Alphatec Holdings, Inc. Announces FDA Clearance for Its Adjustable Bridge Product
18 Juli 2006 - 2:30PM
PR Newswire (US)
CARLSBAD, Calif., July 18 /PRNewswire-FirstCall/ -- Alphatec
Holdings, Inc. (NASDAQ:ATEC), a medical device company that
provides customized spinal products and a broad suite of spine
fusion solutions, announced today that it has received 510(k)
clearance from the U.S. Food and Drug Administration ("FDA")
allowing it to market its adjustable bridge product. As previously
disclosed in Company filings, Alphatec entered in to a private
label distribution agreement with Sea Spine, Inc. for the sale of
Sea Spine's Cross Link(R) adjustable bridge product under
Alphatec's private label. This FDA clearance will allow Alphatec to
begin the immediate commercial sale of its private label adjustable
bridge, which will be used in combination with the Company's
Zodiac(R) line of polyaxial pedicle screws. Ronald G. Hiscock,
President and Chief Executive Officer of Alphatec remarked,
"Receipt of this 510(k) clearance is an important milestone for the
commercial sale of our adjustable bridge product. We believe that
many surgeons prefer the use of an adjustable bridge in order to
provide additional stabilization in multi-level spinal surgeries.
As such, we hope to see increased revenue per surgery as physicians
make use of this excellent product complement. Once again, Alphatec
has demonstrated its commitment to expanding its product offering
with high quality products and services that surgeons require."
About Alphatec Alphatec is a medical device company focused on the
design, development, manufacturing and marketing of products for
the surgical treatment of spine disorders. Alphatec's principal
product offerings are primarily focused on the U.S. spine fusion
market, which is estimated to approach $3 billion in 2007, through
its wholly-owned subsidiary, Alphatec Spine, Inc., and include a
variety of spinal implant products and systems comprised of
components such as spine screws, spinal spacers, and plates that
offer multiple solutions to address patients' needs. In addition to
Alphatec's U.S. presence, it also participates in the Japanese
spine fusion and orthopedic trauma markets through its subsidiary,
Alphatec Pacific, Inc. Our mission is to be a value based leader in
the spinal market by providing unmatched service to and taking
scientific direction from our physician customers. We do so through
our unique in-house manufacturing facility in Carlsbad, California
that allows us to respond quickly and to customize innovative
products needed for spine fusion surgery. Forward Looking
Statements This press release may contain forward-looking
statements that involve risks and uncertainty, including statements
that Alphatec's commercial sale of such private label adjustable
bridge products will occur in the time periods planned, that many
of Alphatec's multi-level fusion surgeries are candidates for use
with an adjustable bridge product, that surgeons will adopt our
private label adjustable bridge product, and that our private label
adjustable bridge product will provide complementary revenues by
increasing our revenue per surgery. Such statements are based on
management's current expectations and are subject to a number of
risks and uncertainties that could cause actual results to differ
materially from those described in the forward-looking statements.
Alphatec cautions investors that there can be no assurance that
actual results or business conditions will not differ materially
from those projected or suggested in such forward-looking
statements as a result of various factors, including, but not
limited to, the following: Alphatec's ability to develop and expand
its spine fusion business in the United States and Japan,
Alphatec's ability to expand and maintain a successful sales and
marketing organization, continuation of favorable third party payor
reimbursement for procedures performed using our products,
unanticipated expenses or liabilities or other adverse events
affecting cash flow or Alphatec's ability to achieve profitability,
uncertainty of additional funding, uncertainty of success in
developing any new products, failure to successfully introduce and
develop new products, including products related to license
agreements, failure to obtain FDA clearance or approval for
particular devices, Alphatec's ability to compete with other
competing products and with emerging new technologies within and
outside of spinal fusion, product liability exposure and patent
infringement claims. Please refer to the risks detailed in
Alphatec's Form S-1 on file with the SEC, as well as the risks
detailed from time to time in Alphatec's SEC reports, including
quarterly reports on Form 10-Q, reports on Form 8-K and annual
reports on Form 10-K. We disclaim any intention or obligation to
update or revise any forward-looking statements, whether as a
result of new information, future events, or otherwise, unless
required by law. Investors: Alphatec Holdings, Inc.: Matt Clawson
Stephen T.D. Dixon Allen & Caron, Inc. Vice President and CFO
(949) 474-4300 (760) 431-9286 DATASOURCE: Alphatec Holdings, Inc.
CONTACT: Investors, Matt Clawson of Allen & Caron, Inc.,
+1-949-474-4300, , for Alphatec Holdings, Inc.; or Stephen T.D.
Dixon, Vice President and CFO of Alphatec Holdings, Inc.,
+1-760-431-9286 Web site: http://www.alphatecspine.com/
Copyright
Alphatec (NASDAQ:ATEC)
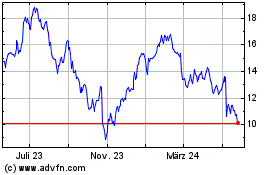
Historical Stock Chart
Von Jun 2024 bis Jul 2024
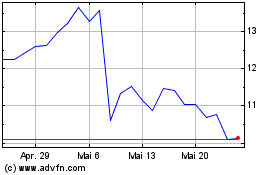
Alphatec (NASDAQ:ATEC)
Historical Stock Chart
Von Jul 2023 bis Jul 2024