Thermo Fisher Scientific to Help Advance Myeloid Cancer Clinical Research and Treatment Utilizing Next Generation Sequencing Technology
11 Juli 2024 - 2:00PM
Business Wire
myeloMATCH Precision Medicine Clinical Trial
Leverages Next-Day Genomic Testing to Quickly Match Patients with
Appropriate Trials Across North America
To help accelerate research into new treatments for Acute
Myeloid Leukemia (AML) and Myelodysplastic Syndrome (MDS), Thermo
Fisher Scientific Inc. (NYSE: TMO), the world leader in serving
science, is partnering with the National Cancer Institute (NCI) –
part of the National Institutes of Health – on the myeloMATCH
(Molecular Analysis for Therapy Choice) precision medicine umbrella
trial. By testing patients’ bone marrow and blood for certain
genetic biomarkers using Thermo Fisher’s next-generation sequencing
(NGS) technology, clinical sites can more quickly match patients
with an appropriate clinical trial that tests a treatment designed
to target specific mutations present in the samples.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240711570854/en/
myeloMATCH Precision Medicine Clinical
Trial Leverages Next-Day Genomic Testing to Quickly Match Patients
with Appropriate Trials Across North America (Photo: Business
Wire)
AML is an aggressive cancer of the bone marrow and blood and is
one of the most common types of leukemia. Because it can advance
quickly with a five-year survival rate of only 30-40% for people
under age 60, rapid detection and effective treatment are essential
to improve patient outcomes. Further, clinical practice guidelines
note the importance of rapid genetic analysis to identify
biomarkers that may help match patients with optimal treatments
based on their unique cancer profile.
“myeloMATCH breaks new ground in many ways, not least by
assembling a portfolio of sub-studies to treat patients with
specific subtypes of AML and MDS through all stages of their
treatment journey,” said Dr. Harry P. Erba, chair of the Southwest
Oncology Group (SWOG) Leukemia Committee and co-chair of the
myeloMATCH Senior Science Council. “Many more treatment options are
available for people with AML and MDS than just 5-10 years ago, and
many more targeted therapies are being developed. However, to
choose the best treatment option for our patients requires
knowledge of the genetic changes that underly the disease, which
vary between patients. This information is needed quickly in order
to begin effective therapy for very aggressive cancers. Our
partnership with Thermo Fisher allows us to obtain the required
genomic profiling rapidly and begin therapies specific for each
subtype of the disease. Through this personalized approach to
treatment, we believe we will increase the number of people who are
leukemia survivors."
The study aims to complete genomic testing and deliver complete
results within a few days across testing modalities to help quickly
enroll patients into specific sub-studies based on their biomarker
profile at time of diagnosis. As the first turnkey NGS solution
that automates the specimen-to-report workflow designed to deliver
results in a single day with just two user touchpoints, the Ion
Torrent™ Genexus™ System* will help accelerate the process of
matching patients with appropriate clinical trials.
myeloMATCH will be open in the U.S. and Canadian sites of the
NCI National Clinical Trials Network, which includes more than
2,200 sites. Further, the NCI’s Division of Cancer Treatment and
Diagnosis has developed cooperative research and development
agreements with many pharmaceutical companies that will provide
different drugs to support myeloMATCH. By conducting multiple
treatment sub-studies specific to genomic types, myeloMATCH may
help fuel the development of promising new therapies.
“myeloMATCH is an immense step forward for patients with
aggressive and rapidly advancing cancers who need better treatment
options,” said John Sos, senior vice president and president, life
sciences solutions at Thermo Fisher Scientific. “Using the Genexus
System, clinical teams across sites can quickly match eligible
patients with the right trials to ultimately better understand the
clinical impact of these therapies. By helping to expedite this
process, we can ensure that more patients have access to
appropriate precision oncology treatments.”
Patient samples will be sequenced in the myeloMATCH Molecular
Diagnostics Laboratory Network (MDNet) using the Genexus System and
reagents along with the Oncomine-based NCI Myeloid Assay as part of
an approved Investigational Device Exemption (IDE) to assign
participants to myeloMATCH treatment studies and has received
Investigational New Drug authorization by the U.S. Federal Drug
Administration. The MDNet sites at the Molecular Characterization
Laboratory, part of the Frederick National Laboratory for Cancer
Research, and the Fred Hutchinson Cancer Center in Seattle, Wash.,
are funded by NCI for this activity. As presented during the 2023
American Society of Hematology (ASH) annual meeting, the assay
demonstrated high sensitivity and reproducibility between
sites.
To learn more about myeloMATCH, please visit
https://clinicaltrials.gov/study/NCT05564390
* Available for research use only in the United States. Not for
use in diagnostic procedures.
About Thermo Fisher Scientific
Thermo Fisher Scientific Inc. is the world leader in serving
science, with annual revenue over $40 billion. Our Mission is to
enable our customers to make the world healthier, cleaner and
safer. Whether our customers are accelerating life sciences
research, solving complex analytical challenges, increasing
productivity in their laboratories, improving patient health
through diagnostics or the development and manufacture of
life-changing therapies, we are here to support them. Our global
team delivers an unrivaled combination of innovative technologies,
purchasing convenience and pharmaceutical services through our
industry-leading brands, including Thermo Scientific, Applied
Biosystems, Invitrogen, Fisher Scientific, Unity Lab Services,
Patheon and PPD. For more information, please visit
www.thermofisher.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240711570854/en/
Media Contact Information: Sandy Pound Thermo Fisher Scientific
Phone: 781-622-1223 E-mail: sandy.pound@thermofisher.com
Investor Contact Information Rafael Tejada Thermo Fisher
Scientific Phone: 781-622-1256 Email:
rafael.tejada@thermofisher.com
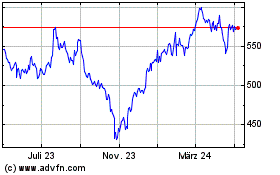
Thermo Fisher Scientific (NYSE:TMO)
Historical Stock Chart
Von Jun 2024 bis Jul 2024
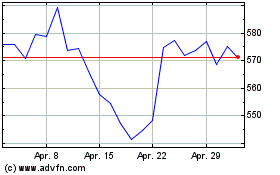
Thermo Fisher Scientific (NYSE:TMO)
Historical Stock Chart
Von Jul 2023 bis Jul 2024