AN2 Therapeutics Signs Grant Agreement to Discover Novel, Boron Based Therapies for Tuberculosis and Malaria
26 September 2023 - 1:00PM
Business Wire
Grant from Bill & Melinda Gates
Foundation
AN2 Therapeutics, Inc. (Nasdaq: ANTX), a clinical-stage
biopharmaceutical company focused on developing treatments for
rare, chronic, and serious infectious diseases with high unmet
needs, today announced that it has received a research grant from
the Bill & Melinda Gates Foundation to discover novel, boron
containing small molecules for the treatment of tuberculosis and
malaria. Funding from the Gates Foundation will support the
discovery of novel inhibitors of aminoacyl-tRNA synthetases using
AN2’s proprietary boron chemistry platform. AN2’s boron chemistry
has the potential to address biological targets for tuberculosis
and malaria that build on the scientific expertise of several AN2
scientists that were involved in discovering novel leucyl-tRNA
synthetase inhibitors including epetraborole, tavaborole, as well
as ganfeborole, which is currently in development for tuberculosis.
AN2’s boron chemistry platform has demonstrated success against
leucyl- tRNA synthetase, a target that has proven difficult to
inhibit using traditional medicinal chemistry approaches.
“Novel drugs that combat drug resistance and shorten therapy are
urgently needed to reduce the impact of tuberculosis and malaria,
diseases that are responsible for over 2 million deaths per year
and disproportionately affect people in low- and middle-income
countries,” said Eric Easom, Co-Founder, President and Chief
Executive Officer of AN2 Therapeutics. “Our boron chemistry
platform and team have a proven track record of delivering novel
compounds targeting serious unmet needs in infectious diseases and
global health that can be game-changers, and we are excited to
engage in this important work. This grant underscores our
commitment to address areas of high unmet need in global health and
capture synergies with our existing research programs using
non-dilutive funding.”
About AN2 Therapeutics, Inc.
AN2 Therapeutics, Inc. is a clinical-stage biopharmaceutical
company developing treatments for rare, chronic, and serious
infectious diseases with high unmet needs. Our initial candidate is
epetraborole, which we are studying as a once-daily, oral treatment
with a novel mechanism of action for patients with nontuberculous
mycobacteria (NTM) lung disease, a rare, chronic, and progressive
infectious disease caused by bacteria known as mycobacteria, that
leads to irreversible lung damage and can be fatal. For more
information, please visit our website at
www.an2therapeutics.com.
Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. Forward-looking statements expressed or implied in this press
release include, but are not limited to, statements regarding: the
timing, progress, and anticipated results AN2's anticipated
progress, business plans, business strategy and planned research
efforts; and other statements that are not historical fact. These
statements are based on AN2's current estimates, expectations,
plans, objectives, and intentions, are not guarantees of future
performance and inherently involve significant risks and
uncertainties. Actual results and the timing of events could differ
materially from those anticipated in such forward-looking
statements as a result of these risks and uncertainties, which
include, but are not limited to, risks and uncertainties related
to: the continuing effects of the COVID-19 pandemic; macroeconomic
conditions; early clinical, preliminary or expected results;
significant adverse events, toxicities or other undesirable side
effects associated with AN2's product candidate; the significant
uncertainty associated with AN2's product candidate ever receiving
any regulatory approvals; AN2's ability to obtain, maintain or
protect intellectual property rights related to its current and
future product candidates; implementation of AN2's strategic plans
for its business and current and future product candidates; the
sufficiency of AN2's capital resources and need for additional
capital to achieve its goals; and other risks, including those
described under the heading “Risk Factors” in AN2's reports to be
filed with the SEC, including AN2’s Report on Form 10-Q for the
quarter ended June 30, 2023. These filings, when available, are
available on the investor relations section of our website at
investor.an2therapeutics.com and on the SEC's website at
www.sec.gov. Forward-looking statements contained in this press
release are made as of this date, and AN2 undertakes no duty to
update such information except as required under applicable
law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230926590651/en/
Lucy O. Day Chief Financial Officer
l.day@an2therapeutics.com
Anne Bowdidge Investor Relations
abowdidge@an2therapeutics.com
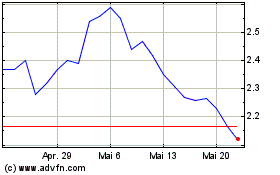
AN2 Therapeutics (NASDAQ:ANTX)
Historical Stock Chart
Von Apr 2024 bis Mai 2024
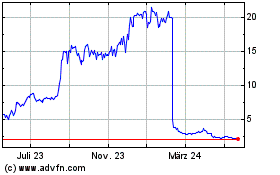
AN2 Therapeutics (NASDAQ:ANTX)
Historical Stock Chart
Von Mai 2023 bis Mai 2024