NEW HAVEN, Conn., Dec. 3, 2018
/PRNewswire/ -- Biohaven Pharmaceutical Holding Company Ltd. (NYSE:
BHVN) today announced positive topline results from a randomized,
controlled Phase 3 clinical trial (BHV3000-303 or Study 303)
evaluating the efficacy and safety of its
Zydis® orally dissolving tablet
(ODT) formulation of rimegepant, an oral calcitonin gene-related
peptide (CGRP) receptor antagonist, for the acute treatment of
migraine. In Study 303, rimegepant Zydis ODT statistically
differentiated from placebo on the two co-primary endpoints as well
as the first 21 consecutive primary and secondary outcome measures
that were prespecified in hierarchical testing. Consistent with the
two previous Phase 3 clinical trials, Study 303 met its co-primary
registrational endpoints of pain freedom and freedom from most
bothersome symptom (MBS) at 2 hours using a single dose (Table
1). Importantly, patients treated with the rimegepant Zydis ODT
formulation began to numerically separate from placebo on pain
relief as early as 15 minutes, and this difference was
statistically significant at 60 minutes (p < 0.0001) (Figure
1). Additionally, a significantly greater percentage of
patients treated with rimegepant Zydis ODT returned to normal
functioning by 60 minutes as compared to placebo (p =
0.0025). Lasting clinical benefit was observed through 48
hours after a single dose of rimegepant on freedom from pain (p
< 0.0001), pain relief (p < 0.0001), freedom from the most
bothersome symptom (p = 0.0018), and freedom from functional
disability (p < 0.0001). Superiority over placebo was also
demonstrated in multiple other secondary endpoints. The vast
majority of patients treated with rimegepant Zydis ODT (85%)
did not use any rescue medications.
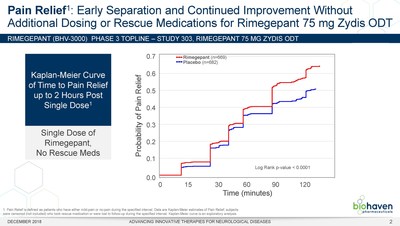
Table 1: Met Co-Primary Endpoints of Pain Freedom
& Freedom from Most Bothersome Symptom
|
2 Hour
Endpoint
|
Rimegepant
(N=669)
|
Placebo
(N=682)
|
Difference
|
p-value
|
|
Pain
Freedom
|
21.2%
|
10.9%
|
10.3%
|
<
0.0001
|
|
Freedom from
MBS1
|
35.1%
|
26.8%
|
8.3%
|
0.0009
|
|
1. Most Bothersome
Symptom of Photophobia, Phonophobia or Nausea
|
The safety and tolerability observations of rimegepant in Study
303 were consistent with the profile previously observed in Studies
301 and 302. Table 2 shows the pooled safety data
across all three trials. In study 303, no single adverse event (AE)
occurred in the rimegepant group with an incidence higher than 1.6%
and overall rates of AEs were similar to placebo. With regard
to liver function tests, one patient
treated with placebo and one patient treated with
rimegepant showed LFTs > 3x ULN in Study 303. Pooled liver
function test results across the three pivotal trials (n=3,556)
performed to date showed that rimegepant was similar to placebo
with regard to aminotransferase (ALT or AST) levels above the upper
limit of normal (ULN) and no patients experienced elevations in
bilirubin > 2x ULN (Table 3).
Table 2: Pooled Adverse Event (AE) Safety Data:
Rimegepant was Similar to Placebo Across Studies
|
AEs from Studies
301, 302 and 303 with an incidence ≥ 1%
|
|
Adverse
Event
|
Rimegepant
N=1,771
|
Placebo
N=1,785
|
|
≥ 1 On-Study
AE1
|
252
(14.2%)
|
209
(13.2%)
|
|
Nausea
|
26 (1.5%)
|
15 (0.8%)
|
|
UTI
|
21 (1.2%)
|
12 (0.7%)
|
|
SAEs2
|
3 (0.2%)
|
3 (0.2%)
|
|
1. No other
individual AEs ≥ 1% in rimegepant treated subjects than those
listed in table. Includes all AEs without attribution to drug
relatedness.
|
|
2. No drug-related
Serious Adverse Events (SAEs). Two of the subjects with SAE in
rimegepant group and one in placebo group had not been dosed before
onset of SAE.
|
Table 3: Pooled Liver Function Test (LFT) Profile:
Rimegepant was Similar to Placebo Across Studies
|
Pooled LFT Results
from Studies 301, 302, and 303*
|
|
ALT or
AST
|
Rimegepant
N=1,771
|
Placebo
N=1,785
|
|
>
ULN1
|
48 (2.7%)
|
52 (2.9%)
|
|
> 3x
ULN
|
2 (0.1%)
|
2 (0.1%)
|
|
> 5x
ULN
|
1
(0.06%) 2
|
0
|
|
> 10x
ULN
|
0
|
0
|
|
> 20x
ULN
|
0
|
0
|
|
1Upper
limit of normal; ALT alanine aminotransferase; AST aspartate
aminotransferase
|
|
2AST
elevation, Not Drug-Related as deemed by the investigator: subject
newly initiated weight-lifting with laboratory results consistent
with muscle injury
*AST/ALT Categories
are not mutually exclusive; No bilirubin elevations > 2x ULN
across Studies 301, 302 and 303
|
Vlad Coric, M.D., Chief Executive
Officer of Biohaven, stated, "Fast-acting and long-lasting pain
relief in an easy to use formulation is important to
patients. Rimegepant provides these characteristics and we
believe is the first oral CGRP receptor antagonist to report
superiority over placebo on pain relief and return to normal
functioning by 60 minutes. These results, combined with the
previous data from the two prior Phase 3 trials, reinforce the
potential of rimegepant to be an important new and differentiated
option for the acute treatment of migraine."
The double-blind, randomized, multicenter, Phase 3 outpatient
trial treated 1,375 patients across sites in the United States. Patients were required to
have at least a one-year history of migraine (with or without
aura), consistent with a diagnosis according to the International
Classification of Headache Disorders. The trial was designed to
evaluate the efficacy and safety of rimegepant Zydis ODT
formulation compared with placebo in the acute treatment of
migraine. Patients were given a single 75 mg dose of rimegepant
Zydis ODT or a matching placebo. They were instructed to take their
study medication when they had a migraine which reached moderate or
severe pain intensity.
Richard B. Lipton, M.D.,
Professor and Vice Chair of Neurology, and Director of the
Montefiore Headache Center, at the Albert Einstein College of
Medicine stated, "Migraine is a serious, incapacitating, and highly
prevalent neurological disease affecting 15% of the population.
People with migraine miss work and lose time with their families
because of disabling pain and associated symptoms. The most
widely used acute prescription drugs, triptans, have cardiovascular
contraindications, do not work in everyone and have been associated
with high recurrence rates in those who respond. Rimegepant
Zydis ODT formulation is rapidly absorbed and has a long
half-life. It is gratifying to see the attractive
pharmacokinetic profile deliver on its promise in a randomized
trial. If approved, rimegepant could potentially play an important
role in helping people with migraine find relief from their pain
and get back to what they need to do in their lives."
Biohaven has an exclusive worldwide license agreement with
Catalent U.K. Swindon Zydis Limited, a subsidiary of Catalent, Inc.
(NYSE: CTLT) ("Catalent") to provide Catalent's Zydis ODT
formulation for the development of rimegepant. The agreement also
provides exclusive rights for developing small molecule CGRP
receptor antagonists with the Zydis ODT technology. Catalent's
proprietary Zydis ODT fast-dissolve formulation is a unique,
freeze-dried, oral solid dosage form that dissolves rapidly in the
mouth, without the need for water. With more than 20 products
launched in 50 countries and a dispersion speed of three seconds or
less, Zydis ODT is generally recognized as the world's fastest and
best-in-class orally dissolving tablet. Zydis is a registered
trademark of R. P. Scherer Technologies, Inc.
Robert Croop, M.D., Chief
Development Officer – Neurology at Biohaven commented, "We are very
pleased with the performance of our novel rimegepant Zydis ODT and
believe that this formulation enhances the benefits offered by
rimegepant and heightens its potential to become a best-in-class
agent for the acute treatment of migraine. The rimegepant Zydis ODT
was designed to enable patients to experience relief wherever and
whenever a migraine strikes. The results of Study 303 demonstrate
the clinical utility of this novel formulation."
Additional secondary and exploratory outcome measures from
this study are anticipated to be presented at upcoming scientific
meetings in 2019.
Conference Call and Webcast
Biohaven will host a conference call and webcast today,
December 3, 2018, at 8:30 a.m. ET (5:30 a.m.
PT) to discuss topline results from the Phase 3 clinical
trial of Rimegepant Zydis Orally Dissolving Tablet (ODT). The call
can be accessed by dialing 877-407-9120 (domestic) or 412-902-1009
(international). To access the audio webcast with slides, please
visit the "Events & Presentations" page in the Investors
section of the Company's website at
https://www.biohavenpharma.com/investors/news-events/events-presentations.
An archive of today's teleconference and webcast will be available
on Biohaven's website for 6 months following the call.
About Migraine
Migraine is both a widespread and disabling neurological
disorder. The Migraine Research Foundation ranks migraine as the
world's third most prevalent illness, affecting approximately 36
million people or 1 out of 4 households in the United States. And the Global Burden of
Disease Study 2015 rates migraine as the seventh highest specific
cause of disability worldwide. More than 90% of people with
migraine are unable to work or function normally during an attack.
Current treatment approaches, such as triptans, can be limited by
headache recurrence within 24 hours after taking migraine
medication, as well as cardiovascular contraindications and
warnings.
About Rimegepant
Rimegepant (formerly known as BHV-3000), Biohaven's lead product
candidate, is an orally available, selective and potent small
molecule CGRP receptor antagonist. The company believes rimegepant
has the potential to be a best-in-class CGRP receptor antagonist
for the acute and preventive treatment of migraine with the ability
to address important unmet needs. The efficacy and safety profile
of rimegepant has been consistently established across four
randomized controlled trials, now including three Phase 3 studies
(ClinicalTrials.gov Identifiers: NCT03235479 (Study 301);
NCT03237845 (Study 302), NCT03461757 (Study 303)) and a Phase
2b study (NCT01430442). The
co-primary endpoints achieved in the Phase 3 trials are consistent
with regulatory guidance from the U.S. Food and Drug Administration
(FDA) and provide the basis for a planned submission of a new drug
application (NDA) to the FDA in 2019.
About Biohaven
Biohaven is a clinical-stage biopharmaceutical company with a
portfolio of innovative, late-stage product candidates targeting
neurological diseases, including rare disorders. Biohaven has
combined internal development and research with intellectual
property licensed from companies and institutions including
Bristol-Myers Squibb Company, AstraZeneca AB, Yale University, Catalent, Rutgers, ALS Biopharma LLC and Massachusetts
General Hospital. Currently, Biohaven's lead development programs
include multiple compounds across its CGRP receptor antagonist,
glutamate modulation, and myeloperoxidase inhibitor platforms.
Biohaven's common shares are listed on the New York Stock Exchange
and traded under the ticker symbol BHVN. More information about
Biohaven is available at www.biohavenpharma.com
Forward-Looking Statements
This news release includes forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
These forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of the Company's management. All
statements, other than statements of historical facts, included in
this press release, including the Company's timing of the expected
NDA submission for rimegepant and its potential to be an improved
treatment option for the acute treatment of migraine, are
forward-looking statements. The use of certain words, including
"believe" and "will" and similar expressions, is intended to
identify forward-looking statements. The Company may not actually
achieve the plans and objectives disclosed in the forward-looking
statements, and you should not place undue reliance on the
Company's forward-looking statements. Various important factors
could cause actual results or events to differ materially from
those that may be expressed or implied by our forward-looking
statements, including that topline data is based on preliminary
analysis of key efficacy and safety data, and such data could
change following a more comprehensive review and evaluation of more
extensive data from the trials that the Company has not yet
received, and these preliminary conclusions may not accurately
reflect the complete results of the clinical trials,
and uncertainties relating to the timing for submitting
an NDA and the potential regulatory approval of rimegepant.
Additional important factors to be considered in connection with
forward-looking statements are described in the "Risk Factors"
section of the Company's Quarterly Report on Form 10-Q filed with
the Securities and Exchange Commission on November 14, 2018 and other filings Biohaven
makes with the U.S. Securities and Exchange Commission from time to
time. The forward-looking statements are made as of this date and
the Company does not undertake any obligation to update any
forward-looking statements, whether as a result of new information,
future events or otherwise, except as required by law.
For further information, contact Dr. Vlad Coric, the Chief Executive Officer at
Vlad.Coric@biohavenpharma.com
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-delivers-positive-phase-3-results-with-rimegepant-zydis-orally-dissolving-tablet-odt-rapid-and-lasting-benefit-for-the-acute-treatment-of-migraine-300758762.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-delivers-positive-phase-3-results-with-rimegepant-zydis-orally-dissolving-tablet-odt-rapid-and-lasting-benefit-for-the-acute-treatment-of-migraine-300758762.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.