Spectral Medical Hosts Successful Investigator Meeting; Expected to Help Bolster Enrollment Activities
24 Mai 2023 - 2:00PM

Spectral Medical Inc. (“Spectral” or the “Company”) (TSX:
EDT), a late-stage theranostic company advancing
therapeutic options for sepsis and septic shock, today reported
hosting an investigator meeting in Charlotte, North Carolina on May
17 - 18, 2023. The in-person meeting was attended by principal
investigators (“PI”) and clinical research coordinators (“CRC”)
from all existing and new trial sites, as well as the Company’s new
CRO, Beaufort. The meeting is expected to help bolster ongoing
enrollment activities related to the Tigris trial, the Company’s
ongoing follow-on study designed to build on knowledge gained from
the earlier EUPHRATES trial, which evaluated the use of Polymyxin B
Hemoperfusion (“PMX”) in a randomized controlled trial of adults
treated for endotoxemia and septic shock. The Tigris trial end
point is a reduction in the 28-day mortality in subjects with
septic shock using the PMX hemoperfusion cartridge versus standard
of care.
Attendance Summary:
- Approximately 70 in-person
attendees.
- Tigris trial sites represented: 15
existing sites and six new or pending sites with site initiation
visits conducted at the meeting for multiple sites.
- Beaufort, Spectral’s new CRO: four
representatives, including the project lead and head of quality
assurance.
- Baxter, Spectral’s strategic
commercial partner: four representatives, including clinical and
marketing teams.
- The Balancing Act, a premier
morning show airing on Lifetime, conducting interviews and filming
discussions.
“We are pleased with the overwhelming success of
our investigator meeting held last week,” commented Chris Seto,
Chief Executive Officer of Spectral Medical. “During the meeting,
participants shared past challenges, as well as best practices and
solutions to overcome recruitment hurdles. Many attendees noted
that all ICU trials are having trouble with recruitment. However,
we now have significant recruitment efforts underway and are taking
steps to accelerate enrollment, including the addition of multiple
new sites, and a new CRO that many of these trial sites have worked
with in the past. Given this progress, the investigator meeting was
an important step, providing us the opportunity to reaffirm our
unwavering commitment to the trial and advancing PMX to FDA
submission and ultimate approval. Participants showed strong
support, as well as shared in our eagerness to bring PMX to
market.”
Dr. John Kellum, Chief Medical Officer of
Spectral Medical, stated, “Tigris is targeting a very important
unmet need in the medical field, as endotoxemic septic shock is the
most malignant form of sepsis. We have actively undertaken specific
initiatives to assist in the acceleration of recruitment and
enrollment. These efforts appear to be paying off as two patients
were just randomized into the trial on May 22 and May 23, bringing
the total enrolment to 60 patients. We continue to close in on
our interim enrollment target of 90 patients. Interim enrollment
would be a significant milestone as Baxter will have the
opportunity to view the data as well as submit a milestone payment
to Spectral. We were encouraged by the outlook of the trial and
felt a high level of support and enthusiasm from participants at
the investigator meeting. We look forward to reaching and reporting
additional milestones in the near-term.”
About Spectral
Spectral is a Phase 3 company seeking U.S. FDA
approval for its unique product for the treatment of patients with
septic shock, Toraymyxin™ (“PMX”). PMX is a therapeutic
hemoperfusion device that removes endotoxin, which can cause
sepsis, from the bloodstream and is guided by the Company’s
Endotoxin Activity Assay (EAA™), the only FDA cleared diagnostic
for the risk of developing sepsis.
PMX is approved for therapeutic use in Japan and
Europe, and has been used safely and effectively on more than
340,000 patients to date. In March 2009, Spectral obtained the
exclusive development and commercial rights in the U.S. for PMX,
and in November 2010, signed an exclusive distribution agreement
for this product in Canada. In July 2022, the U.S. FDA granted
Breakthrough Device Designation for PMX for the treatment of
endotoxemic septic shock. Approximately 330,000 patients are
diagnosed with septic shock in North America each year.
Spectral is listed on the Toronto Stock Exchange
under the symbol EDT. For more information please visit
www.spectraldx.com.
Forward-looking statement
Information in this news release that is not
current or historical factual information may constitute
forward-looking information within the meaning of securities laws.
Implicit in this information, particularly in respect of the future
outlook of Spectral and anticipated events or results, are
assumptions based on beliefs of Spectral's senior management as
well as information currently available to it. While these
assumptions were considered reasonable by Spectral at the time of
preparation, they may prove to be incorrect. Readers are cautioned
that actual results are subject to a number of risks and
uncertainties, including the availability of funds and resources to
pursue R&D projects, the successful and timely completion of
clinical studies, the ability of Spectral to take advantage of
business opportunities in the biomedical industry, the granting of
necessary approvals by regulatory authorities as well as general
economic, market and business conditions, and could differ
materially from what is currently expected.
The TSX has not reviewed and does not accept responsibility for
the adequacy or accuracy of this statement.
For further information, please contact:
|
Ali Mahdavi |
David Waldman/Natalya Rudman |
Blair McInnis |
| Capital Markets & Investor
Relations |
US Investor Relations |
CFO |
| Spinnaker Capital Markets
Inc. |
Crescendo Communications,
LLC |
Spectral Medical Inc. |
| 416-962-3300 |
212-671-1020 |
416-626-3233 |
|
am@spinnakercmi.com |
edt@crescendo-ir.com |
bmcinnis@spectraldx.com |
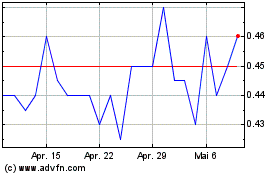
Spectral Medical (TSX:EDT)
Historical Stock Chart
Von Apr 2024 bis Mai 2024
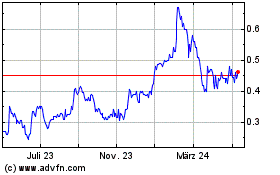
Spectral Medical (TSX:EDT)
Historical Stock Chart
Von Mai 2023 bis Mai 2024