- An adult patient recently received the implant, produced on the
Desktop Health 3D-Bioplotter® Manufacturer Series
- This project is highlighted in a new video showcasing how the
Hannover Medical School’s ENT clinic uses the premium bioprinter at
Team.DM.com/MHH
- The 3D-Bioplotter has now been used for two separate in-patient
applications in Germany and the United States, as bioprinting
applications advance into a new phase of in-patient
applications
Desktop Health — the trusted production-grade medical 3D
printing brand of Desktop Metal, Inc. (NYSE: DM) — today announced
that the ENT clinic at Hannover Medical School (MHH) in Germany has
used its 3D-Bioplotter Manufacturer Series to produce a customized
ear implant with drug release benefits for implantation in an adult
patient.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20231031818640/en/
The ENT clinic at Hannover Medical School
(MHH) in Germany has used its Desktop Health 3D-Bioplotter to
produce a customized ear implant with drug release benefits for
implantation in an adult patient. A growing number of parts
bioprinted on the 3D-Bioplotter are being used in patients. (Photo:
Business Wire)
The ENT clinic at MHH is the first in the world to use the
Desktop Health 3D-Bioplotter to customize an ear implant for a
patient's individual anatomy, print it with integrated
pharmaceutical ingredients, and implant it.
“We have thus opened the door to a new type of pioneering
patient care," said ENT clinic director Prof. Prof. h.c. Thomas
Lenarz, MD.
“Our entire team celebrates this important accomplishment by the
Hannover Medical School ENT clinic, which has been years in the
making,” said Ric Fulop, Founder and CEO of Desktop Metal. MHH
purchased its 3D-Bioplotter in 2020 to develop this and other
applications.
Fulop noted that bioprinting is entering a new phase. “The
benefits of 3D bioprinting are just beginning to move into actual
patients, and we celebrate this important shift in medicine that
MHH and other medical innovators are driving forward,” Fulop said.
“In recent months, we’ve seen the first products developed over
many years on the 3D-Bioplotter moving into commercial and patient
applications. We celebrate these important milestones, and what
they mean for personalized patient care.”
Earlier this month, Chicago-based Dimension Inx announced that
its CMFlex™ synthetic bone graft product has been surgically
implanted in two cases. This product — the first 3D printed
regenerative bone graft product cleared by the FDA — was developed
and is manufactured on the 3D-Bioplotter Manufacturer Series.
Details about the Ear Implant
The MHH patient who received the implant suffered from recurrent
narrowing of the auditory canal, which had been treated surgically
several times without showing the desired success. The new implant
serves a stent function, but also delivers ingredients designed to
accelerate healing.
“The first follow-up has already been very promising,” said PD
Dr. Verena Scheper, whose team from the Otolaryngology Clinic’s
Inner Ear Pharmacology Department designed the implant and
manufactured it using the 3D-Bioplotter Manufacturing Series.
The MHH ENT Clinic is internationally renowned for the world's
largest cochlear implant program for the care of hearing-impaired
patients. To date, the clinic has provided more than 11,111
affected persons with a cochlear implant. Other areas of focus
include the provision of hearing aids and their further
development, the early detection of childhood hearing loss, and the
diagnosis and treatment of sensorineural hearing loss, including
tinnitus. The treatment of patients is based on the close
cooperation between the ENT clinic, the German Hearing Center
Hannover (DHZ), acousticians, product manufacturers and scientists
up to the development of new medical products.
The Desktop Health 3D-Bioplotter is a robust, premium bioprinter
renowned for its high accuracy and repeatability. It’s also the
world’s most cited and researched bioprinter in peer-reviewed
scientific and medical journals, with more than 2,490 citations and
more than 640 research papers directly produced with the system. To
learn more, visit TeamDM.com/biopapers.
About Desktop Metal
Desktop Metal (NYSE:DM) is driving Additive Manufacturing
2.0, a new era of on-demand, digital mass production of industrial,
medical, and consumer products. Our innovative 3D printers,
materials, and software deliver the speed, cost, and part quality
required for this transformation. We’re the original inventors and
world leaders of the 3D printing methods we believe will empower
this shift, binder jetting and digital light processing. Today, our
systems print metal, polymer, sand and other ceramics, as well as
foam and recycled wood. Manufacturers use our technology worldwide
to save time and money, reduce waste, increase flexibility, and
produce designs that solve the world’s toughest problems and enable
once-impossible innovations. Learn more about Desktop Metal and our
#TeamDM brands at www.desktopmetal.com.
Forward-Looking Statements
This press release contains certain forward-looking statements
within the meaning of the federal securities laws. Forward-looking
statements generally are identified by the words “believe,”
“project,” “expect,” “anticipate,” “estimate,” “intend,”
“strategy,” “future,” “opportunity,” “plan,” “may,” “should,”
“will,” “would,” “will be,” “will continue,” “will likely result,”
and similar expressions. Forward-looking statements are
predictions, projections and other statements about future events
that are based on current expectations and assumptions and, as a
result, are subject to risks and uncertainties. Many factors could
cause actual future events to differ materially from the
forward-looking statements in this document, including but not
limited to the risks and uncertainties set forth in Desktop Metal,
Inc.'s filings with the U.S. Securities and Exchange Commission.
Forward-looking statements speak only as of the date they are made.
Readers are cautioned not to put undue reliance on forward-looking
statements, and Desktop Metal, Inc. assumes no obligation and does
not intend to update or revise these forward-looking statements,
whether as a result of new information, future events, or
otherwise.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231031818640/en/
Media and Investor Relations: Sarah Webster
sarahwebster@desktopmetal.com (313) 715-6988
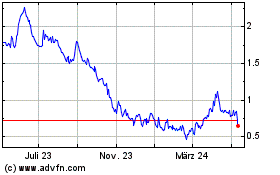
Desktop Metal (NYSE:DM)
Historical Stock Chart
Von Apr 2024 bis Mai 2024
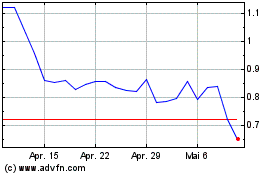
Desktop Metal (NYSE:DM)
Historical Stock Chart
Von Mai 2023 bis Mai 2024