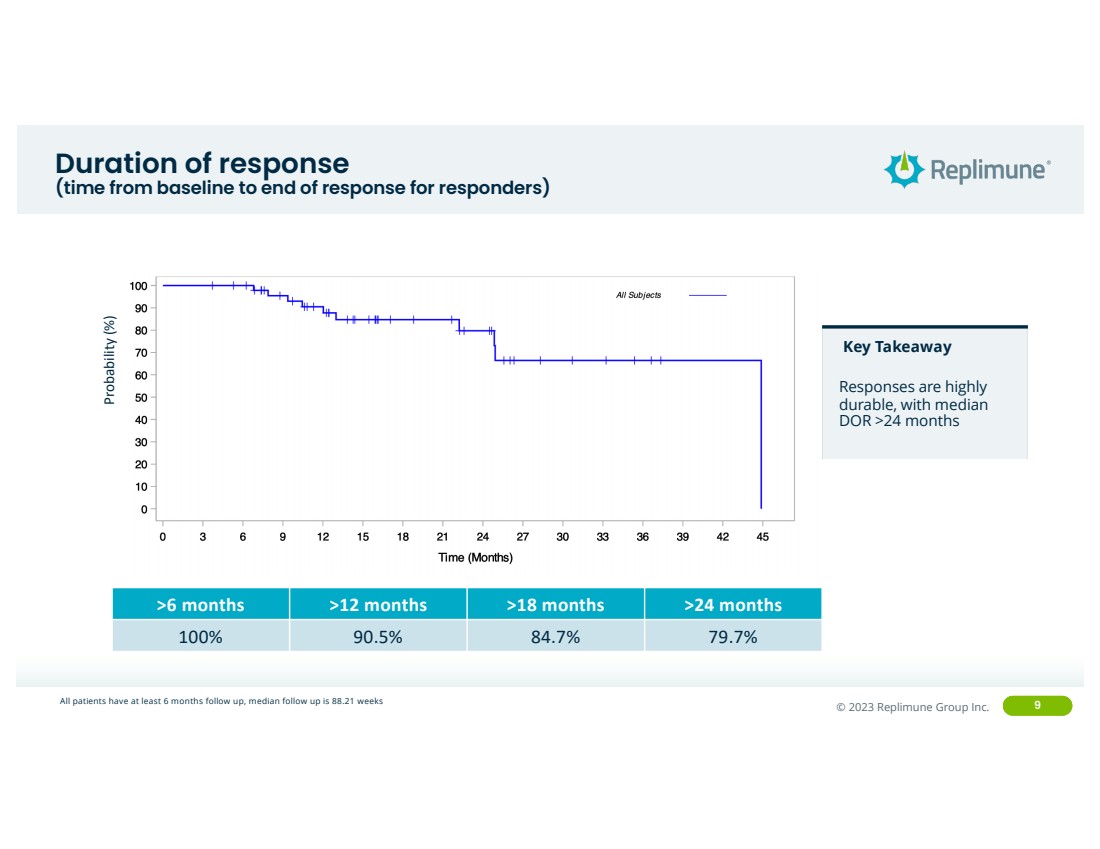
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 9
Duration of response
(time from baseline to end of response for responders) Probability (%)
Key Takeaway
Responses are highly
durable, with median
DOR >24 months
>6 months >12 months >18 months >24 months
100% 90.5% 84.7% 79.7%
All patients have at least 6 months follow up, median follow up is 88.21 weeks |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 10
Promising OS is seen across disease subsets, including
those with the greatest unmet need
Stage IIIb/IIIc/IVM1a vs Stage IV M1b/c/d Prior anti-CTLA-4+anti-PD1 vs prior anti-PD1 alone
All patients have at least 6 months follow up, median follow up is 88.21 weeks |
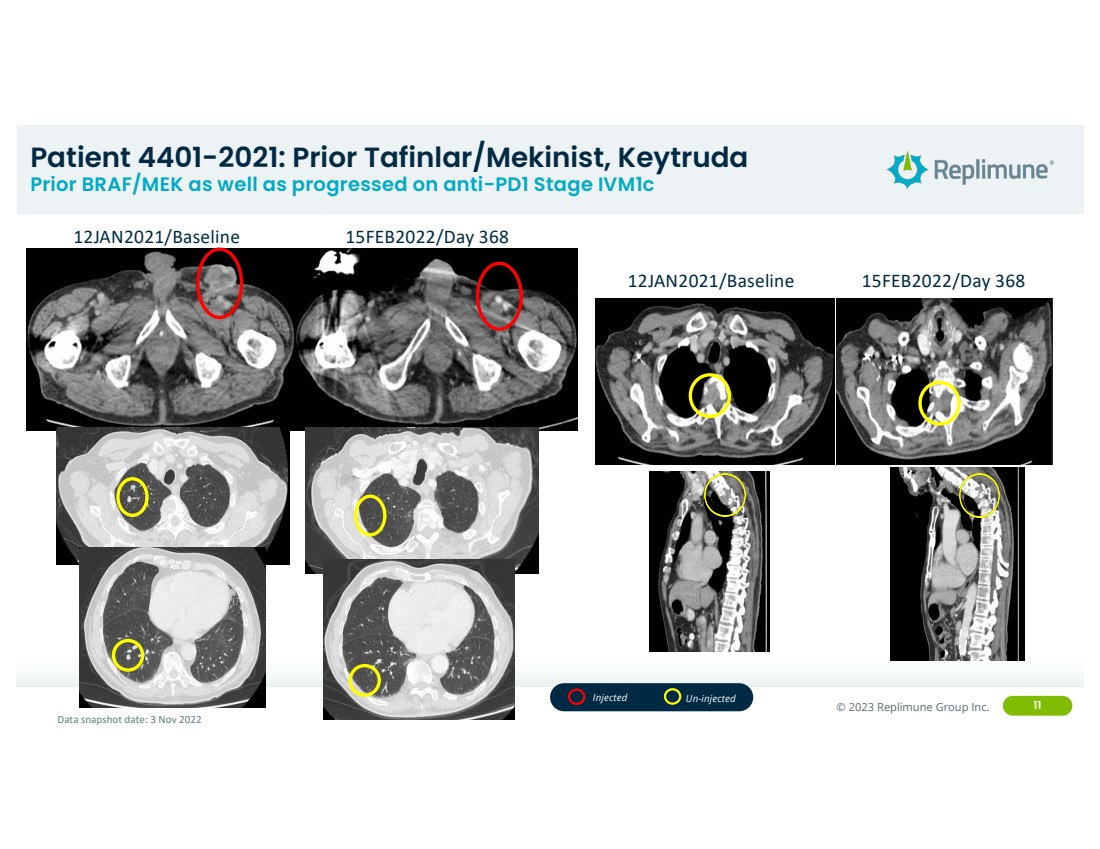
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 11
Patient 4401-2021: Prior Tafinlar/Mekinist, Keytruda Prior BRAF/MEK as well as progressed on anti-PD1 Stage IVM1c
12JAN2021/Baseline 15FEB2022/Day 368
12JAN2021/Baseline 15FEB2022/Day 368
Data snapshot date: 3 Nov 2022
Injected Un-injected |
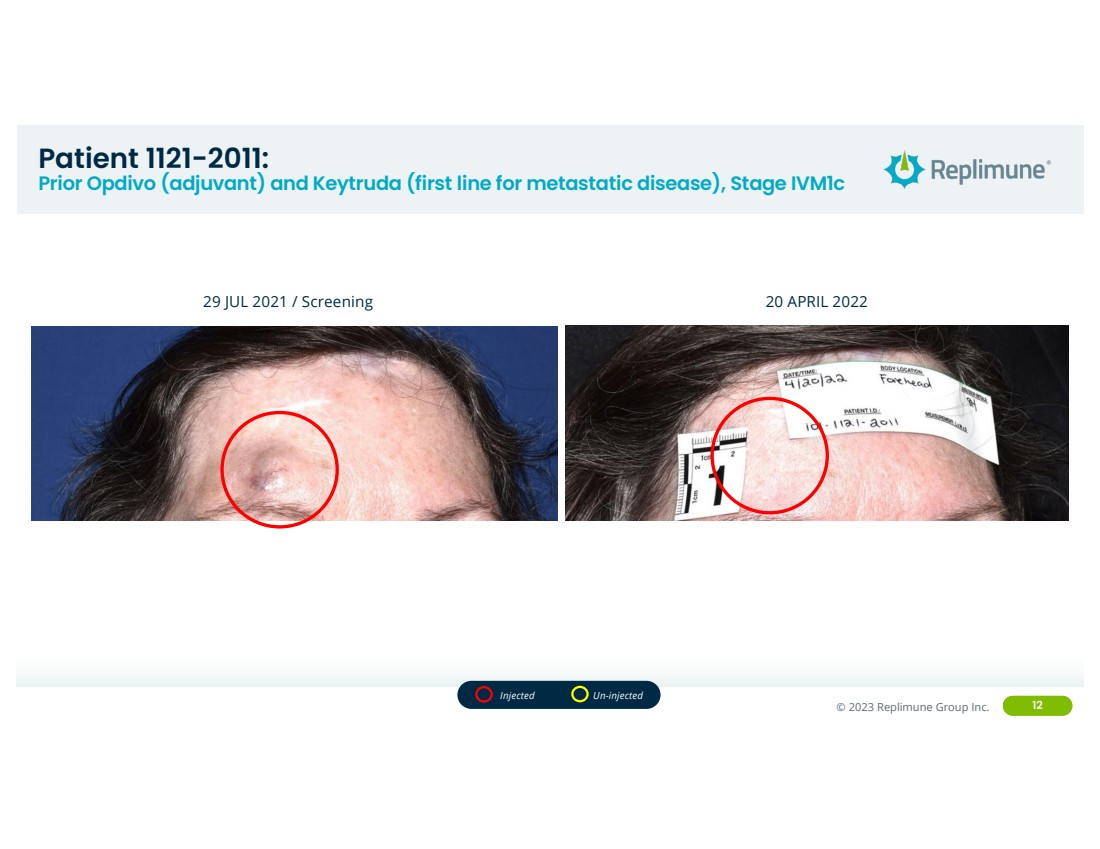
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 12
Patient 1121-2011:
Prior Opdivo (adjuvant) and Keytruda (first line for metastatic disease), Stage IVM1c
29 JUL 2021 / Screening 20 APRIL 2022
Injected Un-injected |
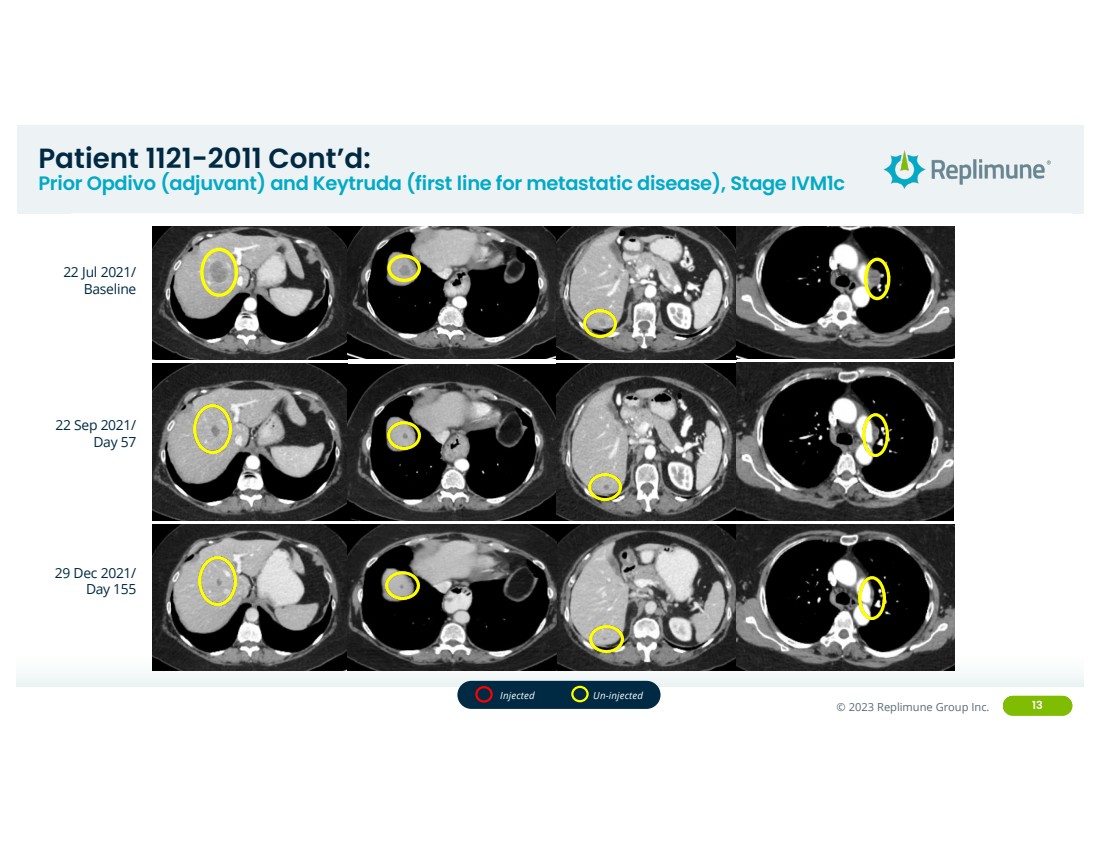
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 13
22 Jul 2021/
Baseline
22 Sep 2021/
Day 57
29 Dec 2021/
Day 155
Injected Un-injected
Patient 1121-2011 Cont’d:
Prior Opdivo (adjuvant) and Keytruda (first line for metastatic disease), Stage IVM1c |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 14
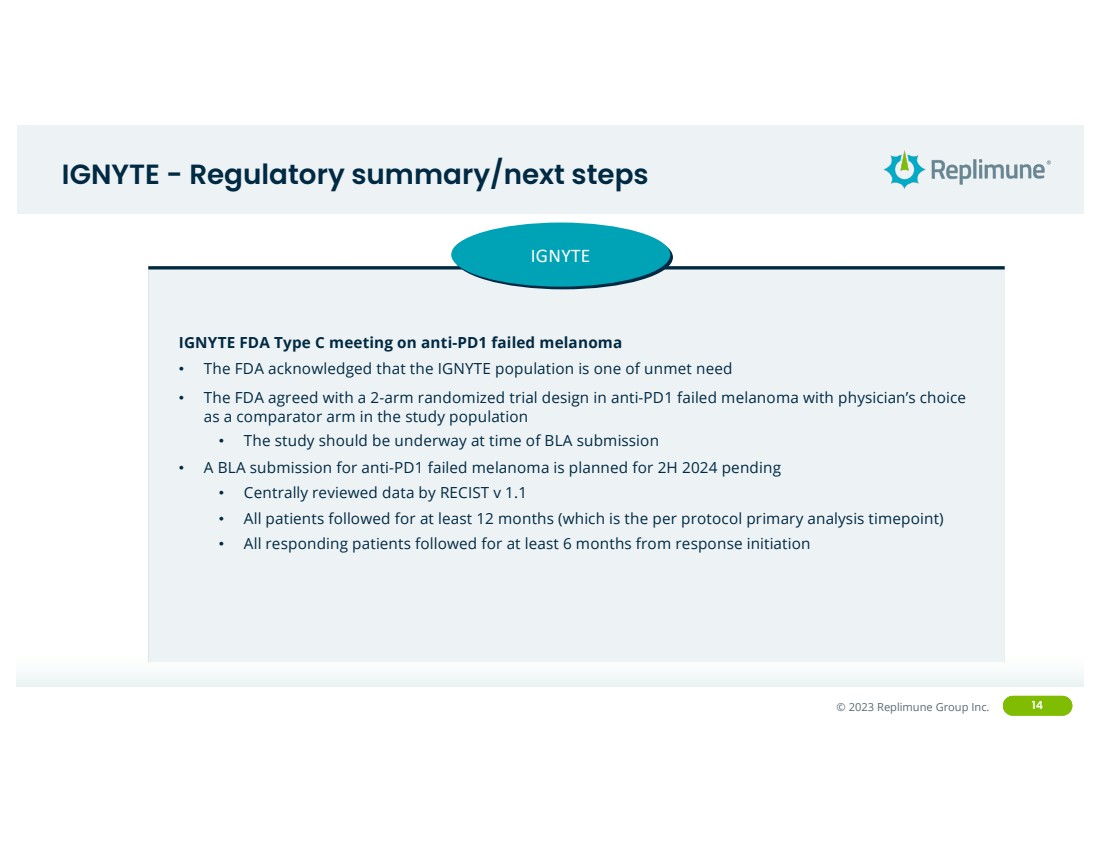
IGNYTE - Regulatory summary/next steps
IGNYTE
IGNYTE FDA Type C meeting on anti-PD1 failed melanoma
• The FDA acknowledged that the IGNYTE population is one of unmet need
• The FDA agreed with a 2-arm randomized trial design in anti-PD1 failed melanoma with physician’s choice
as a comparator arm in the study population
• The study should be underway at time of BLA submission
• A BLA submission for anti-PD1 failed melanoma is planned for 2H 2024 pending
• Centrally reviewed data by RECIST v 1.1
• All patients followed for at least 12 months (which is the per protocol primary analysis timepoint)
• All responding patients followed for at least 6 months from response initiation |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 15
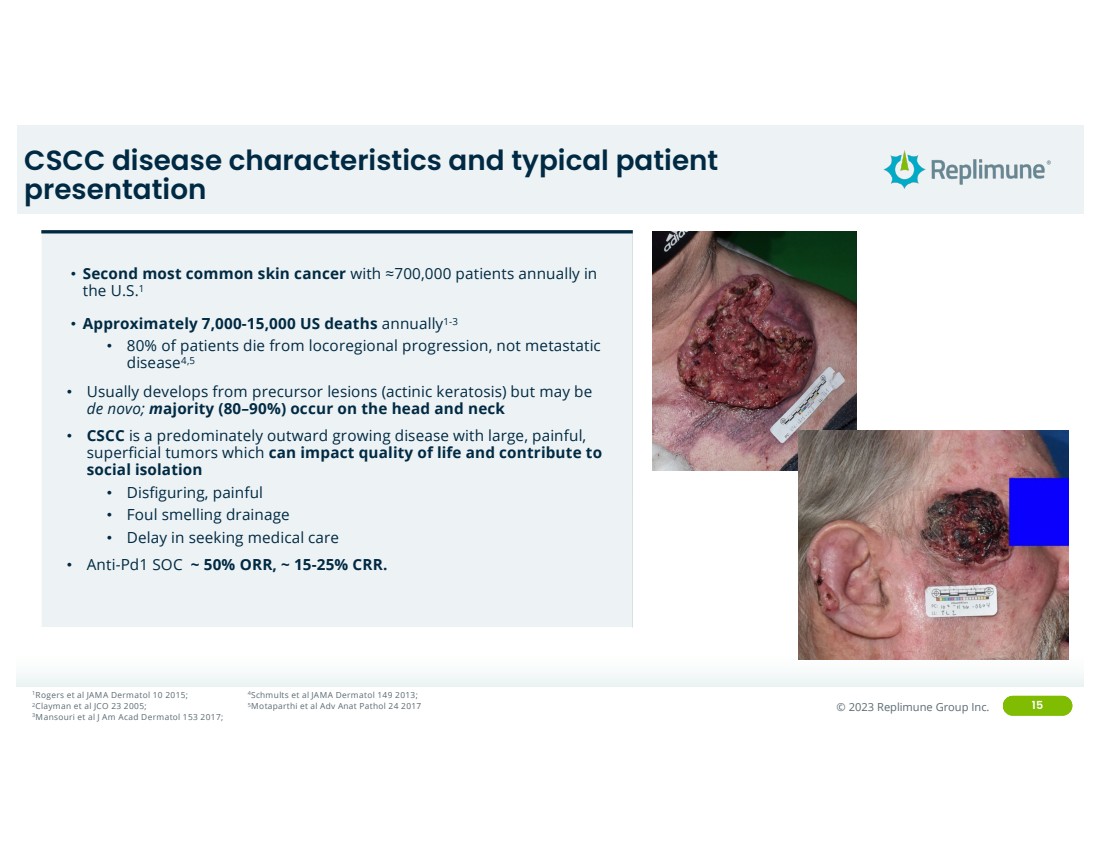
CSCC disease characteristics and typical patient
presentation
• Second most common skin cancer with ≈700,000 patients annually in
the U.S.1
• Approximately 7,000-15,000 US deaths annually1-3
• 80% of patients die from locoregional progression, not metastatic
disease4,5
• Usually develops from precursor lesions (actinic keratosis) but may be
de novo; majority (80–90%) occur on the head and neck
• CSCC is a predominately outward growing disease with large, painful,
superficial tumors which can impact quality of life and contribute to
social isolation
• Disfiguring, painful
• Foul smelling drainage
• Delay in seeking medical care
• Anti-Pd1 SOC ~ 50% ORR, ~ 15-25% CRR.
1Rogers et al JAMA Dermatol 10 2015;
2Clayman et al JCO 23 2005;
3Mansouri et al J Am Acad Dermatol 153 2017;
4Schmults et al JAMA Dermatol 149 2013; 5Motaparthi et al Adv Anat Pathol 24 2017 |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 16
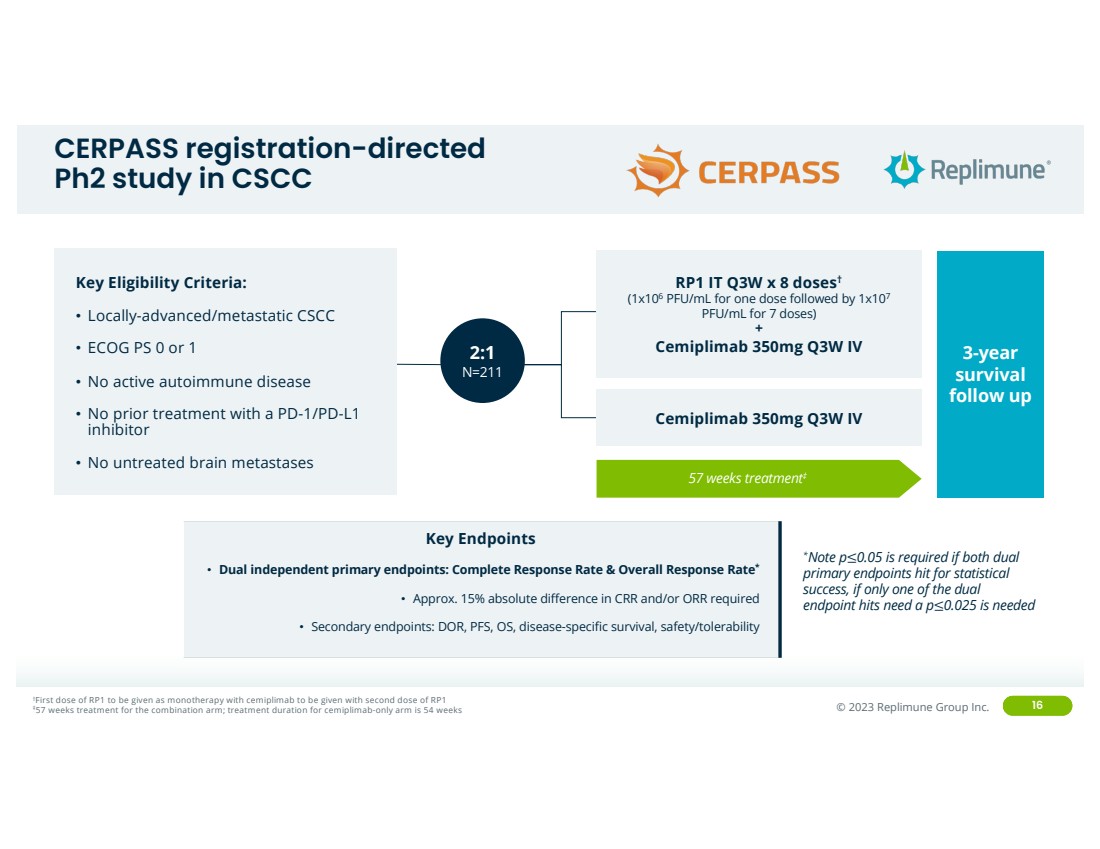
CERPASS registration-directed
Ph2 study in CSCC
Key Eligibility Criteria:
• Locally-advanced/metastatic CSCC
• ECOG PS 0 or 1
• No active autoimmune disease
• No prior treatment with a PD-1/PD-L1
inhibitor
• No untreated brain metastases
57 weeks treatment‡
3-year
survival
follow up
†First dose of RP1 to be given as monotherapy with cemiplimab to be given with second dose of RP1 ‡57 weeks treatment for the combination arm; treatment duration for cemiplimab-only arm is 54 weeks
Key Endpoints
• Dual independent primary endpoints: Complete Response Rate & Overall Response Rate*
• Approx. 15% absolute difference in CRR and/or ORR required
• Secondary endpoints: DOR, PFS, OS, disease-specific survival, safety/tolerability
2:1
N=211
RP1 IT Q3W x 8 doses†
(1x106 PFU/mL for one dose followed by 1x107
PFU/mL for 7 doses)
+
Cemiplimab 350mg Q3W IV
Cemiplimab 350mg Q3W IV
*Note p≤0.05 is required if both dual
primary endpoints hit for statistical
success, if only one of the dual
endpoint hits need a p≤0.025 is needed |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 17
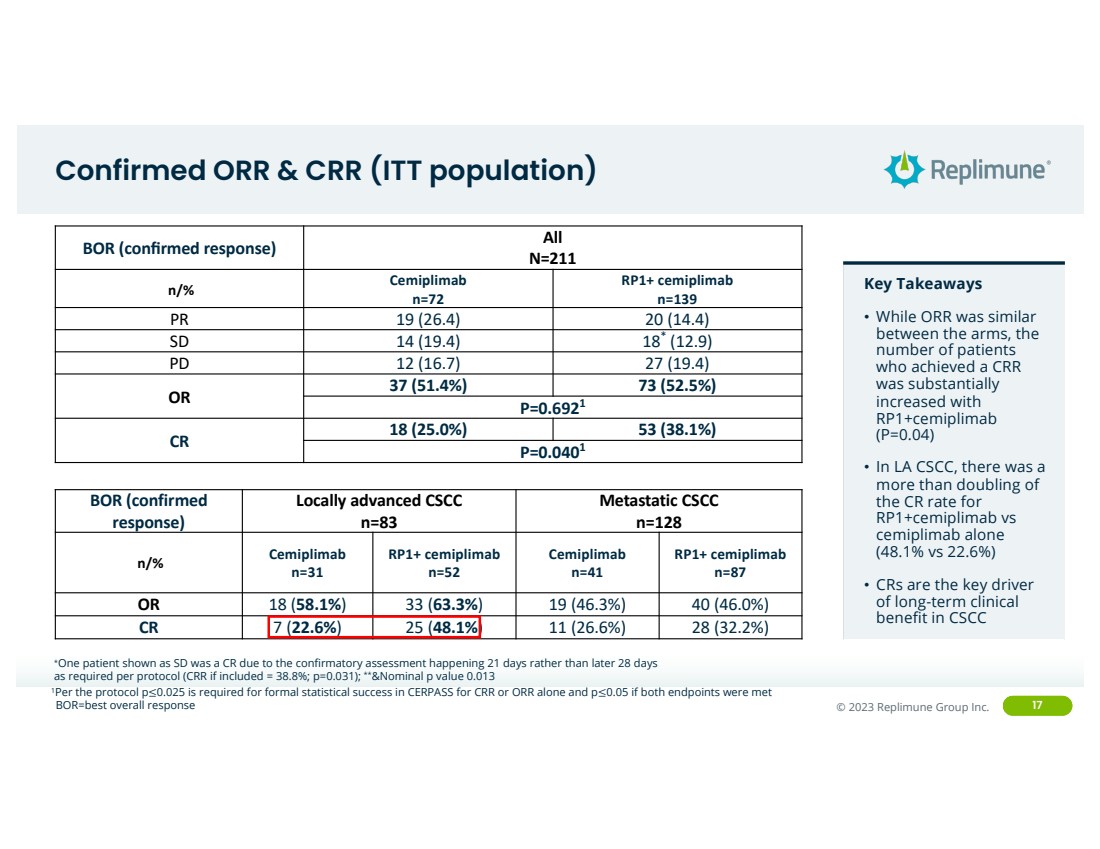
Confirmed ORR & CRR (ITT population)
Key Takeaways
• While ORR was similar
between the arms, the
number of patients
who achieved a CRR
was substantially
increased with
RP1+cemiplimab
(P=0.04)
• In LA CSCC, there was a
more than doubling of
the CR rate for
RP1+cemiplimab vs
cemiplimab alone
(48.1% vs 22.6%)
• CRs are the key driver
of long-term clinical
benefit in CSCC
BOR (confirmed response) All
N=211
n/% Cemiplimab
n=72
RP1+ cemiplimab
n=139
PR 19 (26.4) 20 (14.4)
SD 14 (19.4) 18* (12.9)
PD 12 (16.7) 27 (19.4)
OR 37 (51.4%) 73 (52.5%)
P=0.6921
CR 18 (25.0%) 53 (38.1%)
P=0.0401
*One patient shown as SD was a CR due to the confirmatory assessment happening 21 days rather than later 28 days
as required per protocol (CRR if included = 38.8%; p=0.031); **&Nominal p value 0.013
BOR=best overall response
1Per the protocol p≤0.025 is required for formal statistical success in CERPASS for CRR or ORR alone and p≤0.05 if both endpoints were met
BOR (confirmed
response)
Locally advanced CSCC
n=83
Metastatic CSCC
n=128
n/% Cemiplimab
n=31
RP1+ cemiplimab
n=52
Cemiplimab
n=41
RP1+ cemiplimab
n=87
OR 18 (58.1%) 33 (63.3%) 19 (46.3%) 40 (46.0%)
CR 7 (22.6%) 25 (48.1%) 11 (26.6%) 28 (32.2%) |
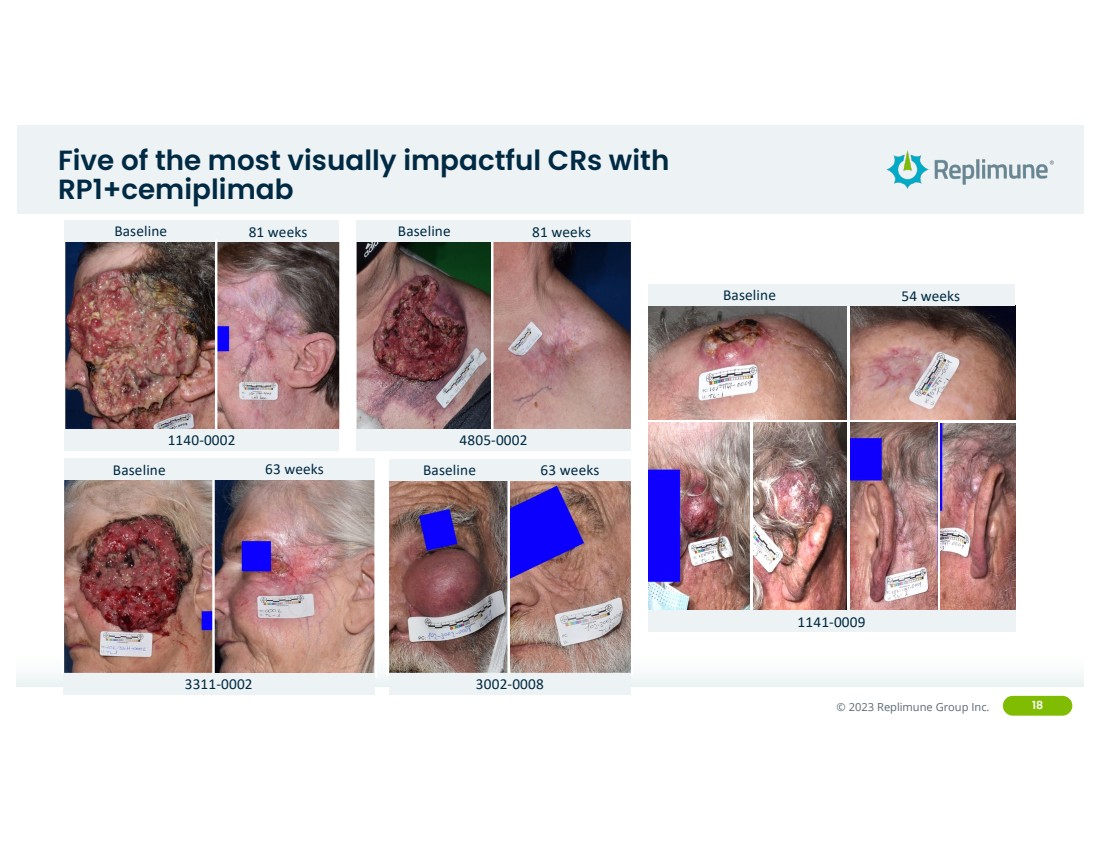
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 18
Five of the most visually impactful CRs with
RP1+cemiplimab
Baseline 81 weeks
4805-0002
Baseline 81 weeks
1140-0002
Baseline 63 weeks
3311-0002
Baseline 63 weeks
3002-0008
Baseline 54 weeks
1141-0009 |
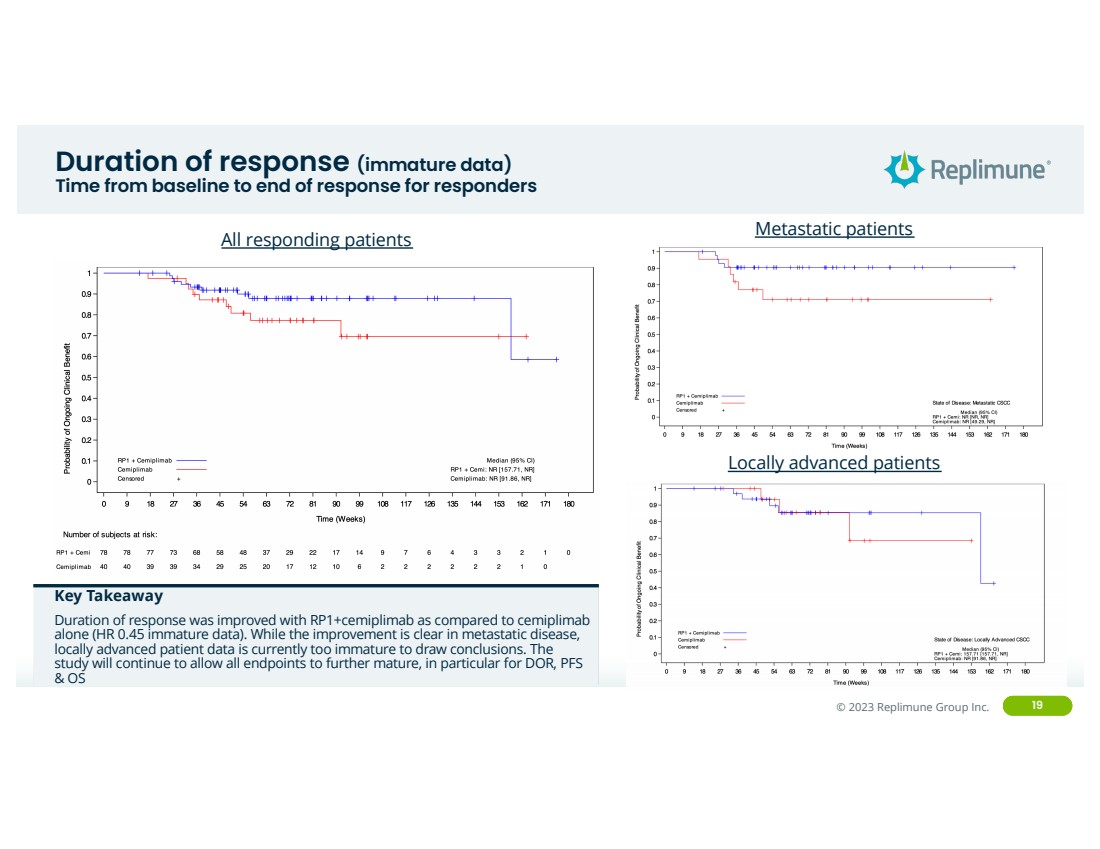
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 19
Duration of response (immature data)
Time from baseline to end of response for responders
All responding patients
Key Takeaway
Duration of response was improved with RP1+cemiplimab as compared to cemiplimab
alone (HR 0.45 immature data). While the improvement is clear in metastatic disease,
locally advanced patient data is currently too immature to draw conclusions. The
study will continue to allow all endpoints to further mature, in particular for DOR, PFS
& OS
Metastatic patients
Locally advanced patients |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 20
CERPASS - next steps
CERPASS
• CERPASS missed its primary endpoints while demonstrating treatment effects suggesting clinical benefit
• CR rate
• Duration of response
• All time-based endpoints are immature (DOR, PFS and OS) and will be followed to maturity
• Mature data required to determine whether any filing or compendia listing strategy is warranted |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 21
Additional unmet needs in CSCC/NMSC
ARTACUS STUDY
• Treatment of high risk immune compromised populations who develop skin cancers
• Anti-PD1 use can lead to loss of graft
• RP1 monotherapy; 35% ORR (N=23)
IGNYTE anti-PD1 failed NMSC
• No FDA approved options for anti-PD1-failed CSCC/NMSC; ~ 70% of treated patients still ultimately progress
• RP1 + nivo 30% ORR (N=30)
1Lam JKS, et al. Head Neck. 2018;40:985-992. 2Friman T, et al. Int J Cancer. 2022;150(11):1779-91. 3Marin-Acevedo et al Cancers 2023, 15(12), 3180 |

| AGENDA
© 2023 Replimune Group Inc.
RP1 Commercial Opportunity |
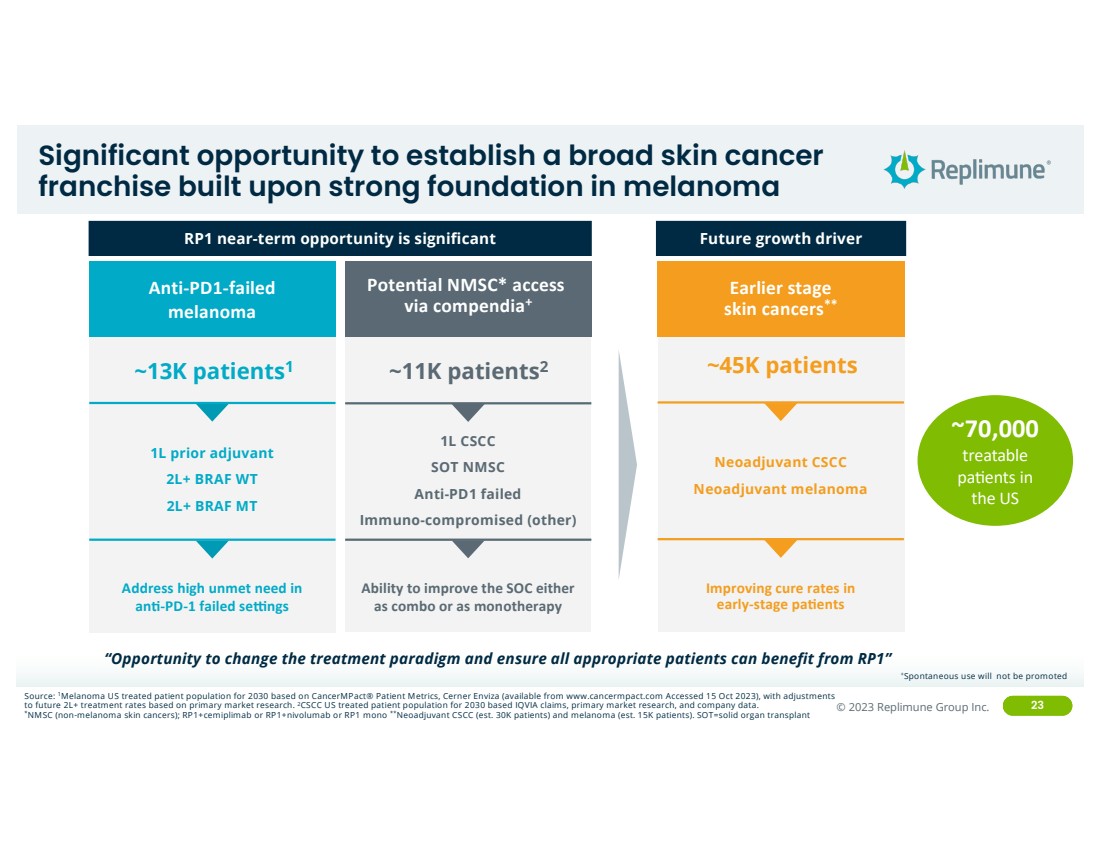
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 23
Significant opportunity to establish a broad skin cancer
franchise built upon strong foundation in melanoma
Future growth driver
~11K patients2 ~13K patients1
Ability to improve the SOC either
as combo or as monotherapy
Address high unmet need in
anG-PD-1 failed seKngs
Poten0al NMSC* access
via compendia+
1L prior adjuvant
2L+ BRAF WT
2L+ BRAF MT
~45K patients
Improving cure rates in
early-stage paGents
Earlier stage
skin cancers**
Anti-PD1-failed
melanoma
1L CSCC
SOT NMSC
Anti-PD1 failed
Immuno-compromised (other)
Neoadjuvant CSCC
Neoadjuvant melanoma
RP1 near-term opportunity is significant
Source: 1Melanoma US treated patient population for 2030 based on CancerMPact® Patient Metrics, Cerner Enviza (available from www.cancermpact.com Accessed 15 Oct 2023), with adjustments
to future 2L+ treatment rates based on primary market research. 2CSCC US treated patient population for 2030 based IQVIA claims, primary market research, and company data. *NMSC (non-melanoma skin cancers); RP1+cemiplimab or RP1+nivolumab or RP1 mono **Neoadjuvant CSCC (est. 30K patients) and melanoma (est. 15K patients). SOT=solid organ transplant
“Opportunity to change the treatment paradigm and ensure all appropriate patients can benefit from RP1”
~70,000
treatable
pa;ents in
the US
+Spontaneous use will not be promoted |

| © 2023 Replimune Group Inc. 24
Investment in manufacturing to support full
commercialization
• 63,000 square foot state-of-the-art facility for GMP manufacturing
• RP1-2 technology transfer from CMO successfully completed
• RP1 released to clinic post comparability analysis
• RP1 BLA consistency lot runs complete
• Scale expected to be sufficient to cover global commercialization
of all Replimune’s product candidates at full capacity
• Commercially attractive cost of goods & ‘off the shelf’ product
practicality
Commercial
scale in-house
manufacturing
established |

| © 2022 Replimune Group Inc. 25
AGENDA
© 2023 Replimune Group Inc.
RP2 update |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 26
RP2 leverages Replimune’s platform to express
anti-CTLA-4
• Anti-CTLA-4 prevents immune blockade at the APC / T cell interface
§ Anti-CTLA-4 is clinically validated; Ipilimumab, tremelimumab
• RP2 has shown durable mono-therapy responses in multiple immune insensitive tumor types
§ Salivary gland cancer
§ Chordoma
§ Uveal melanoma
§ Esophogeal cancer
• 30% ORR (N=17) in 2L uveal melanoma with impressive duration
§ Randomized control trial planned ; foundation of rare disease strategy
• Rare head and neck cancers
• Sarcomas
• HPV associated ; vulvar, anal |
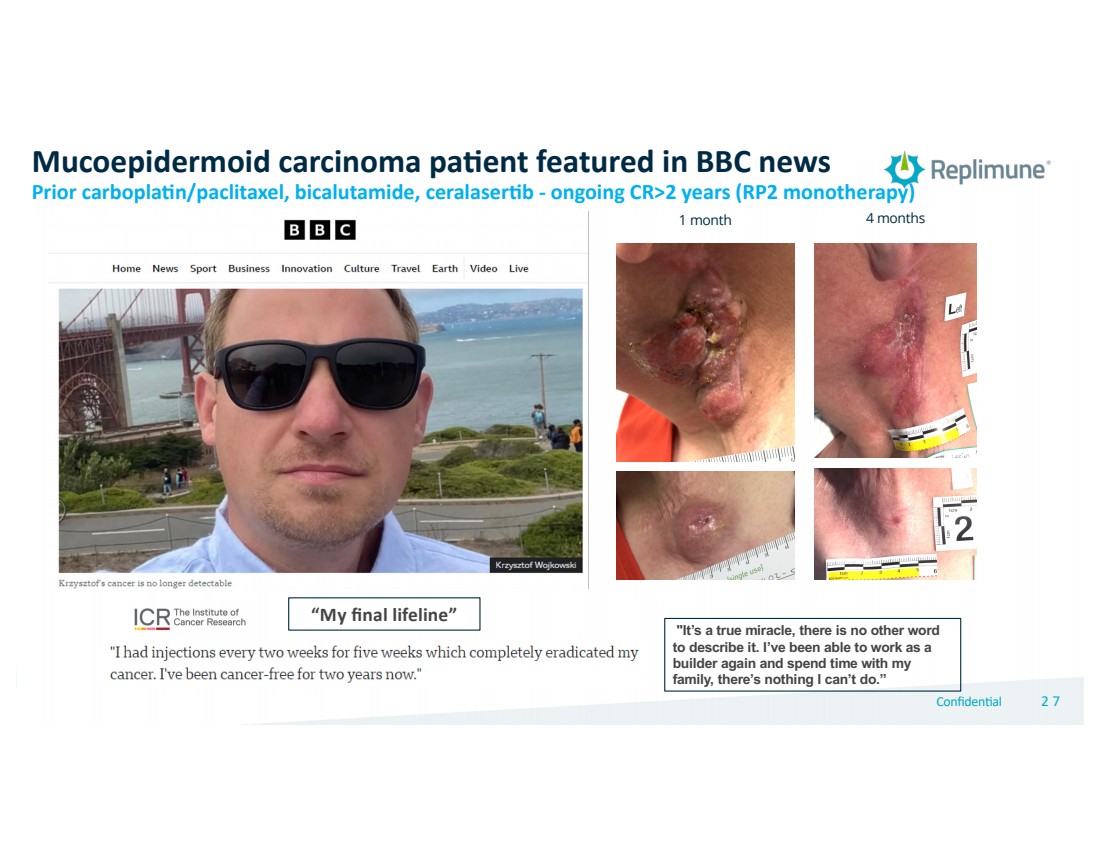
| © 2022 Replimune Group Inc. Confiden'al 272 7
"It’s a true miracle, there is no other word
to describe it. I’ve been able to work as a
builder again and spend time with my
family, there’s nothing I can’t do.”
“My final lifeline”
Mucoepidermoid carcinoma pa.ent featured in BBC news
Prior carbopla+n/paclitaxel, bicalutamide, ceralaser+b - ongoing CR>2 years (RP2 monotherapy)
1 month 4 months |
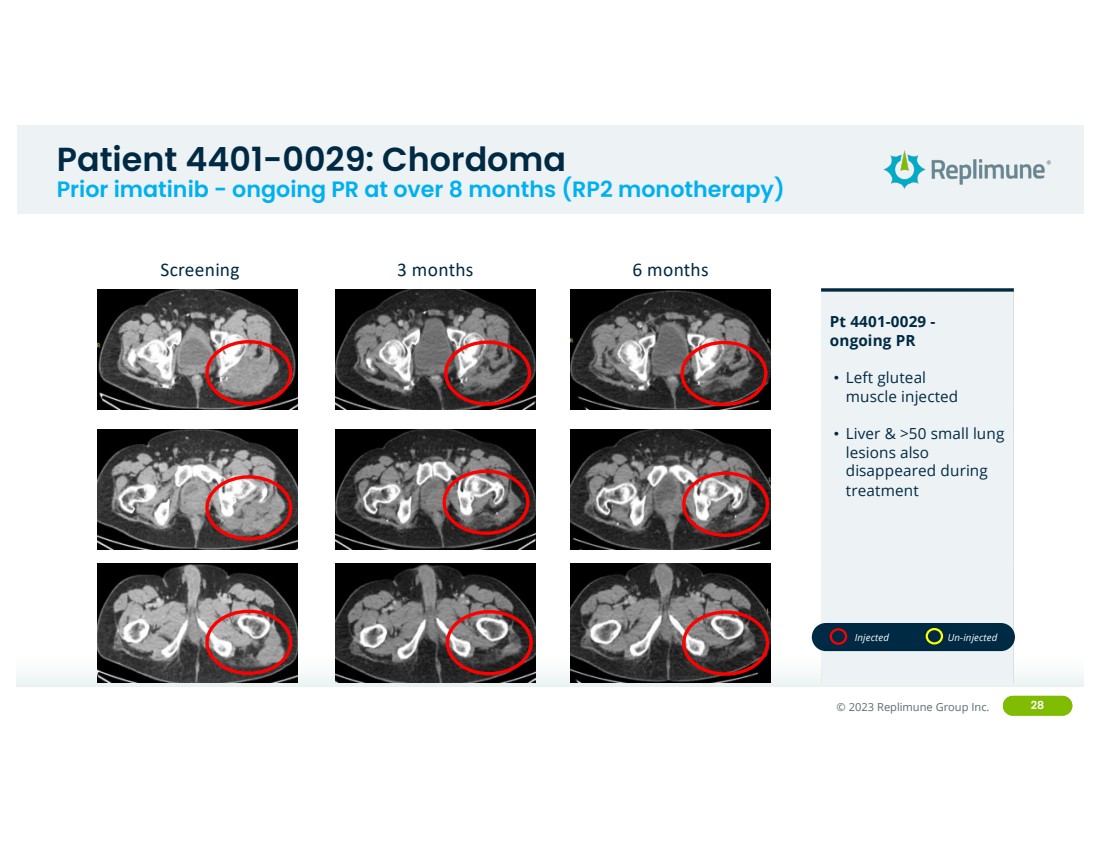
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 28
Patient 4401-0029: Chordoma
Prior imatinib - ongoing PR at over 8 months (RP2 monotherapy)
Pt 4401-0029 -
ongoing PR
• Left gluteal
muscle injected
• Liver & >50 small lung
lesions also
disappeared during
treatment
Screening 3 months 6 months
Injected Un-injected |

| © 2022 Replimune Group Inc. Confiden'al 292 9
Pa.ent 4401-0029: Chordoma
Prior ima+nib - ongoing PR at over 8 months (RP2 monotherapy)
Pt 4401-0029 -
ongoing PR
• Left gluteal muscle
lesion injected
• Liver & >50 small lung
lesions also
disappeared during
treatment
Injected Un-injected
Baseline 3 months 6 months |

| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 30
RP2 in uveal melanoma
1Carvajal RD et al. Br J Ophthalmol 2017; 2Nathan P et al. N Engl J Med. 2021;385(13):1196-1206; 3PelsterMS et al. J Clin Oncol. 2021;39(6):599-607; 4Lukzky J et al SMR 2022;
* Versus investigator’s choice, pembrolizumab, ipilimumab, or dacarbazine
• Ocular or “uveal” melanoma is a rare cancer with approx. 1,000
cases in the US per year1
• Originates from melanocytes and can occur in several eye locations
• The historic median OS is approx. 12 months1
• Uveal melanoma behaves quite differently from skin
melanoma
• Mostly metastasizes to the liver (approx. 70-90% of cases) and once this
occurs only about 10% of these patients survive beyond a year
• A difficult to treat tumor where CPIs have previously demonstrated limited
activity2,3,4
• Kimmtrak (tebentafusp) is the 1st approved agent in uveal melanoma in
HLA-A-02:01-positive adult patients (approx. 50% of the total population)*
• Unmet need for uveal melanoma patients remains high,
including improved efficacy/tolerability, effective options for
HLA negative patients, and options for Kimmtrak and anti-PD1
failed patients |
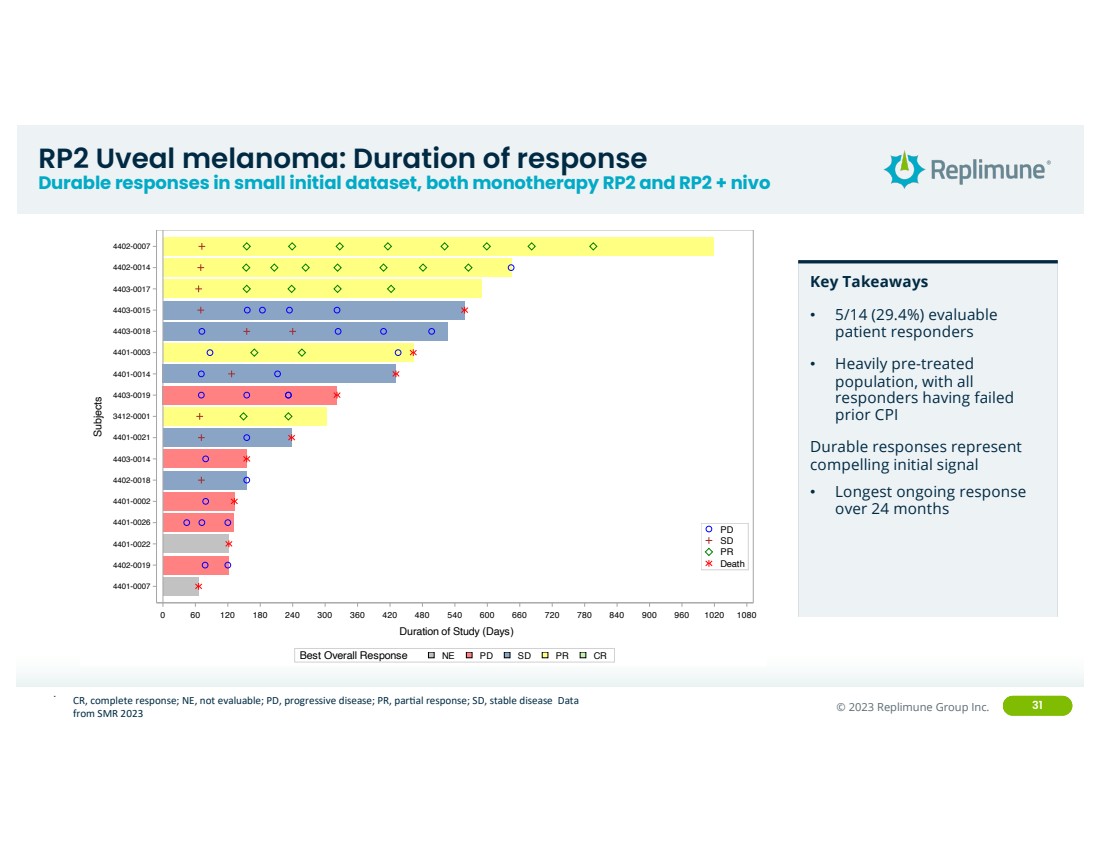
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 31
RP2 Uveal melanoma: Duration of response
Durable responses in small initial dataset, both monotherapy RP2 and RP2 + nivo
..
0 60 120 180 240 300 360 420 480 540 600 660 720 780 840 900 960 1020 1080
Duration of Study (Days)
4401-0007
4402-0019
4401-0022
4401-0026
4401-0002
4402-0018
4403-0014
4401-0021
3412-0001
4403-0019
4401-0014
4401-0003
4403-0018
4403-0015
4403-0017
4402-0014
4402-0007
Subjects
Death
PR
SD
PD
Best Overall Response NE PD SD PR CR
Replimune
Study No. RP2-001-18
Figure: Duration of Study (Ocular Melanoma: Uveal)
Key Takeaways
• 5/14 (29.4%) evaluable
patient responders
• Heavily pre-treated
population, with all
responders having failed
prior CPI
Durable responses represent
compelling initial signal
• Longest ongoing response
over 24 months
CR, complete response; NE, not evaluable; PD, progressive disease; PR, parEal response; SD, stable disease Data
from SMR 2023 |
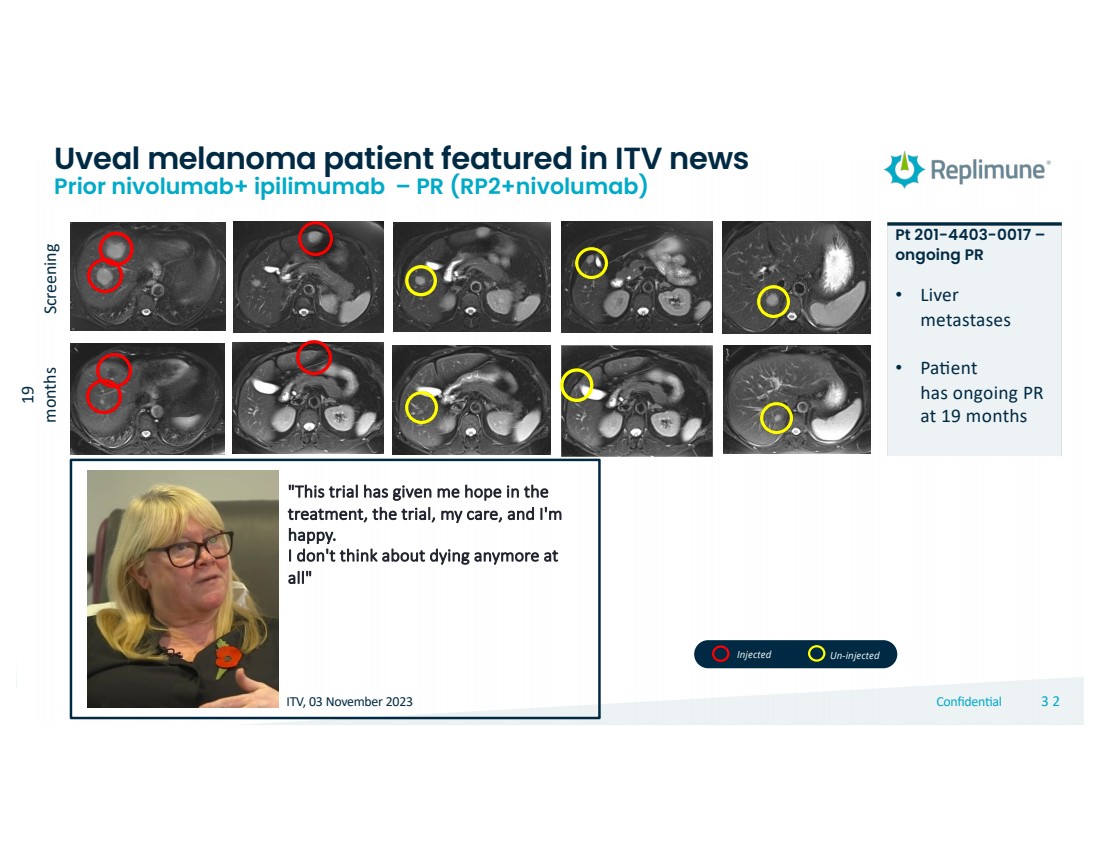
| © 2022 Replimune Group Inc. Confiden'al 323 2
Screening
19
months
Pt 201-4403-0017 –
ongoing PR
• Liver
metastases
• PaNent
has ongoing PR
at 19 months
Uveal melanoma patient featured in ITV news Prior nivolumab+ ipilimumab – PR (RP2+nivolumab)
"This trial has given me hope in the
treatment, the trial, my care, and I'm
happy.
I don't think about dying anymore at
all"
ITV, 03 November 2023
Injected Un-injected |
| ©© 2022 Replimune Group Inc. 2023 Replimune Group Inc. 33
RP1 in skin cancer Mid-stage pipeline Strong cash position
• Strong balance sheet; $466m (1) as
of 31 December 2023
• Cash Runway into H2 2026
• Initial snapshot of data from all 156
anti-PD1 failed melanoma patients
demonstrate that RP1+nivolumab
maintains transformative potential in
this high unmet need setting
• BLA submission planned for 2H
2024
• While CERPASSS missed its primary
endpoints at P>0.025, a clinically
meaningful benefit in CRR (P=0.04) and
DOR in CSCC was demonstrated
• Other skin cancer data in hard-to-treat
settings such as solid organ transplant
recipients & anti-PD1 failed melanoma
& NMSC demonstrate compelling
clinical activity
• Strong data with RP2 in uveal
melanoma
• Planning for a randomized controlled
pivotal study in uveal melanoma
underway
• Plan to investigate other rare
cancer opportunities
(1) Unaudited es,mate |

| MISSION
To enable tumor directed oncolytic
immunotherapy (TDOI) to become a
cornerstone in the treatment of cancer
VISION
To deliver transformational results for patients across
cancers using tumor directed oncolytic
immunotherapy to induce a powerful and durable
systemic anti-tumor immune response resulting in
quality survival and a chance for a cure
THANK YOU |
v3.23.4
Cover
|
Jan. 08, 2024 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Jan. 08, 2024
|
| Entity File Number |
001-38596
|
| Entity Registrant Name |
REPLIMUNE GROUP, INC.
|
| Entity Central Index Key |
0001737953
|
| Entity Tax Identification Number |
82-2082553
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
500 Unicorn Park Drive
|
| Entity Address, Address Line Two |
Suite 303
|
| Entity Address, City or Town |
Woburn
|
| Entity Address, State or Province |
MA
|
| Entity Address, Postal Zip Code |
01801
|
| City Area Code |
781
|
| Local Phone Number |
222-9600
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common Stock, par value $0.001 per share
|
| Trading Symbol |
REPL
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
true
|
| Elected Not To Use the Extended Transition Period |
true
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Replimune (NASDAQ:REPL)
Historical Stock Chart
Von Mai 2024 bis Jun 2024

Replimune (NASDAQ:REPL)
Historical Stock Chart
Von Jun 2023 bis Jun 2024
