Filed Pursuant to Rule 424(b)(4)
Registration Statement No. 333-255112
PROSPECTUS
ABVC BIOPHARMA, INC.
225,500 Shares of Common Stock Underlying Previously
Issued Warrants
This prospectus relates to the offer and sale
by ABVC Biopharma, Inc., a Nevada corporation of up to 225,500 shares of common stock underlying warrants previously issued by us that
are issuable at a price of $62.5 per share from time to time upon exercise of outstanding warrants issued to investors and the underwriters
in our public offering that was consummated in August 2021 (the “August 2021 Public Offering”), the issuance of which were
previously registered on a Registration Statement on Form S-1 (File No. 333-255112).
We are not selling any shares of our common stock in this offering and, as a result, we will not receive any proceeds from the sale of the common stock covered by this prospectus. All of the net proceeds from the sale of our common stock will go to the warrant holders. Upon exercise of the warrants, however, we will receive proceeds from the exercise of such warrants if exercised for cash and not on a cashless basis. Any proceeds received from the exercise of such warrants will be used for general working capital and other corporate purposes.
Our
common stock issued in the initial public offering are listed on the Nasdaq Capital Market under the symbol “ABVC.” On April
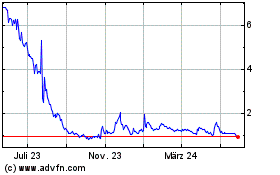
18, 2024, the last reported sale price of our common stock on the Nasdaq Capital Market was $1.02 per share. There is no established
public trading market for the Warrants and we will not seek to list them. We urge prospective purchasers of our common stock to obtain
current information about the market prices of our common stock issued in connection with our initial public offering in August 2021.
Investing in our securities involves a high
degree of risk. See “Risk Factors” starting on page 9 of this prospectus.
See the “Plan of Distribution” section
of this prospectus beginning on page 99 for more information on this offering.
No underwriter or person has been engaged to facilitate the sale of Shares in this offering. All costs associated with the registration were borne by us.
You should read this prospectus, together with additional information described under the headings “Incorporation of Certain Information by Reference” and “Where You Can Find More Information”, carefully before you invest in any of our securities.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is May 22, 2024
TABLE OF CONTENTS
You should rely only on the information contained in this prospectus or in any free writing prospectus that we may specifically authorize to be delivered or made available to you. We and our Underwriter have not authorized anyone to provide you with any information other than that contained in this prospectus or in any free writing prospectus we may authorize to be delivered or made available to you. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus may only be used where it is legal to offer and sell our securities. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery of this prospectus or any sale of our securities. Our business, financial condition, results of operations and prospects may have changed since that date. We are not making an offer of these securities in any jurisdiction where the offer is not permitted.
Unless the context otherwise requires, the terms “ABVC,” “we,” “us” and “our” in this prospectus refer to ABVC BioPharma, Inc., and “this offering” refers to the offering contemplated in this prospectus.
PROSPECTUS CONVENTIONS
Except where the context otherwise requires and for purposes of this prospectus only:
“American BriVision Corporation” refers to a Delaware corporation and wholly-owned subsidiary of ABVC;
“APR” or “annual percentage rate” refers to the annual rate that is charged to borrowers, including a fixed interest rate and a transaction fee rate, expressed as a single percentage number that represents the actual yearly cost of borrowing over the life of a loan;
“BioKey” means BioKey, Inc. refers to a California corporation and wholly-owned subsidiary of ABVC;
“BioLite” means BioLite Holding, Inc. refers to a Nevada corporation and a wholly-owned subsidiary of ABVC;
The “Board” or “Board of Directors” refers to the board of directors of the Company;
“China”
and “P.R.C.” refer to the People’s Republic of China, including Hong Kong Special Administrative Region or Macau Special
Administrative Region, unless referencing specific laws and regulations adopted by the PRC and other legal or tax matters only applicable
to mainland China, excluding Taiwan for purposes of this prospectus;
“Common Stock” is the Common Stock of ABVC Biopharma, Inc., par value US$0.001 per share;
“Merger Agreement” means the Agreement and Plan of Merger dated as of January 31, 2018, pursuant to which the Company, BioLite, BioKey, “BioLite Acquisition Corp.” a Nevada corporation, and BioKey Acquisition Corp.” a California corporation completed a business combination on February 8, 2019 where ABVC acquired BioLite and BioKey via the issuance of additional shares of Common Stock to the shareholders of BioLite and BioKey;
“Series A Convertible Preferred Stock” is the Series A convertible preferred stock of ABVC Biopharma, Inc., par value US$0.001 per share;
The terms “we,” “us,” “our,” “the Company,” “our Company” or “ABVC” refers to ABVC Biopharma, Inc., a Nevada corporation, and all of the Subsidiaries as defined herein unless the context specifies;
“R.O.C.” or “Taiwan” refers to Taiwan, the Republic of China;
“Subsidiary” or “Subsidiaries,” refer to American BriVision Corporation, sometimes referred to as “BriVision”, BioLite Holding, Inc. or BioLite and BioKey, Inc. or BioKey;
All references to “NTD” and “New Taiwan Dollars” are to the legal currency of R.O.C.; and
All references to “U.S. dollars”, “dollars”, and “$” are to the legal currency of the U.S.
This prospectus specifies certain NTD amounts and in parenthesis the approximate U.S. dollar amounts at the exchange rate on the date of this prospectus. The conversion rates regarding NTD and U.S. dollars are subject to change and, therefore, we can provide no assurance that U.S. dollar amounts specified in this prospectus will not change.
For clarification, this prospectus follows English naming convention of first name followed by last name, regardless of whether an individual’s name is Chinese or English.
This prospectus does not discuss any affiliates of the Company that are not controlled by the Company, such as BioFirst and BioFirst Australia.
INDUSTRY AND MARKET DATA
This prospectus includes information with respect to market and industry conditions and market share from third-party sources or based upon estimates using such sources when available. We have not, directly or indirectly, sponsored or participated in the publication of any of such materials. We believe that such information and estimates are reasonable and reliable. We also assume the information extracted from publications of third-party sources has been accurately reproduced. We understand that the Company would be liable for the information included in this prospectus if any part of the information was incorrect, misleading or imprecise to a material extent.
PROSPECTUS SUMMARY
This summary highlights information contained elsewhere in this prospectus and does not contain all of the information that you should consider in making your investment decision. Before investing in our securities, you should carefully read this entire prospectus, including our financial statements and the related notes and the information set forth under the headings “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in each case included elsewhere in this prospectus.
Company Overview
Our Mission
We
devote our resources to building a sophisticated biotech company and becoming a pioneer in the biopharmaceutical industry. Dr. Uttam
Patil, our Chief Executive Officer, and Dr. Tsung-Shann Jiang, the founder and majority shareholder of the Company, understand the challenges
and opportunities of the biotech industry and intend to provide therapeutic solutions to significant unmet medical needs and to improve
health and quality of human life by developing innovative botanical drugs to treat central nervous system (“CNS”) and oncology/
hematology diseases.
Business Overview
As of the date hereof, the Company’s minimal revenue has come from the sale of CDMO services through BioKey. However, the Company’s focus is on developing a pipeline of products by carefully tracking new medical discoveries or medical device technologies in research institutions in the Asia-Pacific region. Pre-clinical, disease animal model and Phase I safety studies are examined closely by the Company’s scientists and other specialists known to the Company to identify drugs or medical devices that it believes demonstrate efficacy and safety based on the Company’s internal qualifications. Once a drug or medical device is shown to be a good candidate for further development and ultimately commercialization, ABVC licenses the drug or medical device from the original researchers and introduces the drug or medical device clinical trial plan to highly respected principal investigators in the United States, Australia and Taiwan. In almost all cases, ABVC has found that research institutions in each of those countries are eager to work with the Company to move forward with Phase II clinical trials.
Institutions
that have or are now conducting phase II clinical trials in partnership with ABVC include:
|
● |
Drug: ABV-1504, Major
Depressive Disorder (MDD), Phase II completed. NCE drug Principal Investigators: Charles DeBattista M.D. and Alan F. Schatzberg,
MD, Stanford University Medical Center, Cheng-Ta Li, MD, Ph.D – Taipei Veterans General Hospital |
|
● |
Drug: ABV-1505, Adult Attention-Deficit
Hyperactivity Disorder (ADHD), Phase II Part 1 completed. Principal Investigators: Keith McBurnett,
Ph.D. and Linda Pfiffner, Ph.D., University of California San Francisco (UCSF), School of Medicine.
Phase II, Part 2 clinical study sites include UCSF and 5 locations in Taiwan. The Principal Investigators
are Keith McBurnett, Ph.D. and Linda Pfiffner, Ph.D., University of California San Francisco (UCSF),
School of Medicine; Susan Shur-Fen Gau, M.D., National Taiwan University Hospital; Xinzhang Ni, M.D.
Linkou Chang Gung Memorial Hospital; Wenjun Xhou, M.D.; Kaohsiung Chang Gung Memorial Hospital; Ton-Ping
Su, M.D., Cheng Hsin General Hospital; Cheng-Ta Li, M.D., Taipei Veterans General Hospital. Phase
II, Part 2 began in the 1st quarter of 2022 at the 5 Taiwan sites. The UCSF site joined the study
in the 2nd quarter of 2023. The subjects enrolled in the study has reached the number for interim
analysis in 2023 December, and the interim analysis of the study is in progress. |
|
● |
Drug: ABV-1601, Major Depression
in Cancer Patients, Phase I/II, NCE drug Principal Investigator: Scott Irwin, MD, Ph.D. – Cedars
Sinai Medical Center (CSMC). The Phase I clinical study will be initiated in the 1st quarter of 2024. |
|
● |
Medical Device: ABV-1701,
Vitargus® in vitrectomy surgery, Phase II Study has been initiated in Australia and Thailand,
Principal Investigator: Duangnate Rojanaporn, M.D., Ramathibodi Hospital; Thuss Sanguansak, M.D.,
Srinagarind Hospital of the two Thailand Sites and Professor/Dr. Matthew Simunovic, Sydney Eye Hospital;
Dr. Elvis Ojaimi, East Melbourne Eye Group & East Melbourne Retina. The Phase II study started
in the 2nd quarter of 2023, and the company is working on improvements to the Vitargus Product through
the new batch of investigational product. |
The following trials
are expected to begin in the third quarter of 2024:
|
● |
Drug: ABV-1519, Non-Small Cell
Lung Cancer treatment, Phase I/II Study in Taiwan, Principal Investigator: Dr. Yung-Hung Luo, M.D.,
Taipei Veterans General Hospital (TVGH) |
|
● |
Drug: ABV-1703, Advanced
Inoperable or Metastatic Pancreatic Cancer, Phase II, Principal Investigator: Andrew E. Hendifar,
MD – Cedars Sinai Medical Center (CSMC) |
Upon successful completion
of a Phase II trial, ABVC will seek a partner, typically a large pharmaceutical company, to complete a Phase III study and commercialize
the drug or medical device upon approval by the US FDA, Taiwan TFDA and other country regulatory authorities.
Corporate Structure
ABVC was incorporated under the laws of the State of Nevada on February 6, 2002 and has three wholly-owned Subsidiaries: BriVision, BioLite Holding, Inc. and BioKey, Inc. BriVision was incorporated in July 2015 in the State of Delaware and is in the business of developing pharmaceutical products in North America.
BioLite Holding was incorporated under the laws of the State of Nevada on July 27, 2016, with 500,000,000 shares authorized, par value $0.0001. Its key Subsidiaries include BioLite BVI, Inc. (“BioLite BVI”) that was incorporated in the British Virgin Islands on September 13, 2016 and BioLite Inc. (“BioLite Taiwan”), a Taiwanese corporation that was founded in February 2006. BioLite Taiwan has been in the business of developing new drugs for over twelve years. Certain shareholders of BioLite Taiwan exchanged approximately 73% of equity securities in BioLite Taiwan for the Common Stock in BioLite Holding in accordance with a share purchase/ exchange agreement (the “Share Purchase/ Exchange Agreement”). As a result, BioLite Holding owns via BioLite BVI approximately 73% of BioLite Taiwan. The other shareholders who did not enter this Share Purchase/ Exchange Agreement retain their equity ownership in BioLite Taiwan.
Incorporated in California on November 20, 2000, BioKey has chosen to initially focus on developing generic drugs to ride the opportunity of the booming industry.
Upon closing of the Mergers on February 8, 2019, BioLite and BioKey became two wholly-owned subsidiaries of ABVC.
In November 2023,
the Company and one of its subsidiaries, BioLite, Inc. (“BioLite”) each entered into a multi-year, global licensing agreement
with AiBtl BioPharma Inc. (“AIBL”) for the Company and BioLite’s CNS drugs with the indications of MDD (Major Depressive
Disorder) and ADHD (Attention Deficit Hyperactivity Disorder) (the “Licensed Products”). The license covers the Licensed
Products’ clinical trial, registration, manufacturing, supply, and distribution rights. The parties are determined to collaborate
on the global development of the Licensed Products. The parties are also working to strengthen new drug development and business collaboration,
including technology, interoperability, and standards development. As per each of the respective agreements, each of ABVC and BioLite
received 23 million shares of AIBL stock at $10 per share, and if certain milestones are met, each may receive $3,500,000 and royalties
equaling 5% of net sales, up to $100 million. Upon the issuance of the shares, AIBL became a subsidiary of ABVC.
The following chart illustrates the corporate structure of ABVC:

Effective March 5, 2022, the Company’s Board for Directors approved amending the Company’s Bylaws to remove Section 2.8, which permitted cumulative voting for directors since cumulative voting is specifically prohibited by our Articles of Incorporation. Since it is not otherwise stated in our Articles of Incorporation or Bylaws, directors shall be elected by a plurality of the votes cast at the election, as provided in the Nevada Revised Statutes.
Effective March 14, 2024, the Company’s
Board for Directors approved amending Section 2.8 of the Company’s Bylaws to revise the number of shares needed to establish a
quorum at shareholder meetings. The amendment changes the quorum requirement from a majority to 33-1/3% of the votes entitled to be cast
on a matter. The full text of our current Bylaws, as amended is attached hereto as Exhibit 3.2.
Recent Developments
On March 25, 2024,
we, and one of our co-development partners, BIOFIRST CORPORATION, a company registered in Taiwan (“BIOFIRST”), each entered
into a twenty-year, global definitive licensing agreement (the “Licensing Agreement”) with ForSeeCon Eye Corporation, a company
registered in the British Virgin Islands (“FEYE”) for the products in the Company and BIOFIRST’s Ophthalmology pipeline,
including Vitargus (the “Licensed Products”). The license covers the Licensed Products’ clinical trial, registration,
manufacturing, supply, and distribution rights; FEYE also has the rights to sublicense or partner with a third party to develop the
Licensed Products.
As per each of the
respective Agreements, each of the Company and BIOFIRST shall receive a total licensing fee of $33,500,000, composed of an upfront payment
of $30,000,000, which can instead be paid with 5 million shares of FEYE stock at $6/share within 30 days after the execution of the Agreement,
and a $3,500,000 cash milestone payment, due 30 days upon completion of next round fundraising, of which there can be no guarantee. Additionally,
each of the Company and BIOFIRST are eligible to receive royalties of 5% of net sales.
NASDAQ Listing
On August 5, 2021, we closed a public offering (the “Offering”) of 1,100,000 units (the “Units”), with each Unit consisting of one share of our common stock (the “Common Stock”), one Series A warrant (the “Series A Warrants”) to purchase one share of common stock at an exercise price equal to $6.30 per share, exercisable until the fifth anniversary of the issuance date, and one Series B warrant (the “Series B Warrants,” and together with the Series A Warrants, the “Public Warrants”) to purchase one share of common stock at an exercise price equal to $10.00 per share, exercisable until the fifth anniversary of the issuance date; the exercise price of the Public Warrants are subject to certain adjustment and cashless exercise provisions as described therein. The Company completed the Offering pursuant to its registration statement on Form S-1 (File No. 333-255112), originally filed with the Securities and Exchange Commission (the “SEC”) on April 8, 2021 (as amended, the “Original Registration Statement”), that the SEC declared effective on August 2, 2021 and the registration statement on Form S-1 (File No. 333-258404) that was filed and automatically effective on August 4, 2021 (the “S-1MEF,” together with the Original Registration Statement, the “Registration Statement”). The Units were priced at $6.25 per Unit, before underwriting discounts and offering expenses, resulting in gross proceeds of $6,875,000. The Offering was conducted on a firm commitment basis. The Common Stock was approved for listing on The Nasdaq Capital Market and commenced trading under the ticker symbol “ABVC” on August 3, 2021.
On
August 19, 2022, we received a deficiency letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq
Stock Market LLC (“Nasdaq”) notifying us that, for the last 30 consecutive business days, the closing bid price for our common
stock was below the minimum $1.00 per share required for continued listing on The Nasdaq Capital Market pursuant to Nasdaq Listing Rule
5550(a)(2) (“Rule 5550(a)(2)”). In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were initially given until February
14, 2023 to regain compliance with Rule 5550(a)(2). Since we did not regain compliance by such date, we requested and received an additional
180 days, until August 14, 2023, to comply with Rule 5550(a)(2).
On
May 24, 2023, the Company received a deficiency letter from the Nasdaq Listing Qualifications Department (the “Staff”) of
the Nasdaq Stock Market LLC (“Nasdaq”) notifying the Company that it was not in compliance with the minimum stockholders’
equity requirement, or the alternatives of market value of listed securities or net income from continuing operations, for continued
listing on the Nasdaq Capital Market. Nasdaq Listing Rule 5550(b)(1) requires listed companies to maintain stockholders’ equity
of at least $2,500,000, and the Company’s stockholders’ equity was $1,734,507 as of March 31, 2023. In accordance with Nasdaq
rules, the Company had 45 calendar days, or until July 10, 2023, to submit a plan to regain compliance. In response to the submitted
plan, Nasdaq granted us an extension until August 31, 2023 to evidence compliance. Following several transactions we then completed,
on September 6, 2023, Nasdaq informed us that they determined that we are in compliance with Nasdaq Listing Rule 5550(b)(1).
Strategy
Key elements of our business strategy include:
|
● |
Advancing to the pivotal trial phase of ABV-1701 Vitargus® for the treatments of Retinal Detachment or Vitreous Hemorrhage, which we expect to generate revenues in the future. |
|
● |
Focusing on licensing ABV-1504 for the treatment of major depressive disorder, MDD, after the successful completion of its Phase II clinical trials. |
|
● |
Completing Phase II, Part 2 clinical trial for ABV-1505 for the treatment of attention deficit hyperactivity disorder, ADHD. |
|
● |
Out licensing drug candidates and medical device candidates to major pharmaceutical companies for phase III and pivotal clinical trials, as applicable, and further marketing if approved by the FDA. |
We
plan to augment our core research and development capability and assets by conducting Phase I and II clinical trials for investigational
new drugs and medical devices in the fields of CNS, Hematology/Oncology and Ophthalmology.
Our
management team has extensive experiences across a wide range of new drug and medical device development and we have in-licensed new
drug and medical device candidates from large research institutes and universities in both the U.S. and Taiwan. Through an assertive
product development approach, we expect that we will build a substantial portfolio of Oncology/ Hematology, CNS and Ophthalmology products.
We primarily focus on Phase I and II research of new drug candidates and out license the post-Phase-II products to pharmaceutical companies;
we do not expect to devote substantial efforts and resources to building the disease-specific distribution channels.
Summary
Risk Factors
The
below is a summary of principal risks to our business and risks associated with this offering. It is only a summary. You should read
the more detailed discussion of risks set forth below and elsewhere in this prospectus for a more complete discussion of the risks listed
below and other risks.
|
● |
Risk associated with our profitability including, but not limited to: |
|
○ |
We have never generated revenue and will continue to be unprofitable in the foreseeable future. |
|
○ |
Our business, operations and plans and timelines could be adversely affected by the effects of health epidemics, including the recent COVID-19 pandemic. |
|
● |
Risk associated with clinical trials and the development of our products, including but not limited to: |
|
○ |
Clinical trials are expensive and time consuming, and their outcome is uncertain. |
| |
○ |
Our
clinical trials could be delayed or unsuccessful, and we may not be able to obtain regulatory approval for any of our drug candidates
when expected, or at all. |
|
○ |
We may experience delays in our clinical trials that could adversely affect our business and operations. |
|
○ |
We rely on third parties to conduct our preclinical studies and clinical trials and if such third parties do not meet our deadlines or otherwise conduct the studies as required, we may be delayed in progressing, or ultimately may not be able to progress, our drug candidates to clinical trials. |
|
○ |
We may not be able to secure and maintain research institutions to conduct our future trials. |
| |
○ |
We
may not be able to secure co-developers or partners to further post-Phase II clinical trials and eventually commercialize our drug
candidates. |
|
○ |
We may need to prioritize the development of our most promising candidates at the expense of the development of other products. |
| |
○ |
Physicians, patients, third-party payors or others in the medical community may not be receptive to our products, and we may not generate any future revenue from the sale or licensing of our products. |
|
● |
Risks associated with intellectual property including but not limited to: |
|
○ |
We may not be successful in obtaining or maintaining patent or other relating rights necessary to the development of our drug candidates in the pipeline; |
|
○ |
The intellectual property rights underlying our exclusive licensing rights may expire or be terminated due to lack of maintenance; |
|
● |
Risks associated with competition and manufacturing including, but not limited to: |
|
○ |
We face competition from entities that have developed or are developing products for our target disease indications, including companies developing novel treatments and technologies similar to ours; and |
|
○ |
We depend primarily upon a sole supplier of our key extract for three drug candidates and could incur significant costs and delays if we are unable to promptly find a replacement for such supplier if the supplier fails to deliver the extract pursuant to our orders. |
|
● |
Risks associated with government regulations including without limitation: |
|
○ |
If we do not obtain the necessary governmental approvals, we will be unable to sub-license or commercialize our pharmaceutical products; and |
|
○ |
Even if we obtain regulatory approval for a drug candidate, our products may remain subject to regulatory scrutiny. |
|
● |
Risk associated with our Common Stock including without limitation: |
|
○ |
The market prices and trading volumes of the Common may be volatile and may be affected by economic conditions beyond our control; and, |
|
○ |
There is only a limited trading market for our Common Stock and such market may never develop. |
| |
● |
Risk
associated with our competition, including, but not limited to: |
| |
○ |
Many
of our current and potential competitors have substantially greater financial, technical and human resources than we do, which could
place us at a significant competitive disadvantage or deny our marketing exclusivity rights. |
| |
○ |
Many
of our current and potential competitors have significantly more experience in the marketing, commercialization, discovery, development
and regulatory approvals of products, which could place us at a significant competitive disadvantage or deny our marketing exclusivity
rights |
These and other risks
described in this prospectus could materially and adversely impact our business, financial condition, operating results and cash flow,
which could cause the trading price of our Common Stock to decline and could result in a loss of your investment. In addition, you should
carefully consider the risks described under “Risk Factors” beginning on page 9.
Corporate Information
ABVC was incorporated under the laws of the State of Nevada on February 6, 2002. BriVision was incorporated in the State of Delaware on July 21, 2015. BioLite was incorporated in the State of Nevada on July 27, 2016. BioKey was incorporated in the State of California on November 20, 2000. BriVision, BioLite and BioKey are three operating subsidiaries that are wholly owned by the Company.
The Company’s shareholders approved an amendment to the Company’s Articles of Incorporation to change the Company’s corporate name from American BriVision (Holding) Corporation to ABVC BioPharma, Inc. and approved and adopted the Certificate of Amendment to affect same at the 2020 annual meeting of shareholders (the “Annual Meeting”). The name change amendment to the Company’s Articles of Incorporation was filed with Nevada’s Secretary of State and became effective on March 8, 2021 and FINRA approved our application for the name change as of May 3, 2021.
The Common Stock was approved for listing on The Nasdaq Capital Market and commenced trading under the ticker symbol “ABVC” on August 3, 2021. The Company’s CUSIP number is 0091F106.
Our principal executive office is located at 44370 Old Warm Springs Blvd., Fremont, CA 94538. Our telephone number at our principal executive office is (510) 668-0881. Our corporate website of BriVision is http://www.abvcpharma.com. The information on our corporate website is not part of, and is not incorporated by reference into, this prospectus.
THE OFFERING
| Securities offered by us |
|
Up
to 225,500 shares of common stock issuable upon exercise of the outstanding warrants issued in our August 2021 public offering, consisting
of (a) 110,000 shares underlying Series A warrant (the “Series A Warrants”) with an exercise price equal to $63.0 per
share, exercisable until the fifth anniversary of the issuance date, (b) 110,000 shares underlying Series B warrant with an exercise
price equal to $100.00 per share, exercisable until the fifth anniversary of the issuance date and subject to certain adjustment
and cashless exercise provisions as described herein and (c) 5,500 shares underlying the warrants issued to the Representative, with
an exercise price equal to $62.5 per share, exercisable until the third anniversary of the issuance date. |
| |
|
|
| Common Stock outstanding prior to completion of this
offering (1) |
|
10,560,421 |
| |
|
|
| Common Stock outstanding immediately after this offering (2) |
|
10,785,921 shares (assuming full exercise of the warrants issued in our August
2021 offering) |
| |
|
|
| Nasdaq Trading Symbol |
|
Our Common Stock is currently quoted on the Nasdaq Capital Market under the symbol “ABVC”. |
| |
|
|
| No Listing for Purchase Warrants |
|
There is no established public trading market for the Purchase Warrants and we will not seek to list them. |
| |
|
|
| Use of proceeds |
|
We will receive approximately $6,875,000 of proceeds if all the currently outstanding warrants issued in the August 2021 public offering are exercised for cash, of which there is no guarantee. We currently intend to use these proceeds for general corporate purposes. See “Use of Proceeds.” |
| |
|
|
| Risk factors |
|
Investment
in our securities involves a high degree of risk and could result in a loss of your entire
investment. See “Risk Factors” beginning on page 9 to read about factors you
should consider before buying our shares of common stock. |
| (1) | The
number of Common Stock to be outstanding before this offering is based on 10,560,421 shares
of Common Stock outstanding as of the date of this prospectus, and excludes:
|
| |
● |
1,307,102 shares of our Common Stock issuable upon the exercise of stock options outstanding; and |
| |
|
|
| |
● |
4,878,442 shares of our Common Stock underlying any outstanding warrants. |
| (2) | The
number of Common Stock to be outstanding after this offering is based on 10,560,421 shares
of Common Stock outstanding as of the date of this prospectus, and excludes: |
| |
● |
1,307,102 shares of our Common Stock issuable upon the exercise of stock options outstanding; |
| |
|
|
| |
● |
7,038,442 shares of our Common Stock underlying any outstanding
warrants;
|
| |
|
|
| |
● |
600,020 shares of our Common Stock issuable upon the exercise of the Purchase Warrants; and |
| |
|
|
| |
● |
92,000 shares of our Common Stock issuable upon the exercise of the Representative’s Warrants issued in this Offering; |
Except as otherwise indicated,
all information in this prospectus assumes:
| |
● |
no exercise of the 1,307,102 shares of our Common Stock issuable
upon the exercise of stock options outstanding. |
OUR AUGUST 2021 PUBLIC OFFERING
On August 5, 2021, we closed our August 2021 public offering of 1,100,000 units (the “Units”), with each Unit consisting of one share of the Company’s common stock, par value $0.001 per share (the “Common Stock”), one Series A warrant (the “Series A Warrants”) to purchase one share of common stock at an exercise price equal to $6.30 per share, exercisable until the fifth anniversary of the issuance date, and one Series B warrant (the “Series B Warrants,” and together with the Series A Warrants, the “Public Warrants”) to purchase one share of common stock at an exercise price equal to $10.00 per share, exercisable until the fifth anniversary of the issuance date; the exercise price of the Public Warrants are subject to certain adjustment and cashless exercise provisions as described therein. The Company completed the Offering pursuant to its registration statement on Form S-1 (File No. 333-255112), originally filed with the Securities and Exchange Commission (the “SEC”) on April 8, 2021 (as amended, the “Original Registration Statement”), that the SEC declared effective on August 2, 2021 and the registration statement on Form S-1 (File No. 333-258404) that was filed and automatically effective on August 4, 2021 (the “S-1MEF,” together with the Original Registration Statement, the “Registration Statement”). The Units were priced at $6.25 per Unit, before underwriting discounts and offering expenses, resulting in gross proceeds of $6,875,000. The Offering was conducted on a firm commitment basis. We also issued a Warrant to WallachBeth Capital, LLC, as underwriter, pursuant to which they may purchase up to 55,000 shares of Common Stock at an exercise price of $6.25 per share.
As of the date hereof, 225,500 shares of our
common stock remain issuable upon exercise of the warrants issued in our offering of August 2021.
RISK FACTORS
Investing in our securities includes a high degree of risk. Prior to making a decision about investing in our securities, you should consider carefully the specific factors discussed below, together with all of the other information contained in this prospectus. If any of the following risks actually occurs, our business, financial condition, results of operations and future prospects would likely be materially and adversely affected. This could cause the market price of our Common Stock to decline and could cause you to lose all or part of your investment.
Risks Related to the Company’s Business
Unfavorable global economic conditions, including as a result of health and safety concerns, could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy, including conditions that are outside of our control, such as the impact of health and safety concerns from the current outbreak of the COVID-19 coronavirus (“COVID-19”). The spread of the COVID-19, which was declared a pandemic by the World Health Organization in March 2020, has caused different countries and cities to mandate curfews, including “shelter-in-place” and closures of most non-essential businesses as well as other measures to mitigate the spread of the virus.
The negative impact of COVID-19 on our operations is ongoing and the extent of which remains uncertain and potentially wide-spread, including:
| |
● |
our ability to successfully execute our long-term growth strategy during these uncertain times; |
| |
● |
our ability to recruit the necessary number of patients to complete future clinical trials; |
| |
|
|
| |
● |
supply chain disruptions in projects ABV-1504, ABV-1505 and ABV-1601, resulting from reduced workforces, scarcity of raw materials, and scrutiny or embargoing of goods produced in infected areas; |
|
● |
our ability to perform on-site due-diligence for project ABV-1505 (MDD Phase II completed new drug candidate) and ABV-1701 (Vitargus FIH completed medical device) with our potential partners/collaborators in US, Mainland China, and Japan; |
|
● |
our ability to access capital sources, as well as the ability of our key customers, suppliers, and vendors to do the same in regard to their own obligations; and |
|
● |
diversion of management and employee attention and resources from key business activities and risk management outside of COVID-19 response efforts, including maintenance of internal controls. |
The COVID-19 pandemic remains highly volatile and continues to evolve on a daily basis and therefore, despite our efforts and developments to combat the virus, there can be no assurance that these measures will prove successful. The extent to which COVID-19 continues to impact the Company’s business, sales, and results of operations will depend on future developments, which are highly uncertain and cannot be predicted.
The Company is a development stage biopharmaceutical company and is thus subject to the risks associated with new businesses in that industry.
The Company acquired the sole licensing rights to develop and commercialize for therapeutic purposes six compounds from BioLite and the right to co-develop with BioFirst a medical device (collectively the “ABVC Pipeline Products”). As such, the Company is a clinical stage biopharmaceutical company with operations that generate unsubstantial revenues. The Company is establishing and implementing many important functions necessary to operate a business, including the clinical research and development of the ABVC Pipeline Products, further establishment of the Company’s managerial and administrative structure, accounting systems and internal financial controls
BioLite and BioKey are expected to continue to have limited revenue and remain unprofitable for an indefinite period of time.
Accordingly, you should consider the Company’s prospects in light of the risks and uncertainties that a pharmaceutical company with a limited operating history and revenue faces. In particular, potential investors should consider that there are significant risks that the Company will not be able to:
|
● |
implement or execute its current business plan, or generate profits; |
|
● |
attract and maintain a skillful management team; |
|
● |
raise sufficient funds in the capital markets or otherwise to effectuate its business plan; |
|
● |
determine that the processes and technologies that it has developed are commercially viable; and/or |
|
● |
enter into contracts with commercial partners, such as licensors and suppliers. |
If any of the above risks occurs, the Company’s business may fail, in which case you may lose the entire amount of your investment in the Company. The Company cannot assure that any of its efforts in business operations will be successful or result in the timely development of new products, or ultimately produce any material revenue and profits.
As a pre-profit biopharmaceutical company, the Company needs to transition from a company with a research and development focus to a company capable of supporting commercial activities. The Company may not be able to reach such transition point or make such a transition, which would have affect our business, financial condition, results of operations and prospects.
If the Company fails to raise additional capital, its ability to implement its business model and strategy could be compromised.
The Company has limited capital resources and operations. The CDMO services provided by BioKey generates a limited amount of revenue that can only partially support the operations of the Company. To date, the Company’s operations have been funded partially from the proceeds from financings or loans from its shareholders. From time to time, we may seek additional financing to provide the capital required to expand research and development (“R&D”) initiatives and/or working capital, as well as to repay outstanding loans if cash flow from operations is insufficient to do so. We cannot predict with certainty the timing or amount of any such capital requirements.
If the Company does not raise sufficient capital
to fund its ongoing development activities, it is likely that it will be unable to carry out its business plans, including R&D development
and expansion of production facilities. Currently, the Company has had to put several projects on hold due to a lack of funding. Even
if the Company obtains financing for near term operations and product development, the Company may require additional capital beyond
the near term. Furthermore, additional capital may not be available in sufficient amounts or on reasonable terms, if at all, and our
ability to raise additional capital may be adversely impacted by potential worsening global economic conditions and the recent disruptions
to and volatility in the credit and financial markets in the United States and worldwide resulting from the ongoing COVID-19 pandemic.
If the Company is unable to raise capital when needed, its business, financial condition and results of operations would be materially
adversely affected, and it could be forced to reduce or discontinue our operations.
The Company has no history in obtaining regulatory approval for, or commercializing, any new drug candidate.
With limited operating history, the Company has never obtained regulatory approval for, or commercialized, any new drug candidate. It is possible that the FDA may refuse to accept our planned New Drug Application (or “NDA”) for any of the six drug products for substantive review or may conclude after review of our data that our application is insufficient to obtain regulatory approval of the new drug candidates or the medical device. Although our CDMO strategic business department has experience in obtaining abbreviated new drug application (or “ANDA”) approvals, the processes and timelines of obtaining an NDA approval and ANDA approval can differentiate substantially. If the FDA does not accept or approve our planned NDA for our product candidates, it may require that we conduct additional clinical, preclinical or manufacturing validation studies, which may be costly. Depending on the FDA required studies, approval of any NDA or application that we submit may be significantly delayed, possibly for several years, or may require us to expend more resources than we have. Any delay in obtaining, or inability to obtain, regulatory approvals of any of our drug candidate will prevent us from sublicensing such product. It is also possible that additional studies, if performed and completed, may not be considered sufficient by the FDA. If any of these outcomes occurs, we may be forced to abandon our planned NDA for such drug candidate, which materially adversely affects our business and could potentially cause us to cease operations. We face similar regulatory risks in a foreign jurisdiction.
Our growth is dependent on our ability to successfully develop, acquire or license new drugs.
Our growth is supported by continuous investment in time, resources and capital to identify and develop new products or new formulations for the market and market penetration. If we are unable to either develop new products on our own or acquire licenses for new products from other parties, our ability to grow revenues and market share will be adversely affected. In addition, we may not be able to recover our investment in the development of new drugs and medical devices, given that projects may be interrupted, unsuccessful, not as profitable as initially contemplated or we may not be able to obtain necessary financing for such development. Similarly, there is no assurance that we can successfully secure such rights from third parties on an economically feasible basis.
Our current products have certain side effects. If the side effects associated with our current or future products are not identified prior to their marketing and sale, we may be required to withdraw such products from the market, perform lengthy additional clinical trials or change the labeling of our products, any of which could adversely impact our growth.
The Company researches and develops the following
seven drug products and one medical device: ABV-1501, ABV-1504, ABV-1505, ABV-1519, ABV-1702, ABV-1601 and ABV-1703. Each of these seven
products may cause serious adverse effects to their users. For example, the API of ABV-1501, ABV-1702 and ABV-1703 is Maitake mushroom
extract. Side effects, or adverse events, associated with Maitake mushroom extract include blood bilirubin increase, lymphocyte count
decrease, neutrophil count decrease, platelet count decrease, white blood cell decrease, headache, and hyperglycemia. Serious adverse
events (collectively, the “SAE”) associated with this compound include leukocytosis, platelet count decrease, eye disorders,
abdominal pain, gastrointestinal disorders, aphonia, lung infection, muscle weakness right-sided, confusion, edema cerebral, stroke,
dyspnea, wheezing, and pruritus.
ABV-1504 and ABV-1505 have the same API, “Radix Polygala”, which is known as Polygala tenuifolia Willd or PDC-1421 Capsule (“Polygala tenuifolia Willd”). Side effects, or adverse events, associated with ABV-1504 and ABV-1505, coming from administration of the trial medicine or examination procedure such as the procedure of taking blood (fainting, pain and/or bruising), may lead to gastrointestinal disorders (abdominal fullness and constipation), nervous system disorders (drowsiness, sleepiness, and oral ulcer). In addition, long-term use may cause miscarriages.
The safety and preliminary efficacy findings from this study, combined with the unique properties of ABV-1701, are supportive of further investigation for its use following vitrectomy surgery in patients requiring vitreous replacement. However, new serious side effects of ABV-1701 may be uncovered as the clinical trials continue.
The occurrence of any of those adverse events would harm our future sales of these medicines and substantially increase the costs and expenses of marketing these medicines, which in turn could cause our revenues and net income to decline. In addition, the reputation and sales of our future medicines could be adversely affected due to the severe side effects discovered.
We may be subject to product liability claims in the future, which could divert our resources, cause us to incur substantial liabilities and limit commercialization of any products that we may develop.
We face an inherent business risk of exposure to product liability claims in the event that the uses of our products are alleged to have caused adverse side effects. Side effects or marketing or manufacturing problems pertaining to any of our products could result in product liability claims or adverse publicity. These risks will exist for those products in clinical development and with respect to those products that receive regulatory approval for commercial sale. Furthermore, although we have not historically experienced any problems associated with claims by users of our products, we do not currently maintain product liability insurance and there could be no assurance that we are able to acquire product liability insurance with terms that are commercially feasible.
We face an inherent risk of product liability claims as a result of the clinical testing of our products and potentially commercially selling any products that we may develop. For example, we may be sued if any product we develop allegedly causes injury or is found to be otherwise unsuitable during clinical testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability or a breach of warranties. Claims could also be asserted under state consumer protection acts. If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our product candidate. Regardless of the merits or eventual outcome, liability claims may result in:
| |
● |
decreased demand for our product candidates or products that we may develop; |
| |
|
|
| |
● |
injury to our reputation and significant negative media attention; |
| |
● |
withdrawal of clinical trial participants; |
| |
● |
significant costs to defend resulting litigation; |
| |
|
|
| |
● |
substantial monetary awards to trial participants or patients; |
| |
|
|
| |
● |
loss of revenue; |
| |
|
|
| |
● |
reduced resources of our management to pursue our business strategy; and |
| |
|
|
| |
● |
the inability to commercialize any products that we may develop. |
We currently have insurance policies to cover liabilities under the clinic trials but do not maintain general liability insurance; and even if we have a general liability insurance in the future, this insurance may not fully cover potential liabilities that we may incur. The cost of any product liability litigation or other proceeding, even if resolved in our favor, could be substantial. We would need to increase our insurance coverage if and when we begin selling any product candidate that receives marketing approval. In addition, insurance coverage is becoming increasingly expensive. If we are unable to obtain or maintain sufficient insurance coverage at an acceptable cost or to otherwise protect against potential product liability claims, it could prevent or inhibit the development and commercial production and sale of our product candidate, which could adversely affect our business, financial condition, results of operations and prospects.
We have conducted, and may in the future conduct, clinical trials for certain of our product candidates at sites outside the United States, and the FDA may not accept data from trials conducted in such locations.
We have conducted and may in the future choose to conduct one or more of our clinical trials outside the United States. Although the FDA may accept data from clinical trials conducted outside the United States, acceptance of this data is subject to certain conditions imposed by the FDA. For example, the clinical trial must be well designed and conducted and performed by qualified investigators in accordance with ethical principles. The trial population must also adequately represent the U.S. population, and the data must be applicable to the U.S. population and U.S. medical practice in ways that the FDA deems clinically meaningful. In addition, while these clinical trials are subject to the applicable local laws, FDA acceptance of the data will be dependent upon its determination that the trials also complied with all applicable U.S. laws and regulations. There can be no assurance that the FDA will accept data from trials conducted outside of the United States. If the FDA does not accept the data from any of our clinical trials that we determine to conduct outside the United States, it would likely result in the need for additional trials, which would be costly and time-consuming and delay or permanently halt our development of the product candidate.
In addition, the conduct of clinical trials outside the United States could have a significant impact on us. Risks inherent in conducting international clinical trials include:
| |
● |
foreign regulatory requirements that could restrict or limit our ability to conduct our clinical trials; |
| |
|
|
| |
● |
administrative burdens of conducting clinical trials under multiple foreign regulatory schema; |
| |
|
|
| |
● |
foreign exchange fluctuations; and |
| |
|
|
| |
● |
diminished protection of intellectual property in some countries. |
If clinical trials of our product candidates fail to demonstrate safety and efficacy to the satisfaction of the FDA and comparable non-U.S. regulators, we may incur additional costs or experience delays in completing, or ultimately be unable to complete the development and commercialization of our product candidates.
We are not permitted to commercialize, market, promote or sell any product candidate in the United States without obtaining marketing approval from the FDA. Comparable non-U.S. regulatory authorities impose similar restrictions. We may never receive such approvals. We must complete extensive preclinical development and clinical trials to demonstrate the safety and efficacy of our product candidate in humans before we will be able to obtain these approvals.
Clinical testing is expensive, difficult to design and implement, can take many years to complete and is inherently uncertain as to outcome. Any inability to successfully complete preclinical and clinical development could result in additional costs to us and impair our ability to generate revenues from product sales, regulatory and commercialization milestones and royalties. In addition, if (1) we are required to conduct additional clinical trials or other testing of our product candidate beyond the trials and testing that we contemplate, (2) we are unable to successfully complete clinical trials of our product candidate or other testing, (3) the results of these trials or tests are unfavorable, uncertain or are only modestly favorable, or (4) there are unacceptable safety concerns associated with our product candidate, we, in addition to incurring additional costs, may:
|
● |
be delayed in obtaining marketing approval for our product candidates; |
|
● |
not obtain marketing approval at all; |
|
● |
obtain approval for indications or patient populations that are not as broad as we intended or desired; |
|
● |
obtain approval with labeling that includes significant use or distribution restrictions or significant safety warnings, including boxed warnings; |
|
● |
be subject to additional post-marketing testing or other requirements; or |
|
● |
be required to remove the product from the market after obtaining marketing approval. |
Even if any of our product candidates receives marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third party payors and others in the medical community necessary for commercial success and the market opportunity for the product candidate may be smaller than we estimate.
We have never completed a new drug or new medical device FDA application process from Phase I to FDA approval and commercialization. Even if our products are approved by the appropriate regulatory authorities for marketing and sale, they may nonetheless fail to gain sufficient market acceptance by physicians, patients, third party payors and others in the medical community. For example, physicians are often reluctant to switch their patients from existing therapies even when new and potentially more effective or convenient treatments enter the market. Further, patients often acclimate to the therapy that they are currently taking and do not want to switch unless their physicians recommend switching products or they are required to switch therapies due to lack of reimbursement for existing therapies.
The potential market opportunities for our products are difficult to estimate precisely. Our estimates of the potential market opportunities are predicated on many assumptions, including industry knowledge and publications, third party research reports and other surveys. While we believe that our internal assumptions are reasonable, these assumptions involve the exercise of significant judgment on the part of our management, are inherently uncertain and the reasonableness of these assumptions has not been assessed by an independent source. If any of the assumptions proves to be inaccurate, the actual markets for our products could be smaller than our estimates of the potential market opportunities.
We may seek to enter into collaborations with third parties for the development and commercialization of our product candidates. If we fail to enter into such collaborations, or such collaborations are not successful, we may not be able to capitalize on the market potential of our product candidates.
We may seek third-party collaborators for development and commercialization of our products. Our likely collaborators for any marketing, distribution, development, licensing or broader collaboration arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies, non-profit organizations, government agencies, and biotechnology companies. Our ability to generate revenues from these arrangements will depend on our collaborators’ abilities to successfully perform the functions assigned to them in these arrangements.
Collaborations involving our products will pose the following risks to us:
|
● |
collaborators may have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
|
● |
collaborators may not pursue development and commercialization of our product candidate or may elect not to continue or renew development or commercialization programs based on preclinical or clinical trial results, changes in the collaborators’ strategic focus or available funding, or external factors such as an acquisition that diverts resources or creates competing priorities; |
|
● |
collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
|
● |
collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidate if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
|
● |
collaborators with marketing and distribution rights to one or more products may not commit sufficient resources to the marketing and distribution of such product or products; |
|
● |
collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
|
● |
collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability; |
|
● |
disputes may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization of our product candidate or that result in costly litigation or arbitration that diverts management attention and resources; and |
|
● |
collaborations may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product candidates. |
Collaborative agreements may not lead to development or commercialization of our product candidate in the most efficient manner or at all. If a collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product development or commercialization program could be delayed, diminished or terminated.
ABVC, through BioLite, may not be able to receive the full amounts available under the collaboration agreement by and between BioLite, Inc. and BioHopeKing, which could increase its burden to seek additional capital to fund the business operations.
In February and December 2015, BioLite, Inc., a subsidiary of BioLite, entered into a total of three collaboration agreements with BioHopeKing to jointly develop ABV-1501 for TNBC (or BLI-1401-2 as used by BioLite internally) and ABV-1504 for MDD (or BLI-1005 as used by BioLite internally) in most Asian countries and BLI-1006, which has been later replaced with BLI-1008 for ADHD in Asia, excluding Japan. ABVC and BioLite are co-developing ABV-1501 for TNBC and ABV-1504 for MDD pursuant to the Collaboration Agreement and its Addendum entered by and between BriVision and BioLite Taiwan where ABVC and BriVision are responsible for the clinical trials of such two new drug candidates. In accordance with the terms of the BioHopeKing Collaboration Agreement for ABV-1501 or BLI-1401-2 and the Addendum thereto, BioLite shall receive payments of a total of $10 million in cash and equity of BioHopeKing or equity securities owned by it at various stages on a schedule dictated by BioLite’s achievements of certain milestones and twelve per cent (12%) of net sales of the drug products when ABV-1501 or BLI-1401-2 is approved for sale in the licensed territories. If BioLite fails to reach any of the milestones in a timely manner, it may not receive the rest of the payments from BioHopeKing. As a result of BioLite’s potential inability to receive the full payments under those collaboration agreements with BioHopeKing, ABVC may have to seek other sources of financing to fund its operation activities.
ABVC and its Subsidiaries may not be successful in establishing and maintaining additional strategic partnerships, which could adversely affect ABVC’s ability to develop and commercialize products, negatively impacting its operating results.
In addition to ABVC’s current collaboration
with BioHopeKing for selected Asian markets, a part of its strategy is to evaluate and, as deemed appropriate, enter into additional
partnerships in the future with major biotechnology or pharmaceutical companies. ABVC’s products may prove to be difficult to effectively
license out as planned. Various regulatory, commercial and manufacturing factors may impact ABVC’s ability to seek co-developers
of or grow revenues from licensing out any of the seven products in the pipeline, none of which has been fully licensed out. Specifically,
ABVC may encounter difficulty by virtue of:
| ● | its
inability to effectively identify and align with commercial partners in the U.S. to collaborate
the development of ABV-1504 for the treatment of Major Depressive Disorder, ABV-1505 to treat
Attention-Deficit Hyperactivity Disease, ABV-1501 for the treatment of Triple Negative Breast
Cancer, ABV-1519 to treat of Non-Small Cell Lung Cancer, ABV-1703 to the treatment of Pancreatic
Cancer, ABV-1601 to treat Depression in Cancer Patients and ABV-1702 to treat Myelodysplastic
syndromes and ABV-1701 Vitargus for the treatments of Retinal Detachment or Vitreous Hemorrhage; |
|
● |
its inability to secure appropriate contract research organizations (“CRO”s) to conduct data analysis, lab research and FDA communication; and |
|
● |
its inability to effectively continue clinical studies on and secure positive research results of all of our investigational new drugs to attract additional commercial collaborators outside the U.S. |
ABVC faces significant competition in seeking appropriate partners for its therapeutic candidates, and the negotiation process is time-consuming and complex. In order for ABVC to successfully partner its autoimmune, CNS and hematology therapeutic candidates, as well as Vitargus, its medical device, potential partners must view these medicinal candidates as economically valuable in markets they determine to be attractive in light of the terms that ABVC is seeking and compared to other available products for licensing by other companies. Even if ABVC is successful in its efforts to establish new strategic partnerships, the terms that ABVC agrees upon may not be favorable, and it may not be able to maintain such strategic partnerships if, for example, development or approval of an autoimmune therapeutic is delayed or sales of an approved product are disappointing. Any delay in entering into new strategic partnership agreements related to any of ABVC’s therapeutic candidates could delay the development and commercialization of such candidates and reduce its competitiveness even if it reaches the market.
If ABVC fails to establish and maintain additional strategic partnerships or collaboration related to its therapeutic candidates that have not been fully licensed, it will bear all of the risk and costs related to the development of any such drug candidate, and it may need to seek additional financing, hire additional employees and otherwise develop expertise for which it has not budgeted. This could negatively affect the development of any incompletely partnered new drug candidates.
ABVC’s licensors may choose to terminate any of the license agreements with ABVC. As a result, ABVC’s research and development of new drug candidates that contain the underlying API may be terminated abruptly.
If ABVC’s Subsidiary BioLite materially breaches any license agreements it has with Yukiguni Maitake Co. (“Yukiguni”), Medical and Pharmaceutical Industry Technology and Development Center (“MPITDC”) or Industrial Technology Research Institute (“ITRI”), or any of such license agreement terminates unexpectedly, BioLite may not be able to continue its research and development of the new drug candidate which contains the underlying API whose license has been terminated. Pursuant to the Yukiguni License Agreement, if BioLite fails to meet the milestone sales requirement or submit certain applications to the appropriate health authorities on a schedule prescribed therein, Yukiguni shall have the right to terminate the Yukiguni License Agreement. If the Yukiguni License Agreement is terminated involuntarily, BioLite will be forced to discontinue its new drug development of ABV-1703, ABV-1502 and ABV-1501 and terminate the collaboration agreements relating to the three new drug candidates. The termination of the right to use the underlying API will materially disrupt the operations of ABVC. Pursuant to the license agreement between BioLite Taiwan and ITRI, if BioLite Taiwan fails to complete the research submission milestones according to the schedule set forth therein without reasons or with reasons unstatisfied with ITRI, ITRI shall have the right to terminate the license agreement with BioLite Taiwan without refund to BioLite Taiwan. BioLite Taiwan and BioLite have submitted the IND for PDC-1421 and subsequently conducted Phase II clinical trials of two drug candidiates developed from PDC-1421 according to the schedule listed in the license agreement between BioLite Taiwan and MPITDC.
ABVC’s Subsidiary BioLite depends
on one supplier for the API of ABV-1703, ABV-1519, ABV-1502 and ABV-1501 and any failure of such supplier to deliver sufficient quantities
of the API that meets its quality standard could have a material adverse effect on its research of these four drug candidates.
Currently BioLite relies primarily on Yukiguni,
a Japanese supplier, to provide Yukiguni Maitake Extract 404, the API which is contained in ABV-1703, ABV-1519, ABV-1502 and ABV-1501,
four of the seven drug candidates in BioLite’s oncology/hematology portfolio. It has entered into the Yukiguni License Agreement,
among other things, for the delivery of Yukiguni Maitake Extract 404. BioLite agrees to fulfill its demand of the Yukiguni Maitake Extract
404 by purchasing first from Yukiguni respecting the therapeutic products and Yukiguni represents that it will provide sufficient quantities
of such API that meets cGMP standards. If the supplies of Yukiguni Maitake Extract 404 were interrupted for any reason, BioLite’s
research and development activities of these four drug candidates could be delayed. These delays could be extensive and expensive, especially
in situations where a substitution is not readily available.
BioLite is currently
negotiating with another supplier of Yukiguni Maitake Extract 404 that is located in Canada. However, there can be no assurance that
the negotiation will be successful. Failure to obtain adequate supplies of high quality Yukiguni Maitake Extract 404 in a timely manner
could have a disruptive effect on ABVC and BioLite’s research and development activities of ABV-1703, ABV-1519, ABV-1502 and ABV-1501,
resulting in a material adverse effect on the Company’s business, financial condition and results of operations.
ABVC may use hazardous chemicals and biological materials in its business. Any claims relating to improper handling, storage or disposal of these materials could be time consuming and costly.
ABVC’s research and development may involve the controlled use of hazardous materials, including chemicals and biological materials. ABVC cannot eliminate the risk of accidental contamination or discharge and any resulting injury from these materials. ABVC may be sued for any injury or contamination that results from its use or the use by third parties of these materials, and its liability may exceed any insurance coverage and its total assets. Federal, state and local laws and regulations govern the use, manufacture, storage, handling and disposal of these hazardous materials and specified waste products, as well as the discharge of pollutants into the environment and human health and safety matters. Although ABVC makes its best efforts to comply with environmental laws and regulations despite the associated high costs and inconvenience, ABVC cannot guarantee that it will not mishandle any hazardous materials in the future. If it fails to comply with these requirements or any improper handling of hazardous materials occurs, it could incur substantial costs, including civil or criminal fines and penalties, clean-up costs or capital expenditures for control equipment or operational changes necessary to achieve and maintain compliance. In addition, ABVC cannot predict the impact on its business of new or amended environmental laws or regulations or any changes in the way existing and future laws and regulations are interpreted and enforced.
The facilities where the samples of drug candidates are manufactured need to be maintained and monitored in compliance with the good manufacturing practice standards, the failure of such maintenance could contaminate the results of our clinical trials and adversely affect our operations.
ABVC’s Subsidiary BioKey operates a laboratory facility that is a certified good manufacturing practice facility (“cGMP”) and some of its contract clinical trial service providers use cGMP facilities to conduct clinical studies. ABVC cannot be certain that ABVC or its present or future contract manufacturers or suppliers will be able to comply with cGMPs regulations and other FDA regulatory requirements. Failure to comply with these requirements may result in, among other things, total or partial suspension of production activities, failure of the FDA to grant approval for marketing, and withdrawal, suspension, or revocation of marketing approvals.
Risks Related to Intellectual Property
Pharmaceutical patents and patent applications involve highly complex legal and factual questions, which, if determined adversely to the Company, could negatively impact its respective licensors’ patent position and interrupt its research activities.
The patent positions of pharmaceutical companies and research institutions can be highly uncertain and involve complex legal and factual questions. The interpretation and breadth of claims allowed in some patents covering pharmaceutical compositions may be uncertain and difficult to determine, and are often affected materially by the facts and circumstances that pertain to the patented compositions and the related patent claims. The standards of the U.S. Patent and Trademark Office, or USPTO, are sometimes uncertain and could change in the future. Consequently, the issuance and scope of patents cannot be predicted with certainty. Patents, if issued, may be challenged, invalidated or circumvented. U.S. patents and patent applications may also be subject to interference proceedings, and U.S. patents may be subject to re-examination proceedings, post-grant review and/or inter parties review in the USPTO. Foreign patents may be subject to opposition or comparable proceedings in the corresponding foreign patent office, which could result in either loss of the patent or denial of the patent application or loss or reduction in the scope of one or more of the claims of the patent or patent application. In addition, such interference, re-examination, post-grant review, inter parties review and opposition proceedings may be costly. Accordingly, rights under any issued patents may not provide the Company with sufficient protection against competitive products or processes.
In addition, changes in or different interpretations of patent laws in the U.S. and foreign countries may permit others to use discoveries of the Company or to develop and commercialize their new drug candidates without providing any compensation thereto, or may limit the number of patents or claims the Company can obtain. The laws of some countries do not protect intellectual property rights to the same extent as U.S. laws and those countries may lack adequate rules and procedures for defending the intellectual property rights of the Company.
If the Company fails to obtain and maintain patent protection and trade secret protection of its respective products, the Company could lose their competitive advantages and competition it faces would increase, reducing any potential revenues and adversely affecting its ability to attain or maintain profitability.
Developments in patent law could have a negative impact on the Company’s Licensors’ patent positions and the Company’s business.
From time to time, the U.S. Supreme Court, other federal courts, the U.S. Congress or the USPTO may change the standards of patentability and any such changes could have a negative impact on the Company’s business.
In addition, the Leahy-Smith America Invents Act, or the America Invents Act, which was signed into law in 2011, includes a number of significant changes to U.S. patent law. These changes include a transition from a “first-to-invent” system to a “first-to-file” system, changes the way issued patents are challenged, and changes the way patent applications are disputed during the examination process. These changes may favor larger and more established companies that have greater resources to devote to patent application filing and prosecution. The USPTO has developed regulations and procedures to govern the full implementation of the America Invents Act, and many of the substantive changes to patent law associated with the America Invents Act, and, in particular, the first-to-file provisions, became effective on March 16, 2013. Substantive changes to patent law associated with the America Invents Act may affect the Company, BioLite and BioKey’s ability to obtain patents, and if obtained, to enforce or defend them. Accordingly, it is not clear what, if any, impact the America Invents Act will ultimately have on the cost of prosecuting the Company’s patent applications, its ability to obtain patents based on its discoveries and its ability to enforce or defend its patents.
If the Company is unable to protect the confidentiality of its trade secrets, its business and competitive position would be harmed, respectively.
In addition to patent protection, because the Company operates in the highly technical field of discovery and development of therapies, it relies in part on trade secret protection in order to protect its proprietary technology and processes. However, trade secrets are difficult to protect. The Company has entered into confidentiality and non-disclosure agreements with its employees, consultants, outside scientific and commercial collaborators, sponsored researchers, and other advisors. These agreements generally require that the other party keep confidential and not disclose to third parties any confidential information developed by the party or made known to the party by the Company during the course of the party’s relationship therewith. These agreements also generally provide that inventions conceived by the party in the course of rendering services to the Company will be ABVC’s exclusive property. However, these agreements may not be honored and may not effectively assign intellectual property rights to the Company.
In addition to contractual measures, the Company tries to protect the confidential nature of its proprietary information using physical and technological security measures. Such measures may not, for example, in the case of misappropriation of a trade secret by an employee or third party with authorized access, provide adequate protection for the Company. The Company’s security measures may not prevent an employee or consultant from misappropriating its trade secrets and providing them to a competitor, and recourse it takes against such misconduct may not provide an adequate remedy to protect the Company’s interests fully. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret can be difficult, expensive, and time-consuming, and the outcome is unpredictable. In addition, courts outside the U.S. may be less willing to protect trade secrets. Trade secrets may be independently developed by others in a manner that could prevent legal recourse by the Company. If the Company’s confidential or proprietary information, such as the trade secrets, were to be disclosed or misappropriated, or if any such information was independently developed by a competitor, its competitive position could be harmed.
Third parties may assert that the Company’s employees or consultants have wrongfully used or disclosed confidential information or misappropriated trade secrets.
The Company might employ individuals who were previously employed at universities or other biopharmaceutical companies, including its competitors or potential competitors. Although through certain non-disclosure covenants and employment agreements with its officers and employees, the Company tries to ensure that its employees and consultants do not use the proprietary information or know-how of others in the work for the Company, the Company may be subject to claims that it or its employees, consultants or independent contractors have inadvertently or otherwise used or disclosed intellectual property, including trade secrets or other proprietary information, of a former employer or other third parties. Litigation may be necessary to defend against these claims. If the Company fails in defending any such claims, in addition to paying monetary damages, the Company may lose valuable intellectual property rights or personnel. Even if the Company is successful in defending against such claims, litigation could result in substantial costs and be a distraction to the Company’s management and other employees.
ABVC’s ability to compete may decline if it does not adequately protect its proprietary rights or if it is barred by the intellectual property rights of others.
ABVC’s commercial success depends on obtaining and maintaining proprietary rights to its drug candidates as well as successfully defending these rights against third-party challenges. ABVC obtains its rights to use and research certain proprietary information to further develop the drug candidates primarily from three institutions, MPITDC, ITRI and Yukiguni (collectively the “Licensors”). These three institutions own the intellectual property rights in the products that have been licensed to us and may prosecute new patents of the drug candidates that are invented or discovered within the licensed scope of use under the respective license agreements. ABVC will only be able to protect its new drug candidates from unauthorized use by third parties to the extent that its valid and enforceable patents, or effectively protected trade secrets and know-how, cover them.
ABVC’s ability to obtain new patent protection for its new drug candidates is uncertain due to a number of factors, including that:
|
● |
ABVC may not have been the first to make the inventions covered by pending patent applications or issued patents; |
|
● |
ABVC may not have been the first to file patent applications for its new drug candidates; |
|
● |
others may independently develop identical, similar or alternative products or compositions and uses thereof; |
|
● |
ABVC’s disclosures in patent applications may not be sufficient to meet the statutory requirements for patentability; |
|
● |
any or all of ABVC’s pending patent applications may not result in issued patents; |
|
● |
ABVC may not seek or obtain patent protection in countries that may eventually provide a significant business opportunity; |
|
● |
any patents issued to ABVC may not provide a basis for commercially viable products, may not provide any competitive advantages, or may be successfully challenged by third parties; |
|
● |
ABVC’s methods may not be patentable; |
|
● |
ABVC’s licensors may successfully challenge that ABVC’s new patent application fall outside the licensed use of the products; or |
|
● |
others may design around ABVC’s patent claims to produce competitive products which fall outside of the scope of its patents. |
Even if ABVC has or obtains new patents covering its new drug candidates, ABVC may still be barred from making, using and selling them because of the patent rights of others. Others may have filed, and in the future may file, patent applications covering products that are similar or identical to ABVC. There are many issued U.S. and foreign patents relating to therapeutic products and some of these relate to ABVC’s new drug candidates. These could materially affect ABVC’s ability to develop its drug candidates. Because patent applications can take many years to issue, there may be currently pending applications unknown to ABVC that may later result in issued patents that its new drug candidates may infringe. These patent applications may have priority over patent applications filed by ABVC.
The Company and its respective licensors may not be able to enforce their intellectual property rights throughout the world.
The laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the U.S. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. The legal systems of some countries, particularly developing countries, do not favor the enforcement of patents and other intellectual property protection, especially those relating to pharmaceuticals and medical devices. This could make it difficult for the Company and its respective licensors to stop the infringement of some of the Licensors’ patents, or the misappropriation of their other intellectual property rights. For example, many foreign countries have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, many countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time-consuming process with uncertain outcomes. Accordingly, the Company and its licensors have chosen in the past and may choose in the future not to seek patent protection in certain countries, and as a result the Company will not have the benefit of patent protection in such countries. Moreover, the Company may choose in the future not to seek patent protection in certain countries, and as a result it will not have the benefit of patent protection in such countries.
Proceedings to enforce the Company’s and its licensors’ patent rights in foreign jurisdictions could result in substantial costs and divert its efforts and attention from other aspects of the businesses. Accordingly, the efforts to protect the Company’s intellectual property rights in such countries may be inadequate. In addition, changes in the law and legal decisions by courts in the U.S. and foreign countries may affect the Company’s ability to obtain adequate protection for its technology and the enforcement of intellectual property.
Regulatory Risks Relating to Biopharmaceutical Business
The Company is subject to various government regulations.
The manufacture and sale of human therapeutic and diagnostic products in the U.S. and foreign jurisdictions are governed by a variety of statutes and regulations. These laws require approval of manufacturing facilities, controlled research and testing of products and government review and approval of a submission containing manufacturing, preclinical and clinical data in order to obtain marketing approval based on establishing the safety and efficacy of the product for each use sought, including adherence to current PIC/S Guide to Good Manufacturing Practice for Medicinal products during production and storage, and control of marketing activities, including advertising and labeling.
The products the Company is currently developing will require significant development, preclinical and clinical testing and investment of substantial funds prior to its commercialization. The process of obtaining required approvals can be costly and time-consuming, and there can be no assurance that future products will be successfully developed and will prove to be safe and effective in clinical trials or receive applicable regulatory approvals. Markets other than the U.S. have similar restrictions. Potential investors and shareholders should be aware of the risks, problems, delays, expenses and difficulties which we may encounter in view of the extensive regulatory environment which controls our business.
The Company cannot be certain that it will be able to obtain regulatory approval for, or successfully commercialize, any of its current or future product candidates.
The Company may not be able to develop any current or future product candidates. The Company’s new drug candidates will require substantial additional clinical development, testing, and regulatory approval before the commencement of commercialization. The clinical trials of the Company’s drug candidates are, and the manufacturing and marketing of our new drug candidates will be subject to extensive and rigorous review and regulation by numerous government authorities in the U.S. and in other countries where the Company intend to test and, if approved, market any new drug candidate. Before obtaining regulatory approvals for the commercial sale of any product candidate, the Company must demonstrate through pre-clinical testing and clinical trials that the product candidate is safe and effective for use in each target indication. This process can take many years and may include post-marketing studies and surveillance, which will require the expenditure of substantial resources. Of the large number of drugs in development in the U.S., only a small percentage successfully completes the FDA regulatory approval process and is commercialized. Accordingly, even if the Company is able to obtain the requisite financing to continue to fund its development and clinical programs, it cannot assure the investors that any of the product candidates will be successfully developed or commercialized.
The Company is not permitted to market a therapeutic product in the U.S. until it receives approval of an NDA or ANDA, for that product from the FDA, or in any foreign countries until they receive the requisite approval from such countries. Obtaining approval of an NDA is a complex, lengthy, expensive and uncertain process, and the FDA may delay, limit or deny approval of any product candidate for many reasons, including, among others:
|
● |
Unable to demonstrate that a product candidate is safe and effective to the satisfaction of the FDA; |
|
● |
the results of the Company’s clinical trials may not meet the level of statistical or clinical significance required by the FDA for marketing approval; |
|
● |
the FDA may not approve the formulation of any product candidate; |
|
● |
the CROs, that BioLite or the Company retains to conduct its clinical trials may take actions outside of its control that materially adversely impact its clinical trials; |
|
● |
delays in patient enrollment, variability in the number and types of patients available for clinical trials, and lower-than anticipated retention rates for patients in clinical trials; |
|
● |
the FDA may find the data from pre-clinical studies and clinical trials insufficient to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks, such as the risk of drug abuse by patients or the public in general; |
|
● |
the FDA may disagree with the interpretation of data from the Company’s pre-clinical studies and clinical trials; |
|
● |
the FDA may not accept data generated at the Company’s clinical trial sites; |
|
● |
if an NDA, if and when submitted, is reviewed by an advisory committee, the FDA may have difficulties scheduling an advisory committee meeting in a timely manner or the advisory committee may recommend against approval of our application or may recommend that the FDA require, as a condition of approval, additional pre-clinical studies or clinical trials, limitations on approved labeling or distribution and use restrictions; |
|
● |
the FDA may require development of a Risk Evaluation and Mitigation Strategy, or REMS, as a condition of approval or post-approval; or |
|
● |
the FDA may change its approval policies or adopt new regulations. |
These same risks apply to applicable foreign regulatory agencies from which the Company, through BioLite, may seek approval for any of our new drug candidates.
Any of these factors, many of which are beyond the Company’s control, could jeopardize its ability to obtain regulatory approval for and successfully market any new drug candidate. As a result, any such setback in the Company’s pursuit of initial or additional regulatory approval would have a material adverse effect on its business and prospects.
If the Company does not successfully complete pre-clinical and Phase I and II clinical development, it will be unable to receive full payments under their respective collaboration agreements, find future collaborators or partners to take the drug candidates to Phase III clinical trials. Even if the Company successfully completes all Phase I and II clinical trials, those results are not necessarily predictive of results of additional trials that may be needed before an NDA for Phase III trials may be submitted to the FDA. Although there are a large number of drugs in development in the U.S. and other countries, only a very small percentage result in commercialization, and even fewer achieve widespread physician and consumer acceptance following the regulatory approval.
In addition, the Company may encounter delays or drug candidate rejections based on new governmental regulations, future legislative or administrative actions, or changes in FDA policy or interpretation during the period of product development. If the Company obtains required regulatory approvals, such approvals may later be withdrawn. Delays or failures in obtaining regulatory approvals may result in:
|
● |
varying interpretations of data and commitments by the FDA and similar foreign regulatory agencies; and |
|
● |
diminishment of any competitive advantages that such drug candidates may have or attain. |
Furthermore, if the Company fails to comply with applicable FDA and other regulatory requirements at any stage during this regulatory process, the Company may encounter or be subject to:
|
● |
delays or termination in clinical trials or commercialization; |
|
● |
refusal by the FDA or similar foreign regulatory agencies to review pending applications or supplements to approved applications; |
|
● |
product recalls or seizures; |
|
● |
suspension of manufacturing; |
|
● |
withdrawals of previously approved marketing applications; and |
|
● |
fines, civil penalties, and criminal prosecutions. |
The Company faces substantial competition from companies with considerably more resources and experience than the Company has, which may result in others discovering, developing, receiving approval for, or commercializing products before or more successfully than the Company.
The Company competes with companies that research, develop, manufacture and market already-existing and new pharmaceutical products in the fields of CNS, hematology/oncology and autoimmune. The Company anticipates that it will face increased competition in the future as new companies enter the market with new drugs and/or technologies and/or their competitors improve their current products. One or more of their competitors may offer new drugs superior to the Company’s and render the Company’s drugs uneconomical. A lot of the Company’s current competitors, as well as many of its respective potential competitors, have greater name recognition, more substantial intellectual property portfolios, longer operating histories, significantly greater resources to invest in new drug development, more substantial experience in product marketing and new product development, greater regulatory expertise, more extensive manufacturing capabilities and the distribution channels to deliver products to customers. If the Company is not able to compete successfully, it may not generate sufficient revenue to become profitable. The Company’s ability to compete successfully will depend largely on its ability to:
|
● |
successfully commercialize its drug candidates with commercial partners; |
|
● |
discover and develop new drug candidates that are superior to other products in the market; |
|
● |
with its collaborators, obtain required regulatory approvals; |
|
● |
attract and retain qualified personnel; and |
|
● |
obtain patent and/or other proprietary protection for its product candidates. |
Established pharmaceutical companies devote significant financial resources to discovering, developing or licensing novel compounds that could make the Company’s products and product candidates obsolete. Our competitors may obtain patent protection, receive FDA approval, and commercialize medicines before we do. Other companies are or may become engaged in the discovery of compounds or botanical materials that may compete with the drug candidates the Company is developing.
The Company competes with a large number of well-established pharmaceutical companies that may have more resources than the Company does in developing therapeutics in the fields of CNS, oncology/hematology and ophthalmology.
Any new drug candidate the Company is developing or commercializing that competes with a currently-approved product must demonstrate compelling advantages in efficacy, convenience, tolerability and/or safety in order to address price competition and be commercially successful. If the Company is not able to compete effectively against its current and future competitors, its business will not grow and its financial condition and operations will suffer.
Risks Relating to Doing Business Outside the United States
Because part of ABVC’s pharmaceutical research and development is conducted outside of the U.S., the Company is subject to the risks of doing business internationally, including periodic foreign economic downturns and political instability, which may adversely affect the Company’s revenue and cost of doing business in Taiwan.
ABVC collaborates with partners whose primary place of business is in Taiwan, Republic of China and the Company has certain key employees in Taiwan. Foreign economic downturns may affect our results of operations in the future. Additionally, other facts relating to the operation of the Company’s business outside of the U.S. may have a material adverse effect on the Company’s business, financial condition and results of operations, including:
|
● |
international economic and political changes; |
|
● |
the imposition of governmental controls or changes in government regulations, including tax laws, regulations and treaties; |
|
● |
changes in, or impositions of, legislative or regulatory requirements regarding the pharmaceutical industry; |
|
● |
compliance with U.S. and international laws involving international operations, including the Foreign Corrupt Practices Act and export control laws; |
|
● |
difficulties in achieving headcount reductions due to unionized labor and works councils; |
|
● |
restrictions on transfers of funds and assets between jurisdictions; and |
|
● |
China- Taiwan geo-political instability. |
As the Company continues to operate its business globally, its success will depend in part, on its ability to anticipate and effectively manage these risks. The impact of any one or more of these factors could materially adversely affect the Company’s business, financial condition and results of operations.
The Company may be exposed to liabilities under the U.S. Foreign Corrupt Practices Act (“FCPA”) and Chinese anti-corruption law.
The Company is subject to the FCPA, and other laws that prohibit improper payments or offers of payments to foreign governments, foreign government officials and political parties by U.S. persons as defined by the statute for purposes of obtaining or retaining businesses. The Company may have agreements with third parties who may make sales in mainland China and the U.S., during the process of which the Company may be exposed to corruption. Activities in Taiwan create the risk of unauthorized payments or offers of payments by an employee, consultant or agent of the Company, because these parties are not always subject to the Company’s control.
Although the Company believes to date it has complied in all material aspects with the provisions of the FCPA and Chinese anti-corruption law, the existing safeguards and any future improvements may prove to be less than effective and any of the Company’s employees, consultants or agents may engage in corruptive conduct for which the Company might be held responsible. Violations of the FCPA or Chinese anti-corruption law may result in severe criminal or civil sanctions against the Company and individuals and therefore could negatively affect the Company’s business, operating results and financial condition. In addition, the Taiwanese government may seek to hold the Company liable as a successor for FCPA violations committed by companies in which the Company invests or acquires.
International operations expose the Company to currency exchange and repatriation risks, and the Company cannot predict the effect of future exchange rate fluctuations on its business and operating results.
The Company has business operations in Taiwan and collaborative activities in the U.S. and Japan. Substantial amounts of revenues are received and expenses are incurred in New Taiwan Dollars and U.S. dollars. Thus, the Company has exposure to currency fluctuations. The Company cannot assure you that the effect of currency exchange fluctuations will not materially affect its revenues and net income in the future.
We conduct our operations internationally and the effect of business, legal and political risks associated with international operations may seriously harm our business.
Sales to customers outside the United States
accounted for 93 % and 66% for the year ended December 31, 2023 and 2022, respectively. Our international sales and operations are subject
to a wide range of risks, which may vary from country to country or region to region. These risks include the following:
|
● |
export and import duties, changes to import and export regulations, and restrictions on the transfer of funds; |
|
● |
political and economic instability; |
|
● |
issues arising from cultural or language differences and labor unrest; |
|
● |
longer payment cycles and greater difficulty in collecting accounts receivable; |
|
● |
compliance with trade and
technical standards in a variety of jurisdictions; |
|
● |
difficulties in staffing
and managing international operations, including the risks associated with fraud, theft and other illegal conduct; |
|
● |
compliance with laws and
regulations, including environmental, employment and tax laws, which vary from country to country and over time, increasing the costs
of compliance and potential risks of non-compliance; |
|
● |
difficulties enforcing
our contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual
property rights to the same extent as the United States and European countries; |
|
● |
operations may be affected
by political tensions, trade disputes and similar matters, particularly between China and Taiwan or between China and the United
States; |
|
● |
United States and foreign
trade restrictions, including those that may limit the importation of technology or components to or from various countries or impose
tariffs or quotas; and |
|
● |
imposition of currency
exchange controls or taxes that make it impracticable or costly to repatriate funds from foreign countries. |
We cannot assure you that risks relating to our international operations will not seriously harm our business.
If the Company becomes directly subject to the recent scrutiny, criticism and negative publicity involving U.S.-listed Chinese companies, we may have to expend significant resources to investigate and resolve the matters. Any unfavorable results from the investigations could harm our business operations, this offering and our reputation.
Recently, U.S. public companies that have substantially all of their operations in China, have been subjects of intense scrutiny, criticism and negative publicity by investors, financial commentators and regulatory agencies, such as the SEC. Much of the scrutiny, criticism and negative publicity has centered on financial and accounting irregularities, lack of effective internal control over financial accountings, inadequate corporate governance and ineffective implementation thereof and, in many cases, allegations of fraud. As a result of enhanced scrutiny, criticism and negative publicity, the publicly traded stocks of many U.S. listed Chinese companies have sharply decreased in value and, in some cases, have become virtually worthless or illiquid. Many of these companies are now subject to shareholder lawsuits and SEC enforcement actions and are conducting internal and external investigations into the allegations. It is not clear what effects the sector-wide investigations will have on the Company. If the Company becomes a subject of any unfavorable allegations, whether such allegations are proven to be true or untrue, the Company will have to expend significant resources to investigate such allegations and defend the Company. If such allegations were not proven to be baseless, the Company would be severely hampered and the price of the stock of the Company could decline substantially. If such allegations were proven to be groundless, the investigation might have significantly distracted the attention of the Company’s management.
Risks Related to the Company’s Financial Condition
Our existing indebtedness may adversely affect our ability to obtain additional funds and may increase our vulnerability to economic or business downturns.
We are subject to a number of risks associated
with our indebtedness, including: 1) we must dedicate a portion of our cash flows from operations to pay debt service costs, and therefore
we have less funds available for operations and other purposes; 2) it may be more difficult and expensive to obtain additional funds
through financings, if available at all; 3) we are more vulnerable to economic downturns and fluctuations in interest rates, less able
to withstand competitive pressures and less flexible in reacting to changes in our industry and general economic conditions; and 4) if
we default under any of our existing credit facilities or if our creditors demand payment of a portion or all of our indebtedness, we
may not have sufficient funds to make such payments. As of December 31, 2023, our outstanding current liabilities were approximately
$5.6 million, respectively, which consisted primarily of short-term bank loans and accrued expenses. On April 5 and 20, 2020, we entered
into certain exchange agreements separately with certain U.S. and non-U.S. holders of certain convertible promissory notes in the aggregate
amount of $1,446,780; pursuant to the exchange agreements, we issued to the Holders an aggregate of 795,735 shares of Common Stock and
warrants to purchase 795,735 shares of Common Stock. On November 9, 2020, we entered into an exchange agreement with a certain non-U.S.
holder of certain convertible promissory notes in the amount of $270,272; pursuant to the exchange agreements, we will issue to the holder
an aggregate of 120,121 shares of Common Stock and warrants to purchase 120,121 shares of Common Stock. We also agreed to issue an aggregate
of 545,182 options of common stock to some of our employees in lieu of their deferred salaries in an aggregate amount of $1,090,360.
Failure to remediate a material weakness
in internal accounting controls could result in material misstatements in our financial statements.
Our management has identified a material weakness
in our internal control over financial reporting related to not having sufficient and skilled accounting personnel with appropriate level
of technical accounting knowledge and experience in the application of accounting principles generally accepted in the United States
commensurate with the Company’s financial reporting requirements and has concluded that, due to such material weakness, our disclosure
controls and procedures were not effective as of December 31, 2023. If not remediated, or if we identify further material weaknesses
in our internal controls, our failure to establish and maintain effective disclosure controls and procedures and internal control over
financial reporting could result in material misstatements in our financial statements and a failure to meet our reporting and financial
obligations, each of which could have a material adverse effect on our financial condition and the trading price of our common stock.
Failure to maintain the effectiveness of our disclosure controls and procedures may lead to restatement of our financial statements, harm our operating results, subject us to regulatory scrutiny and sanction, cause investors to lose confidence in our reported financial information and have a negative effect on the market prices for our Common Stock.
The Sarbanes-Oxley Act of 2002 and the Securities and Exchange Commission (SEC) have requirements that we may fail to meet or we may fall out of compliance with, such as the internal controls auditor attestation required under Section 404 of the Sarbanes-Oxley Act of 2002, with which we are not currently required to comply as we are a smaller reporting company. If we fail to achieve and maintain the adequacy of our internal controls, as such standards are modified, supplemented or amended from time to time, we may not be able to ensure that we can conclude on an ongoing basis that we have effective internal controls over financial reporting in accordance with Section 404 of the Sarbanes-Oxley Act of 2002. Moreover, effective internal controls, particularly those related to revenue recognition, are necessary for us to produce reliable financial reports and are important to help prevent financial fraud. If we cannot provide reliable financial reports or prevent fraud, our business and operating results could be harmed, investors could lose confidence in our reported financial information, and the trading price of our stock could drop significantly.
Our articles of incorporation allow for our board to create new series of preferred stock without further approval by our shareholders, which could adversely affect the rights of the holders of our Common Stock.
Our Board of Directors has the authority to fix and determine the relative rights and preferences of preferred stock without shareholder approval. As a result, our Board of Directors could authorize the issuance of a series of preferred stock that would grant to holders the preferred right to our assets upon liquidation, the right to receive dividend payments before dividends are distributed to the holders of Common Stock and the right to the redemption of the shares, together with a premium, prior to the redemption of our Common Stock. In addition, our Board of Directors could authorize the issuance of a series of preferred stock that has greater voting power than our Common Stock or that is convertible into our Common Stock, which could decrease the relative voting power of our Common Stock or result in dilution to our existing shareholders.
We may create any additional series of preferred stock and issue such shares in the future although we do not have any present intention of doing so.
We may not be able to secure financing needed for future operating needs on acceptable terms, or on any terms at all.
From time to time, we may seek additional financing to provide the capital required to expand our production facilities, Research and development (“R&D”) initiatives and/or working capital, as well as to repay outstanding loans if cash flow from operations is insufficient to do so. We cannot predict with certainty the timing or amount of any such capital requirements. If such financing is not available on satisfactory terms, we may be unable to expand our business or to develop new business at the rate desired. If we are able to incur debt, we may be subject to certain restrictions imposed by the terms of the debt and the repayment of such debt may limit our cash flow and growth. If we are unable to incur debt, we may be forced to issue additional equity, which could have a dilutive effect on our current shareholders.
Our internal computer systems, or those of our third-party contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs.
Despite the implementation of security measures, our internal computer systems and those of our third-party contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we do not believe that we have experienced any such system failure, accident, or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a loss of clinical trial data for our new drug candidates which could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications or other data or applications relating to our technology or new drug candidates, or inappropriate disclosure of confidential or proprietary information, we could incur liabilities and the further development of our product candidates could be delayed.
The elimination of personal liability against our directors and officers under Nevada law and the existence of indemnification rights held by our directors, officers and employees may result in substantial expenses.
ABVC Bylaws eliminate the personal liability of our directors and officers to us and our shareholders for damages for breach of fiduciary duty as a director or officer to the extent permissible under Nevada law. Further, our Bylaws provide that we are obligated to indemnify each of our directors or officers to the fullest extent authorized by Nevada law and, subject to certain conditions, advance the expenses incurred by any director or officer in defending any action, suit or proceeding prior to its final disposition. Those indemnification obligations could expose us to substantial expenditures to cover the cost of settlement or damage awards against our directors or officers, which we may be unable to afford. Further, those provisions and resulting costs may discourage us or our shareholders from bringing a lawsuit against any of our current or former directors or officers for breaches of their fiduciary duties, even if such actions might otherwise benefit our shareholders.
Risks Related to the Company’s Common Stock
The share price of our Common Stock is volatile and may be influenced by numerous factors, some of which are beyond our control.
There is currently only a limited public market for our Common Stock, which is listed on the Nasdaq Capital Market, and there can be no assurance that a trading market will develop further or be maintained for our Common Stock in the future. The trading price of our Common Stock is likely to be highly volatile, and could be subject to wide fluctuations in response to various factors, some of which are beyond our control. In addition to the factors discussed in this “Risk Factors” section and elsewhere herein, these factors include:
|
● |
the new drug candidates we acquire for commercialization; |
|
● |
the product candidates we seek to pursue, and our ability to obtain rights to develop those product candidates; |
|
● |
our decision to initiate a clinical trial, not to initiate a clinical trial or to terminate an existing clinical trial; |
|
● |
actual or anticipated adverse results or delays in our pre-clinical studies and clinical trials; |
|
● |
our failure to get any of our new drug candidates approved; |
|
● |
unanticipated serious safety and environmental concerns related to the use and research activities of any of our new drug candidates; |
|
● |
overall performance of the equity markets and other factors that may be unrelated to our operating performance or the operating performance of our competitors, including changes in market valuations of similar companies; |
|
● |
conditions or trends in the healthcare, biotechnology and pharmaceutical industries; |
|
● |
introduction of new products offered by us or our competitors; |
|
● |
announcements of significant acquisitions, strategic partnerships, joint ventures or capital commitments by us or our competitors; |
|
● |
our ability to maintain an adequate rate of growth and manage such growth; |
|
● |
issuances of debt or equity securities by us; |
|
● |
sales of our securities by us or our shareholders in the future, or the perception that such sales could occur; |
|
● |
trading volume of our Common Stock; |
|
● |
ineffectiveness of our internal control over financial reporting or disclosure controls and procedures; |
|
● |
general political and economic conditions in U.S. and other countries and territories where we conduct our business; |
|
● |
effects of natural or man-made catastrophic events; and |
|
● |
adverse regulatory decisions; |
|
● |
additions or departures of key scientific or management personnel; |
|
● |
changes in laws or regulations applicable to our product candidates, including without limitation clinical trial requirements for approvals; |
|
● |
disputes or other developments relating to patents and other proprietary rights and our ability to obtain protection for our products; |
|
● |
our dependence on third parties, including CROs and scientific and medical advisors; |
|
● |
failure to meet or exceed any financial guidance or expectations regarding development milestones that we may provide to the public; |
|
● |
actual or anticipated variations in quarterly operating results; |
|
● |
failure to meet or exceed the estimates and projections of the investment community; |
|
● |
other events or factors, many of which are beyond our control. |
In addition, the stock market in general, and the stocks of small-cap healthcare, biotechnology and pharmaceutical companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our Common Stock, regardless of our actual operating performance. The realization of any of the above risks or any of a broad range of other risks, including those described in these “Risk Factors,” could have a dramatic and material adverse impact on the market price of our Common Stock.
Insiders have substantial control over us, and they could delay or prevent a change in our corporate control even if our other shareholders wanted it to occur.
Our
executive officers, directors, and principal shareholders own, in the aggregate, approximately 61.4% of our outstanding Common Stock.
As a result of their stockholdings, these shareholders are able to assert substantial control over matters requiring shareholder approval,
including the election of directors and approval of significant corporate transactions. This could delay or prevent an outside party
from acquiring or merging with us even if our other shareholders wanted it to occur.
The market price of our Common Stock may be volatile and there may not be sufficient liquidity in the market for our securities in order for investors to sell their securities.
The market price of our Common Stock has been and will likely continue to be highly volatile, as is the stock market in general. Factors that may materially affect the market price of our Common Stock are beyond our control, these factors may materially adversely affect the market price of our Common Stock, regardless of our performance. In addition, the public stock markets have experienced extreme price and trading volume volatility. These broad market fluctuations may influence the market price of our Common Stock. There is currently only a limited public market for our Common Stock, which is listed on the Nasdaq Capital Market, and there can be no assurance that a trading market will develop further or be maintained in the future.
The stock markets have experienced extreme price and volume fluctuations that have affected and continue to affect the market prices of equity securities of many companies, including very recently in connection with the ongoing COVID-19 pandemic, which has resulted in decreased stock prices for many companies notwithstanding the lack of a fundamental change in their underlying business models or prospects. These fluctuations have often been unrelated or disproportionate to the operating performance of those companies. Broad market and industry factors, including potentially worsening economic conditions and other adverse effects or developments relating to the ongoing COVID-19 pandemic, political, regulatory and other market conditions, may negatively affect the market price of shares of our common stock, regardless of our actual operating performance. The market price of shares of our common stock may decline and you may lose some or all of your investment.
We have not paid dividends in the past and do not expect to pay dividends in the future, and any return on investment may be limited to the value of our shares.
We have never paid any cash dividends on our Common Stock and do not anticipate paying any cash dividends in the foreseeable future, and any return on investment may be limited to the value of our Common Stock. We plan to retain any future earnings to finance growth.
Under applicable Nevada law, we, as a Nevada corporation, generally may not make a distribution if i) we would not be able to pay our debts as they become due in the usual course of business, or ii) our total assets would be less than the sum of our total liabilities plus the amount that would be needed, if we were to be dissolved at the time of distribution, to satisfy the preferential rights upon dissolution of shareholders whose preferential rights are superior to those receiving the distribution.
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and any trading volume could decline.
Any trading market for our Common Stock that may develop will depend in part on the research and reports that securities or industry analysts publish about us or our business. As of the date hereof, there is only 1 publish research report about our business. If securities or industry analysts provide additional coverage, and one or more of those analysts downgrade our stock or publish inaccurate or unfavorable research about our business, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our Common Stock could decrease, which might cause our stock price and any trading volume to decline.
Future sales and issuances of our Common Stock or rights to purchase Common Stock, including pursuant to our equity incentive plan or otherwise, could result in dilution of the percentage ownership of our shareholders and could cause our stock price to fall.
We expect that we will need significant additional
capital in the future to continue our planned operations. To raise capital, we may sell Common Stock, convertible securities or other
equity securities in one or more transactions at prices and in a manner we determine from time to time. If we sell Common Stock, convertible
securities or other equity securities in more than one transaction, including issuance of equity securities pursuant to any future stock
incentive plan to our officers, directors, employees and non-employee consultants for their services to us, investors in a prior transaction
may be materially diluted by subsequent sales. Additionally, any such sales may result in material dilution to our existing shareholders,
and new investors could gain rights, preferences and privileges senior to those of holders of our Common Stock. Further, any future sales
of our Common Stock by us or resales of our Common Stock by our existing shareholders could cause the market price of our Common Stock
to decline. Any future grants of options, warrants or other securities exercisable or convertible into our Common Stock, or the exercise
or conversion of such shares, and any sales of such shares in the market, could have an adverse effect on the market price of our Common
Stock. On October 29, 2021, we filed a registration statement on Form S-3, as amended on November 16, 2021, which was declared effective
on November 29, 2021. On May 11, 2022, we agreed to issue 2,000,000 shares of Common Stock, par value $0.001 per share, at a price of
$2.11 per share and 5-year warrants to purchase up to 2,000,000 shares of Common Stock, exercisable at a price of $2.45 per share pursuant
to certain securities purchase agreement dated May 11, 2022, which was effected as a takedown off the Company’s shelf registration
statement on Form S-3, as amended. We also issued the co-placement agents warrants to purchase up to 160,000 shares of Common Stock,
on the same terms as the investors warrants in connection with the transaction. We may issue shares of Common Stock through the Form
S-3 in the future, which would further dilute your ownership.
Our Common Stock may be subject to the “penny stock” rules of the Securities and Exchange Commission, which may make it more difficult for shareholders to sell our Common Stock.
The SEC has adopted Rule 15g-9 which establishes the definition of a “penny stock,” for the purposes relevant to us, as any equity security that has a market price of less than $5.00 per share, subject to certain exceptions. For any transaction involving a penny stock, unless exempt, the rules require that a broker or dealer approve a person’s account for transactions in penny stocks, and the broker or dealer receive from the investor a written agreement to the transaction, setting forth the identity and quantity of the penny stock to be purchased.
In order to approve a person’s account for transactions in penny stocks, the broker or dealer must obtain financial information and investment experience objectives of the person, and make a reasonable determination that the transactions in penny stocks are suitable for that person and the person has sufficient knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks.
The broker or dealer must also deliver, prior to any transaction in a penny stock, a disclosure schedule prescribed by the SEC relating to the penny stock market, which, in highlight form sets forth the basis on which the broker or dealer made the suitability determination, and that the broker or dealer received a signed, written agreement from the investor prior to the transaction.
Generally, brokers may be less willing to execute transactions in securities subject to the “penny stock” rules. This may make it more difficult for investors to dispose of the Company’s Common Stock if and when such shares are eligible for sale and may cause a decline in the market value of its stock.
Disclosure also has to be made about the risks of investing in penny stocks in both public offerings and in secondary trading and about the commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and the rights and remedies available to an investor in cases of fraud in penny stock transactions. Finally, monthly statements have to be sent disclosing recent price information for the penny stock held in the account and information on the limited market in penny stock.
Our failure to meet the continued listing requirements of the Nasdaq Capital Market could result in a delisting of our Common Stock.
If we fail to satisfy the continued listing
requirements of the Nasdaq Capital Market, such as the corporate governance requirements or the minimum closing bid price requirement,
the Nasdaq Capital Market may take steps to delist our common stock. Such a delisting would likely have a negative effect on the price
of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so.
On August 19, 2022, we received a deficiency
letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq Stock Market LLC (“Nasdaq”)
notifying us that, for the last 30 consecutive business days, the closing bid price for our common stock was below the minimum $1.00
per share required for continued listing on The Nasdaq Capital Market pursuant to Nasdaq Listing Rule 5550(a)(2) (“Rule 5550(a)(2)”).
Under the Nasdaq Listing Rules, we have until February 14, 2023 to regain compliance. Since we did not regain compliance by such date,
we requested and received an additional 180 days, until August 14, 2023, to comply with Rule 5550(a)(2).
On May 24, 2023,
the Company received a deficiency letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq Stock
Market LLC (“Nasdaq”) notifying the Company that it is not currently in compliance with the minimum stockholders’ equity
requirement, or the alternatives of market value of listed securities or net income from continuing operations, for continued listing
on the Nasdaq Capital Market. Nasdaq Listing Rule 5550(b)(1) requires listed companies to maintain stockholders’ equity of at least
$2,500,000, and the Company’s stockholders’ equity was $1,734,507 as of March 31, 2023. In accordance with Nasdaq rules,
the Company had 45 calendar days, or until July 10, 2023, to submit a plan to regain compliance. After submitting a plan to regain
compliance, on July 10, 2023,Nasdaq granted the Company an extension until August 30, 20203, to comply with Listing Rule 5550(b)(1). On
July 31, 2023, the Company issued 300,000 shares of Common Stock and 200,000 pre-funded warrants, at an exercise price of $0.01 per share,
in a registered direct offering. Pursuant to this transaction, the stockholders’ equity was increased by $1.75M. On August 1, 2023,
$500,000 of Notes were converted at $3.50 per share and the holder received 142,857 shares of Common Stock. As a result of this conversion,
the stockholders’ equity was increased by $0.5M. Additionally, on August 14, 2023, the Company entered into a cooperation agreement
with Zhonghui United Technology (Chengdu) Group Co., Ltd., pursuant to which the Company acquired a 20% ownership of certain property
and a parcel of the land owned by Zhonghui in exchange for an aggregate of 370,000 shares of Common Stock. Accordingly, stockholders’
equity increased by $7.4M. On February 23, 2023, the Company entered into a securities purchase agreement with Lind,
pursuant to which the Company issued Lind a secured, convertible note in the principal amount of $3,704,167 (the “Lind Offering”),
for a purchase price of $3,175,000 (the “Lind Note”), that is convertible into shares of Common Stock at an initial conversion
price of $1.05 per share, subject to adjustment. On August 24, 2023, the Company started repaying Lind the monthly installments
due under the Lind Notes; $308,000 was repaid via the issuance of 176,678 shares of Common Stock (the “Monthly Shares”) at
the Redemption Share Price (as defined in the Lind Note) of $1.698 per share. Pursuant to the terms of the Lind Note, Lind increased
the amount of the next monthly payment to one million dollars, such that as of September and together with the Monthly Shares, the Company
repaid Lind a total of $1M by September 2023. As a result, the stockholders’ equity increased by an additional $1M. As a result
of the four transactions referenced above, the Company’ estimated that its stockholders’ equity would increase by approximately
$10.65M. On September 6, 2023, Nasdaq issued a letter that the Company is in compliance with Rule 5550(b)(1), but noted that if at the
time of the Company’s next periodic report the Company does not evidence compliance, it may be subject to delisting.
If our common stock were delisted from the
Nasdaq, trading of our common stock would most likely take place on an over-the-counter market established for unlisted securities, such
as the OTCQB or the Pink Market maintained by OTC Markets Group Inc. An investor would likely find it less convenient to sell, or to
obtain accurate quotations in seeking to buy, our common stock on an over-the-counter market, and many investors would likely not buy
or sell our common stock due to difficulty in accessing over-the-counter markets, policies preventing them from trading in securities
not listed on a national exchange or other reasons. In addition, as a delisted security, our common stock would be subject to SEC rules
as a “penny stock,” which impose additional disclosure requirements on broker-dealers. The regulations relating to penny
stocks, coupled with the typically higher cost per trade to the investor of penny stocks due to factors such as broker commissions generally
representing a higher percentage of the price of a penny stock than of a higher-priced stock, would further limit the ability of investors
to trade in our common stock.
In the event of a delisting, we anticipate
that we would take actions to restore our compliance with the Nasdaq Capital Market or another national exchange’s listing requirements,
but we can provide no assurance that any such action taken by us would allow our Common Stock to remain listed on the Nasdaq Capital
Market, stabilize our market price, improve the liquidity of our common stock, prevent our common stock from dropping below the Nasdaq
Capital Market’s minimum bid price requirement, or prevent future non-compliance with the Nasdaq Capital Market or another national
exchange’s listing requirements.
We will continue to incur significant increased costs as a result of operating as a public company, and our management will be required to devote substantial time to new compliance requirements as a result of our Common Stock being listed on the Nasdaq Capital Market.
We will continue to incur significant increased costs as a result of operating as a public company, and our management will be required to devote substantial time to new compliance requirements of the Nasdaq Capital Market. As a public company, we will continue to incur significant legal, accounting and other expenses. We are subject to mandatory reporting requirements of the Exchange Act, which require, among other things, that we continue to file with the SEC annual, quarterly and current reports with respect to our business and financial condition, that we were not required to file as a voluntary reporting company (though we did file such reports with the SEC on a voluntary basis). We have incurred and will continue to incur costs associated with the preparation and filing of these SEC reports. Furthermore, we are subject to mandatory new corporate governance and other compliance requirements of the Nasdaq Capital Market. In addition, the Sarbanes-Oxley Act, as well as rules subsequently implemented by the SEC, the Dodd-Frank Wall Street Reform and Consumer Protection Act and the Nasdaq Capital Market or another national exchange have imposed various other requirements on public companies. Stockholder activism, the current political environment and the current high level of government intervention and regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact (in ways we cannot currently anticipate) the way we operate our business. Our management and other personnel will need to devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations have and will continue to increase our legal and financial compliance costs and will make some activities more time-consuming and costly.
In
addition, if and when we cease to be a smaller reporting company and become subject to Section 404(b) of the Sarbanes-Oxley Act, we will
be required to furnish an attestation report on internal control over financial reporting issued by our independent registered public
accounting firm. To achieve compliance with Section 404 within the prescribed time period, we will continue to be engaged in a process
to document and evaluate our internal control over financial reporting, which is both costly and challenging. In this regard, we will
need to dedicate substantially greater internal resources, potentially engage outside consultants and adopt a detailed work plan to assess
and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate
through testing that controls are functioning as documented and implement a continuous reporting and improvement process for internal
control over financial reporting. Despite our efforts, there is a risk that our independent registered public accounting firm, when required,
will not be able to conclude within the prescribed timeframe that our internal control over financial reporting is effective as required
by Section 404. This could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our
financial statements.
Future sales and issuances of our Common Stock or rights to purchase Common Stock, including pursuant to our equity incentive plan or otherwise, could result in dilution of the percentage ownership of our shareholders and could cause our stock price to fall.
We expect that we will need significant additional capital in the future to continue our planned operations. To raise capital, we may sell Common Stock, convertible securities or other equity securities in one or more transactions at prices and in a manner we determine from time to time. If we sell Common Stock, convertible securities or other equity securities in more than one transaction, including issuance of equity securities pursuant to any future stock incentive plan to our officers, directors, employees and non-employee consultants for their services to us, investors in a prior transaction may be materially diluted by subsequent sales. Additionally, any such sales may result in material dilution to our existing shareholders, and new investors could gain rights, preferences and privileges senior to those of holders of our Common Stock. Further, any future sales of our Common Stock by us or resales of our Common Stock by our existing shareholders could cause the market price of our Common Stock to decline. Any future grants of options, warrants or other securities exercisable or convertible into our Common Stock, or the exercise or conversion of such shares, and any sales of such shares in the market, could have an adverse effect on the market price of our Common Stock.
An active, liquid, and orderly market for our common stock or Purchase Warrants may not develop.
Our Common Stock trades on Nasdaq. If an active market for our Common Stock does not continue to develop or is not sustained, it may be difficult for investors to sell shares of our Common Stock without depressing the market price and investors may not be able to sell the shares of our Common Stock at all. An inactive market may also impair our ability to raise capital by selling our Common Stock and may impair our ability to acquire other businesses, applications, or technologies using our Common Stock as consideration, which, in turn, could materially adversely affect our business.
The Purchase Warrants may not have any value.
The Series A Warrant has an exercise price of $6.30 per share and the Series B Warrant has an exercise price of $10.00 per share; all of the Purchase Warrants will expire on the fifth anniversary of the date they first become exercisable. In the event our Common Stock price does not exceed the exercise price of the Purchase Warrants during the period when the warrants are exercisable, the Purchase Warrants may not have any value.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements that involve risks and uncertainties, including statements based on our current expectations, assumptions, estimates and projections about us, our industry and the regulatory environment in which we and companies integral to our ecosystem operate. The forward-looking statements are contained principally in the sections entitled “Prospectus Summary,” “Risk Factors,” “Use of Proceeds,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business.” These statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from those expressed or implied by the forward-looking statements. In some cases, these forward-looking statements can be identified by words or phrases such as “may,” “will,” “expect,” “anticipate,” “aim,” “estimate,” “intend,” “plan,” “believe,” “potential,” “continue,” “is/are likely to” or other similar expressions. The forward-looking statements included in this prospectus relate to, among others:
|
● |
risks and uncertainties associated with our research and development activities, including our clinical trials and preclinical studies; |
|
● |
the timing or likelihood of regulatory filings and approvals or of alternative regulatory pathways for our drug candidates; |
|
● |
the potential market opportunities for commercializing our drug candidates; |
|
● |
our expectations regarding the potential market size and the size of the patient populations for our drug candidates, if approved for commercial use, and our ability to serve such markets; |
|
● |
estimates of our expenses, future revenue, capital requirements and our needs for additional financing; |
|
● |
our ability to develop, acquire and advance our product candidates into, and successfully complete, clinical trials and preclinical studies and obtain regulatory approvals; |
|
● |
the implementation of our business model and strategic plans for our business and drug candidates; |
|
● |
the initiation, cost, timing, progress and results of future preclinical studies and clinical trials, and our research and development programs; |
|
● |
the terms of future licensing arrangements, and whether we can enter into such arrangements at all; |
|
● |
timing and receipt or payments of licensing and milestone revenues, if any; |
|
● |
the scope of protection we are able to establish and maintain for intellectual property rights covering our drug candidates and our ability to operate our business without infringing the intellectual property rights of others; |
|
● |
regulatory developments in the United States and foreign countries; |
|
● |
the performance of our third party suppliers and manufacturers; |
|
● |
our ability to maintain and establish collaborations or obtain additional funding; |
|
● |
the success of competing therapies that are currently or may become available; |
|
● |
our ability to continue as a going concern; |
|
● |
the effect of the ongoing COVID-19 pandemic; |
|
● |
our financial performance; and |
|
● |
developments and projections relating to our competitors and our industry. |
We caution you that the forward-looking statements highlighted above do not encompass all of the forward-looking statements made in this prospectus or in the documents incorporated by reference in this prospectus.
There are important factors that could cause actual results to vary
materially from those described in this registration statement as anticipated, estimated or expected, including, but not limited to: the
effects of the COVID-19 outbreak, including on the demand for our products; the duration of the COVID-19 outbreak and severity of
such outbreak in regions where we operate; the pace of recovery following the COVID-19 outbreak; our ability to implement cost
containment and business recovery strategies; the adverse effects of the COVID-19 outbreak on our business or the market price of
our ordinary shares; competition in the industry in which we operate and the impact of such competition on pricing, revenues and margins,
volatility in the securities market due to the general economic downturn; SEC regulations which affect trading in the securities of “penny
stocks,” and other risks and uncertainties described herein and the risk factors set forth in Part I - Item 1A, “Risk Factors”,
in our Annual Report on Form 10-K for the year ended December 31, 2023, as filed with the SEC on March 13, 2024 and elsewhere in the documents
incorporated by reference into this prospectus. Moreover, we operate in a very competitive and challenging environment. New risks and
uncertainties emerge from time to time, and it is not possible for us to predict all risks and uncertainties that could have an impact
on the forward-looking statements contained in this prospectus and in the documents incorporated by reference in this prospectus. We cannot
assure you that the results, events and circumstances reflected in the forward-looking statements will be achieved or occur, and actual
results, events or circumstances could differ materially from those described in the forward-looking statements. Except as required by
law, we assume no obligation to update any forward-looking statements publicly, or to update the reasons actual results could differ materially
from those anticipated in any forward- looking statements, even if new information becomes available in the future. Depending on the market
for our stock and other conditional tests, a specific safe harbor under the Private Securities Litigation Reform Act of 1995 may be available.
Notwithstanding the above, Section 27A of the Securities Act of 1933, as amended (the “Securities Act”) and Section 21E of
the Securities Exchange Act of 1934, as amended (the “Exchange Act”) expressly state that the safe harbor for forward-looking
statements does not apply to companies that issue penny stock. Because we may from time to time be considered to be an issuer of penny
stock, the safe harbor for forward-looking statements may not apply to us at certain times.
The forward-looking statements contained in this prospectus and in the documents incorporated by reference in this prospectus relate only to events as of the date on which the statements are made. We do not undertake any obligation to release publicly any revisions to such forward-looking statements to reflect events or circumstances after the date of this prospectus or to reflect the occurrence of unanticipated events. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements and you should not place undue reliance on our forward-looking statements. Our forward-looking statements do not reflect the potential impact of any future acquisitions, mergers, dispositions, joint ventures, other strategic transactions or investments we may make.
USE OF PROCEEDS
We are not selling any shares of our common stock
in this offering and, as a result, we will not receive any proceeds from the sale of the common stock covered by this prospectus. All
of the net proceeds from the sale of our common stock will go to the holders of the warrants issued in the August 2021 public offering.
Upon exercise of the warrants, however, we will receive proceeds from the exercise of such warrants if exercised for cash and not on a
cashless basis. We will receive approximately $6,875,000 of proceeds if all the currently outstanding warrants issued in the August 2021
public offering are exercised for cash. We currently intend to use these proceeds for general corporate purposes.
DIVIDEND POLICY
We have never declared or paid any cash dividends on our capital stock, and we do not currently intend to pay any cash dividends on our capital stock in the foreseeable future. We currently intend to retain all available funds and any future earnings to support operations and to finance the growth and development of our business. Any future determination to pay dividends will be made at the discretion of our Board, subject to applicable laws and will depend upon, among other factors, our results of operations, financial condition, contractual restrictions and capital requirements. Our future ability to pay cash dividends on our capital stock may also be limited by the terms of any debt instruments or preferred securities issued in the future.
DILUTION
If you invest in our securities, your interest may be immediately and substantially diluted to the extent of the difference between the price you pay per share and the pro forma net tangible book value per share of our common stock.
Our historical net tangible book value as
December 31, 2023 was $8,130,972, or $1.02 per share.
Our pro forma net tangible book value as of
December 31, 2023 was $16,993,350, or $1 per share and includes the following transactions subsequent to December 31, 2023: (i) issuance
of a secured, convertible note in the principal amount of $3,704,167 that is convertible into shares of common stock at an initial conversion
price of $1.05 per share, subject to adjustment, and (ii) issuance of a warrant to purchase up to 5,291,667 shares of common stock at
an initial exercise price of $1.05 per share, subject to adjustment.
Assuming the conversion of convertible notes
into common shares, as well as exercise of warrants issued, which means the issuance of 9,023,844 shares of common stock upon the exercise
of such warrants, our pro forma as adjusted net tangible book value would have been approximately $8,862,378, or $1 per share at December
31, 2023.
Dilution in pro forma as adjusted net tangible book value per share represents the difference between the amount per share of common stock and the pro forma net tangible book value per share of our common stock immediately after this offering.
The following table illustrates the per share dilution to investors purchasing shares in an offering using this prospectus:
| Assumed
price per share |
|
|
|
|
|
$ |
6.25 |
|
| Pro
forma net tangible book value per share as of December 31, 2023 |
|
$ |
1.02 |
|
|
|
|
|
| Increase
in pro forma net tangible book value per share |
|
$ |
-0.2 |
|
|
|
|
|
| Pro
forma as adjusted net tangible book value per share after this offering |
|
|
|
|
|
$ |
1 |
|
| Dilution
per share to new investors |
|
|
|
|
|
$ |
5.25 |
|
The number of shares of common stock that
will be outstanding immediately after this offering is based on 16,993,350 shares of common stock outstanding as of December 31,
2023.
MARKET FOR OUR COMMON STOCK, DIVIDENDS AND
RELATED SHAREHOLDER INFORMATION
Market Information. As of August 3,
2021, our common stock, par value $.001 per share (the “Common Stock”), is quoted on the Nasdaq Capital Markets under the
symbol “ABVC”.
Holders. As of April 19, 2024, we had approximately 656
shareholders of record of our common stock.
Dividends. Holders of our common stock
are entitled to receive such dividends as may be declared by our board of directors. No dividends on our common stock have ever been paid,
and we do not anticipate that dividends will be paid on our common stock in the foreseeable future.
Securities authorized for issuance under equity incentive plans.
Equity Compensation Plan Information
The following
table discloses information as of December 31, 2023, with respect to compensation plans (including individual compensation arrangements)
under which our equity securities are authorized for issuance, aggregated as follows:
| Plan category | |
Number of
securities
to
be issued
upon
exercise of
outstanding
options,
warrants
and rights | | |
Weighted-
average
exercise
price of
outstanding
options,
warrants
and rights | | |
Shares of
common
stock
remaining
available for
future
issuance
under equity
compensation
plans | |
| Equity compensation plans approved by security holders | |
| 2,587,104 | | |
$ | 2.79 | | |
| 3,860,211 | |
| Equity compensation plans not approved by security holders | |
| - | | |
| - | | |
| - | |
| Total | |
| 2,587,104 | | |
$ | 2.79 | | |
| 3,860,211 | |
MANAGEMENT’S DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis should
be read together with our financial statements and the related notes appearing elsewhere in this prospectus. This discussion contains
forward-looking statements reflecting our current expectations that involve risks and uncertainties. See “Cautionary Note Regarding
Forward-Looking Statements and Industry Data” for a discussion of the uncertainties, risks and assumptions associated with these
statements. Actual results and the timing of events could differ materially from those discussed in our forward-looking statements as
a result of many factors, including those set forth under “Risk Factors” and elsewhere in this prospectus.
Overview
From its inception, the Company has
not generated substantial revenue from its medical device and new drug development. For the year ended December 31, 2023, the Company
generated $152,430 in revenue, mainly from the sale of Contract Development & Manufacturing Organization (“CDMO”) services.
Business Overview
ABVC BioPharma Inc., which was incorporated
under the laws of the State of Nevada on February 6, 2002, is a clinical stage biopharmaceutical company focused on development of new
drugs and medical devices, all of which are derived from plants.
Medicines derived from plants have a long
history of relieving or preventing many diseases and, typically, have exhibited fewer side effects than drugs developed from animals
or chemical ingredients. Perhaps the most famous example is aspirin, which evolved from a compound found in the bark and leaves of the
willow tree and was later marketed by Bayer starting in 1899. Aspirin has very few serious side effects and has proven to be one of the
most successful drugs in medical history. Some 50 years later, scientists identified anticancer compounds in the rosy periwinkle, which
Eli Lilly subsequently produced for the treatment of leukemia and Hodgkins disease. Other well-known examples of successful botanical
drugs include the cancer-fighting Taxol, isolated from the Pacific yew tree.
The Company develops its pipeline by carefully
tracking new medical discoveries or medical device technologies in research institutions in the Asia-Pacific region. Pre-clinical, disease
animal model and Phase I safety studies are examined closely by the Company’s scientists and other specialists known to the Company
to identify drugs that it believes demonstrate efficacy and safety based on the Company’s internal qualifications. Once a drug
is shown to be a good candidate for further development and ultimately commercialization, BriVision licenses the drug or medical device
from the original researchers and begins to introduce the drugs clinical plan to highly respected principal investigators in the United
States, Australia and Taiwan. In almost all cases, we have found that research institutions in each of those countries are eager to work
with the Company to move forward with Phase II clinical trials.
Currently, institutions conducting phase II clinical trials in
partnership with ABVC include:
| ● | Medical
Device: ABV-1701, Vitargus® in vitrectomy surgery, Phase II Study in Australia
and Thailand, Principal Investigator: Professor/Dr. Matthew Simunovic, Sydney Eye Hospital;
Dr. Elvis Ojaimi, East Melbourne Eye Group & East Melbourne Retina, Duangnate Rojanaporn,
M.D., Ramathibodi Hospital; Thuss Sanguansak, M.D., Srinagarind Hospital. |
| ● | Drug:
ABV-1505, Adult Attention-Deficit Hyperactivity Disorder (ADHD), Phase II, NCE drug Principal
Investigators: Keith McBurnett, Ph.D. and Linda Pfiffner, Ph.D., University of California
San Francisco (UCSF), School of Medicine |
| ● | Drug:
ABV-1601, Major Depression in Cancer Patients, Phase I/II, NCE drug Principal Investigator:
Scott Irwin, MD, Ph.D. - Cedars Sinai Medical Center (CSMC) |
| ● | Drug:
ABV-1519, A Phase I/II, Open Label Study to Evaluate the Safety and Efficacy of BLEX 404
Oral Liquid Combined with Pemetrexed + Carboplatin Therapy in Patients with Advanced Inoperable
or Metastatic EGFR wild-type Non-Small Cell Lung Cancer Patients |
Upon successful completion of the Phase II
trial, the Company will seek a partner - a large pharmaceutical company - to complete a Phase III study, submit the New Drug Application
(NDA), and commercialize the drug upon approval by the FDA and Taiwan FDAs.
Another part of the Company’s business
is conducted by BioKey, a wholly owned subsidiary, that is engaged in a wide range of services, including, API characterization, pre-formulation
studies, formulation development, analytical method development, stability studies, IND/NDA/ANDA/510K submissions, and manufacturing
clinical trial materials (phase I through phase III) and commercial manufacturing.
On June 21, 2023, Dr. Howard Doong resigned
from his position as the Company’s CEO. The Company’s board of directors appointed Dr. Uttam Patil to replace Dr. Doong as
the Company’s CEO.
On August 14, 2023, the Company entered into
a cooperation agreement with Zhonghui United Technology (Chengdu) Group Co., Ltd., pursuant to which the Company acquired a 20% ownership
of certain property and a parcel of the land (collectively, the “Property”) owned by Zhonghui in exchange for an aggregate
of 370,000 shares of Common Stock at $20 per share (the “Zhonghui Shares”). Accordingly, stockholders’ equity increased
by $7.4M.
The Company and Zhonghui plan to jointly develop
the Property into a healthcare center for senior living, long-term care, and medical care in the areas of ABVCs’ special interests,
such as Ophthalmology, Oncology, and Central Nervous Systems. The plan is to establish a base for the China market and global development
of these interests. The asset ownership certification is in the application process and pending approval from the Chinese government.
During the third quarter of 2023, the Company
issued the Zhonghui Shares. The Zhonghui Shares are subject to a lock-up period of one year following the closing date of this Transaction.
In addition, the parties agreed that, after one year following the closing of the transaction, if the market value of the shares issued
or the value of the Property increase or decrease, the parties will negotiate in good faith to make reasonable adjustments thereto; provided,
however that in no event shall Zhonghui’s ownership exceed 19.99% of the Company.
On July 31, 2023, the Company entered into
a binding term sheet with Xinnovation Therapeutics Co., Ltd., a Company incorporated under the Law of People’s Republic of China.
The term sheet contemplates that, pursuant to definitive agreements, Xinnovation will be granted an exclusive license to develop, manufacture,
market, and distribute ABV-1504 for Major Depressive Disorder (MDD) and ABV-1505 for Attention-Deficit/Hyperactivity Disorder, in the
Chinese market and shall bear the costs for clinical trials and product registration in China and the Company would receive an initial
license fee and royalty payments ranging from 5% to 12% based on the projected annual net sales of the licensed drugs by Xinnovation
in China. This transaction remains subject to the negotiation of definitive documents and therefore there is no guarantee that this transaction
will occur.
In November 2023, the Company and one of its
subsidiaries, BioLite, Inc. (“BioLite”) each entered into a multi-year, global licensing agreement with AIBL for the Company
and BioLite’s CNS drugs with the indications of MDD (Major Depressive Disorder) and ADHD (Attention Deficit Hyperactivity Disorder)
(the “Licensed Products”). The potential license will cover the Licensed Products’ clinical trial, registration, manufacturing,
supply, and distribution rights. The Licensed Products for MDD and ADHD, owned by ABVC and BioLite, were valued at $667M by a third-party
evaluation. The parties are determined to collaborate on the global development of the Licensed Products. The parties are also working
to strengthen new drug development and business collaboration, including technology, interoperability, and standards development. As
per each of the respective agreements, each of ABVC and BioLite received 23 million shares of AIBL stock at $10 per share, and if certain
milestones are met, each of ABVC and BioLite may receive $3,500,000 and royalties equaling 5% of net sales, up to $100 million. Upon
the issuance of the shares, AIBL became a subsidiary of ABVC.
On February 6, 2024, the Company entered into
a definitive agreement with Shuling Jiang (“Shuling”), pursuant to which Shuling shall transfer the ownership of certain
land she owns located at Taoyuan City, Taiwan (the “Land”) to the Company (the “Agreement”). Shuling
is a director of the Company, is married to TS Jiang, the Company’s Chief Strategic Officer and owns approximately 15.4% of the
Company’s issued and outstanding shares of common stock.
In consideration for the Land, the Company
shall pay Shuling (i) 703,495 restricted shares of the Company’s common stock (the “Shares”) at a price of $3.50
per share and (ii) five-year warrants to purchase up to 1,000,000 shares of the Company’s common stock, with an exercise price
of $2.00 per share. Under the Agreement, Shuling will also transfer outstanding liability owed on the Land (approximately $500,000) to
the Company. Thus, the parties value the exchange at approximately $2,962,232.
Use of acquired land
ABVC acquired the real estate described above
for the long-term purpose of supporting its pipeline of products and reducing costs. As per FDA guidelines, the raw material of botanical
drugs must be grown in a specific area under Good Agricultural Practices (GAP) or in an environmentally fully controlled plant factory
to maintain quality. By acquiring land, ABVC plans to grow its botanical drug raw materials under its control; doing this will help the
Company maintain the quality of the product and lower the cost of raw materials, which in turn will lower the cost of the drug substance
and the drug product when it’s botanical drugs become commercialized.
Common Stock Reverse Split
On July 25, 2023, the Company filed a Certificate
of Amendment to its Articles of Incorporation authorizing a 1-for-10 reverse stock split of the issued and outstanding shares of its
common stock. The Company’s stockholders previously approved the Reverse Stock Split at the Company’s Special Shareholder
Meeting held on July 7, 2023. The Reverse Stock Split was effected to reduce the number of issued and outstanding shares and to increase
the per share trading value of the Company’s common stock, although that outcome is not guaranteed. Unless otherwise noted, all
shares and related financial information in this Form 10-K reflect this 1-for-10 reverse stock split.
NASDAQ Listing
In August 2022, we received a deficiency letter
from the Nasdaq Listing Qualifications Department (the “Staff”) notifying us that, for the last 30 consecutive business days,
the closing bid price for our common stock was below the minimum $1.00 per share required for continued listing on The Nasdaq Capital
Market pursuant to Nasdaq Listing Rule 5550(a)(2) (“Rule 5550(a)(2)”). In accordance with Nasdaq Listing Rule 5810(c)(3)(A),
we were initially given until February 14, 2023 to regain compliance with Rule 5550(a)(2). Since the Company did not regain compliance
by such date, it requested and received an additional 180 days, until August 14, 2023, to comply with Rule 5550(a)(2).
The deficiency has no immediate effect on
the listing of the Company’s common stock, and its common stock continues to trade on The Nasdaq Capital Market under the symbol
“ABVC” at this time.
If at any time before August 14, 2023, the
bid price of the Company’s common stock closes at $1.00 per share or more for a minimum of 10 consecutive business days, the Staff
will provide written confirmation that the Company has achieved compliance and the matter will be closed.
If the Company does not regain compliance
with Rule 5550(a)(2) by August 14, 2023, the Staff will provide written notification that the Company’s securities will be delisted,
although the Company maintains the right to appeal such determination. The Company intends to actively monitor the closing bid price
for its common stock and will consider available options to resolve the deficiency and regain compliance with Rule 5550(a)(2).
On August 8, 2023, the Company received a
notification letter from Nasdaq notifying the Company that the Staff has determined that for 10 consecutive business days, from July
25, 2023 to August 7, 2023, the closing bid price of the Company’s common stock has been at least $1.00 per share or greater. Accordingly,
the Staff determined that the Company regained compliance with Listing Rule 5550(a)(2) and indicated that the matter is now closed.
On May 24, 2023, we received a deficiency
letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq Stock Market LLC (“Nasdaq”)
notifying the Company that it is not currently in compliance with the minimum stockholders’ equity requirement, or the alternatives
of market value of listed securities or net income from continuing operations, for continued listing on the Nasdaq Capital Market. Nasdaq
Listing Rule 5550(b)(1) requires listed companies to maintain stockholders’ equity of at least $2,500,000, and the Company’s
stockholders’ equity was $1,734,507 as of March 31, 2023. In accordance with Nasdaq rules, the Company had 45 calendar days, or
until July 10, 2023, to submit a plan to regain compliance. After submitting a plan to regain compliance, on July 10, 2023, Nasdaq granted
the Company an extension until August 30, 20203, to comply with Listing Rule 5550(b)(1). On July 31, 2023, the Company issued 300,000
shares of Common Stock and 200,000 pre-funded warrants, at an exercise price of $0.01 per share, in a registered direct offering. Pursuant
to this transaction, the stockholders’ equity was increased by $1.75M. On August 1, 2023, $500,000 of Notes were converted at $3.50
per share and the holder received 142,857 shares of Common Stock. As a result of this conversion, the stockholders’ equity was
increased by $0.5M. Additionally, on August 14, 2023, the Company entered into a cooperation agreement with Zhonghui United Technology
(Chengdu) Group Co., Ltd., pursuant to which the Company acquired a 20% ownership of certain property and a parcel of the land owned
by Zhonghui in exchange for an aggregate of 370,000 shares of Common Stock. Accordingly, stockholders’ equity increased by $7.4M.
On February 23, 2023, the Company entered into a securities purchase agreement with Lind, pursuant to which the Company issued Lind a
secured, convertible note in the principal amount of $3,704,167 (the “Lind Offering”), for a purchase price of $3,175,000
(the “Lind Note”), that is convertible into shares of Common Stock at an initial conversion price of $1.05 per share, subject
to adjustment. On August 24, 2023, the Company started repaying Lind the monthly installments due under the Lind Notes; $308,000 was
repaid via the issuance of 176,678 shares of Common Stock (the “Monthly Shares”) at the Redemption Share Price (as defined
in the Lind Note) of $1.698 per share. Pursuant to the terms of the Lind Note, Lind increased the amount of the next monthly payment
to one million dollars, such that as of September and together with the Monthly Shares, the Company repaid Lind a total of $1M by September
2023. As a result, the stockholders’ equity increased by an additional $1M. As a result of the four transactions referenced above,
the Company’ estimated that its stockholders’ equity would increase by approximately $10.65M. On September 6, 2023, Nasdaq
issued a letter that the Company is in compliance with Rule 5550(b)(1), but noted that if at the time of the Company’s next periodic
report the Company does not evidence compliance, it may be subject to delisting.
Recent Research Results
Vitargus® Phase II Study has been initiated
in Australia and Thailand, Principal Investigator: Duangnate Rojanaporn, M.D., Ramathibodi Hospital; Thuss Sanguansak, M.D., Srinagarind
Hospital of the two Thailand sites and Professor/Dr. Matthew Simunovic, Sydney Eye Hospital; Dr. Elvis Ojaimi, East Melbourne Eye Group
& East Melbourne Retina of the two Australian sites. The Phase II study has started in the 2nd quarter of 2023. The company
is working on improvements to the Vitargus product through the new batch of investigational product.
Initially the Company will focus on ABV-2002,
a solution utilized to store a donor cornea prior to either penetrating keratoplasty (full thickness cornea transplant) or endothelial
keratoplasty (back layer cornea transplant). Designated ABV-2002 under the Company’s product identification system, the solution
is comprised of a specific poly amino acid that protects ocular tissue from damage caused by external osmolarity exposure during pre-surgery
storage. The specific polymer in ABV-2002 can adjust osmolarity to maintain a range of 330 to 390 mOsM thereby permitting hydration within
the corneal stroma during the storage period. Stromal hydration results in (a) maintaining acceptable corneal transparency and (b) prevents
donor cornea swelling. ABV-2002 also contains an abundant phenolic phytochemical found in plant cell walls that provides antioxidant
antibacterial properties and neuroprotection.
Early testing by BioFirst indicates that ABV-2002
may be more effective for protecting the cornea and retina during long-term storage than other storage media available today and can
be manufactured at lower cost. Further clinical development task was put on hold due to the lack of funding.
In addition, BioFirst was incorporated on
November 7, 2006, focusing on the R&D, manufacturing, and sales of innovative patented pharmaceutical products. The technology of
BioFirst comes from the global exclusive licensing agreements BioFirst maintains with domestic R & D institutions. Currently, BioFirst’s
main research and development product is the vitreous substitute (Vitargus®), licensed by the National Health Research Institutes.
Vitargus is the world’s first bio-degradable vitreous substitute and offers a number of advantages over current vitreous substitutes
by minimizing medical complications and reducing the need for additional surgeries.
Vitargus has started the construction of a
GMP factory in Hsinchu Biomedical Science Park, Taiwan, with the aim at building a production base to supply the global market, and promote
the construction of bio-degradable vitreous substitute manufacturing centers in Taiwan. Completion of this factory would allow ABVC to
manufacture Vitargus with world-class technology in a GMP certified pharmaceutical factory. BioFirst is targeting to complete the construction
in 2025.
On July 12, 2022, the Company announced the
enrollment progress in the Phase II Part II clinical study of the company’s ADHD medicine (ABV-1505). Since the first-treated subject
reported on May 10, 2022, a total of sixty-nine (69) subjects have been enrolled in the study, including 50 who have completed the 56-day
treatment. The study, a randomized, double-blind, placebo-controlled study entitled “A Phase II Tolerability and Efficacy Study
of PDC-1421 Treatment in Adult Patients with Attention-Deficit Hyperactivity Disorder (ADHD), Part II, is expected to eventually involve
approximately 100 patients. Five prestigious research hospitals in Taiwan and the research hospital at the University of California,
San Francisco (UCSF) are participating in the study which is a continuation of the Phase II part 1 study of ABV-1505 completed successfully
at UCSF and accepted by the U.S. Food & Drug Administration in October of 2020. The UCSF Medical Center Institutional Review Board
has approved participation in the Part II study, and the site initiation visit was conducted in March 2023.
Public Offering & Financings
2024 Financings
On January 17, 2024, the Company entered into
a securities purchase agreement with Lind Global Fund II, LP (“Lind”), pursuant to which the Company issued Lind a secured,
convertible note in the principal amount of $1,000,000, for a purchase price of $833,333 (the “3rd Lind Note”),
that is convertible into shares of the Company’s common stock at a conversion price, which shall be the lesser of (i) $3.50 (the
“Fixed Price”) and (ii) 90% of the average of the three lowest VWAPs (as defined in the 3rd Lind Note) during
the 20 trading days prior to conversion (“Variable Price”), subject to adjustment (the “Note Shares”). Notwithstanding
the foregoing, provided that no Event of Default (as defined in the 3rd Lind Note) shall have occurred, conversions under
the 3rd Lind Note shall be at the Fixed Price for the first 180 days following the closing date. Lind will also receive a
5-year, common stock purchase warrant (the “3rd Lind Warrant”) to purchase up to 1,000,000 shares of the Company’s
common stock at an initial exercise price of $2.00 per share, subject to adjustment (each, a “Warrant Share,” together with
the 3rd Lind Note, Note Shares and 3rd Lind Warrant, the “Securities”). The parties later agreed to
a floor price of $1.00 for the Variable Price and that the Company would compensate Lind in cash if the Variable Price was less than
such floor price at the time of conversion.
Upon the occurrence of any Event of Default
(as defined in the 3rd Lind Note), the Company must pay Lind an amount equal to 120% of the then outstanding principal amount
of the 3rd Lind Note, in addition to any other remedies under the 3rd Lind Note or the other Transaction Documents
(as defined below).
The 3rd Lind Warrant may be exercised
via cashless exercise in the event a registration statement covering the Warrant Shares is not available for the resale of such Warrant
Shares or upon exercise of the 3rd Lind Warrant in connection with a Fundamental Transaction (as defined in the 3rd
Lind Warrant).
Pursuant to the terms of the securities purchase
agreement, if at any time prior to a date that is 18 months following the closing of the offering, the Company proposes to offer or sell
any additional securities in a subsequent financing, the Company shall first offer Lind the opportunity to purchase up to 10% of such
new securities.
In connection with the Offering, the Company
and its subsidiaries: (i) Biokey, Inc., a California corporation (“BioKey”), (ii) Biolite Holding, Inc., a Nevada corporation
(“BioLite”), (iii) Biolite BVI, Inc., a British Virgin Islands corporation (“BioLite BVI”) and (iv) American
BriVision Corporation, a Delaware corporation (“American BriVision” and, collectively with the Company, BioKey, BioLite,
and BioLite BVI, the “Guarantors”), jointly and severally guaranteed all of the obligations of the Company in connection
with the offering (the “Guaranty”) with certain collateral, as set forth in the related Transaction Documents (as hereinafter
defined).
The sale of the 3rd Lind Note and the terms of the offering, including the Guaranty are set forth in the securities
purchase agreement, the 3rd Lind Note, the 3rd Lind Warrant, the Second Amendment to Guaranty, the Second Amendment
to Security Agreement, and the Second Amendment to Guarantor Security Agreement (collectively, the “Transaction Documents”).
Allele Capital Partners, LLC (“Allele”)
together with its executing broker dealer, Wilmington Capital Securities, LLC (together with its affiliates, “Wilmington”),
served as the exclusive placement agent (the “Placement Agent”) of the offering. the Company has agreed to pay certain expenses
of the placement agent in connection with the offering and issued them a warrant to purchase up to 25,000 shares of common stock, on
the same terms as set forth in the 3rd Lind Warrant.
The securities purchase agreement also contains
customary representation and warranties of the Company and the Investors, indemnification obligations of the Company, termination provisions,
and other obligations and rights of the parties.
The foregoing description of the Transaction
Documents is qualified by reference to the full text of the forms of the Transaction Documents, which are filed as Exhibits hereto and
incorporated herein by reference.
2023 Financings
On November 17, 2023, the Company entered
into a securities purchase agreement (the “2nd Lind Securities Purchase Agreement”) with Lind Global Fund II,
LP (“Lind”), pursuant to which the Company issued Lind a secured, convertible note in the principal amount of $1,200,000
(the “2nd Lind Offering”), for a purchase price of $1,000,000 (the “2nd Lind Note”), that
is convertible into shares of the Company’s common stock at a conversion price, which shall be the lesser of (i) $3.50 (the “Fixed
Price”) and (ii) 90% of the average of the three lowest VWAPs (as defined in the 2nd Lind Note) during the 20 trading
days prior to conversion, subject to adjustment. Notwithstanding the foregoing, provided that no Event of Default (as defined in the
2nd Lind Note) shall have occurred, conversions under the 2nd Lind Note shall be at the Fixed Price for the first
180 days following the closing date. Lind will also receive a 5-year, common stock purchase warrant (the “2nd Lind Warrant”)
to purchase up to 1,000,000 shares of the Company’s common stock at an initial exercise price of $2 per share, subject to adjustment.
The parties later agreed to a floor price of $1.00 for the Variable Price and that the Company would compensate Lind in cash if the variable
price was less than such floor price at the time of conversion.
Upon the occurrence of any Event of Default
(as defined in the 2nd Lind Note), the Company must pay Lind an amount equal to 120% of the then outstanding principal amount
of the 2nd Lind Note, in addition to any other remedies under the 2nd Lind Note or the other Transaction Documents
(as defined below).
Pursuant to the terms of the 2nd
Lind Securities Purchase Agreement, if at any time prior to a date that is 18 months following the closing of the 2nd Lind
Offering, the Company proposes to offer or sell any additional securities in a subsequent financing, the Company shall first offer Lind
the opportunity to purchase up to 10% of such new securities.
In connection with the 2nd Lind
Offering, the Company and its subsidiaries: (i) Biokey, Inc., a California corporation (“BioKey”), (ii) Biolite Holding,
Inc., a Nevada corporation (“BioLite”), (iii) Biolite BVI, Inc., a British Virgin Islands corporation (“BioLite BVI”)
and (iv) American BriVision Corporation, a Delaware corporation (“American BriVision” and, collectively with the Company,
BioKey, BioLite, and BioLite BVI, the “Guarantors”), jointly and severally guaranteed all of the obligations of the Company
in connection with the 2nd Lind Offering (the “Guaranty”) with certain collateral, as set forth in the related
Transaction Documents (as hereinafter defined).
The sale of the Note and the terms of the
2nd Lind Offering, including the Guaranty are set forth in the 2nd Lind Securities Purchase Agreement, the 2nd
Lind Note, the 2nd Lind Warrant, the First Amendment to Guaranty, the First Amendment to Security Agreement, and the
First Amendment to Guarantor Security Agreement (collectively, the “Transaction Documents”).
Allele Capital Partners, LLC (“Allele”)
together with its executing broker dealer, Wilmington Capital Securities, LLC (together with its affiliates, “Wilmington”),
served as the exclusive placement agent (the “Placement Agent”) of the 2nd Lind Offering. We have agreed to pay
certain expenses of the placement agent in connection with the 2nd Lind Offering.
An amendment was filed on February 29, 2024
to disclose that due to Nasdaq requirements, the parties entered into an amendment to the Note, pursuant to which the conversion price
shall have a floor price of $1.00 (the “Amendment”). Additionally, the Amendment requires the Company to make a cash payment
to Lind if in connection with a conversion, the conversion price is deemed to be the floor price.
The Securities Purchase Agreement also contains
customary representation and warranties of the Company and the Investors, indemnification obligations of the Company, termination provisions,
and other obligations and rights of the parties.
The foregoing description of the Transaction
Documents is qualified by reference to the full text of the forms of the Transaction Documents, which are filed as Exhibits hereto and
incorporated herein by reference.
On February 23, 2023, the Company entered
into a securities purchase agreement (the “Lind Securities Purchase Agreement”) with Lind Global Fund II, LP (“Lind”),
pursuant to which the Company issued Lind a secured, convertible note in the principal amount of $3,704,167 (the “Lind Offering”),
for a purchase price of $3,175,000 (the “Lind Note”), that is convertible into shares of the Company’s common stock
at an initial conversion price of $1.05 per share, subject to adjustment (the “Note Shares”). The Company also issued Lind
a common stock purchase warrant (the “Lind Warrant”) to purchase up to 5,291,667 shares of the Company’s common stock
at an initial exercise price of $1.05 per share, subject to adjustment (each, a “Warrant Share,” together with the Note,
Note Shares and Warrants, the “Lind Securities”).
The Lind Note does not carry any Interest.
Beginning with the date that is six months from the issuance date of the Lind Note and on each one (1) month anniversary thereafter,
the Company shall pay Lind an amount equal to $308,650.58, until the outstanding principal amount of the Lind Note has been paid in full
prior to or on the Maturity Date or, if earlier, upon acceleration, conversion or redemption of the Lind Note in accordance with the
terms thereof (the “Monthly Payments”). At the Company’s discretion, the Monthly Payments shall be made in (i) cash,
(ii) shares of the Company’s common stock, or (iii) a combination of cash and Shares; if made in shares, the number of shares shall
be determined by dividing (x) the principal amount being paid in shares by (y) 90% of the average of the 5 lowest daily VWAPs during
the 20 trading days prior to the applicable payment date. The Lind Notes sets forth certain conditions that must be satisfied before
the Company may make any Monthly Payments in shares of common stock. If the Company makes a Monthly Payment in cash, the Company must
also pay Lind a cash premium of 5% of such Monthly Payment.
Upon the occurrence of any Event of Default
(as defined in the Lind Note), the Company must pay Lind an amount equal to 120% of the then outstanding principal amount of the Lind
Note, in addition to any other remedies under the Note or the other Transaction Documents.
The Lind Warrant may be exercised via cashless
exercise.
Pursuant to the terms of the Lind Securities
Purchase Agreement, if at any time prior to a date that is 18 months following the closing of the Lind Offering, the Company proposes
to offer or sell any additional securities in a subsequent financing, the Company shall first offer Lind the opportunity to purchase
up to 10% of such new securities.
In connection with the Lind Offering, the
Company and its subsidiaries: (i) Biokey, Inc., a California corporation (“BioKey”), (ii) Biolite Holding, Inc., a Nevada
corporation (“BioLite”), (iii) Biolite BVI, Inc., a British Virgin Islands corporation (“BioLite BVI”) and (iv)
American BriVision Corporation, a Delaware corporation (“American BriVision” and, collectively with the Company, BioKey,
BioLite, and BioLite BVI, the “Guarantors”), jointly and severally guaranteed all of the obligations of the Company in connection
with the Lind Offering (the “Guaranty”) with certain collateral, as set forth in the related Transaction Documents (as hereinafter
defined).
The sale of the Lind Note and the terms of
the Lind Offering, including the Guaranty are set forth in the Lind Securities Purchase Agreement, the Note, the Warrant, a Security
Agreement, Guarantor Security, Guaranty, a Trademark Security Agreement with Rgene Corporation, a Trademark Security Agreement with BioFirst,
a Patent Security Agreement, a Copyright Security Agreement and a Stock Pledge Agreement (collectively, the “Transaction Documents”).
Allele Capital Partners, LLC (“Allele”)
together with its executing broker dealer, Wilmington Capital Securities, LLC (together with its affiliates, “Wilmington”),
served as the exclusive placement agent (the “Placement Agent”) of the Lind Offering. As a result of the Lind Offering, the
Company will pay the Placement Agent (i) a cash fee of 6% of the gross proceeds from the sale of the Securities, and (ii) common stock
purchase warrants to purchase 6% of the number of shares of common stock issuable under the Lind Note. We also agreed to pay certain
expenses of the placement agent in connection with the Lind Offering.
Pursuant to the Lind Securities Purchase Agreement,
the Company agreed to register all of the Lind Securities and the shares of common stock underlying the warrant issued to the placement
agent.
The Securities Purchase Agreement also contains
customary representation and warranties of the Company and the Investors, indemnification obligations of the Company, termination provisions,
and other obligations and rights of the parties.
Upon the occurrence of any Event of Default
(as defined in the Lind Note), the Company must pay Lind an amount equal to 120% of the then outstanding principal amount of the Lind
Note (the “Mandatory Default Amount”), in addition to any other remedies under the Note or the other Transaction Documents.
The Company and Lind entered into a letter agreement on September 12, 2023, pursuant to which the Mandatory Default Amount was reduced
to 115% of the then outstanding principal amount of the Lind Note; pursuant to the letter agreement, Lind also agreed to waive any default
associated with the Company’s market capitalization being below $12.5 million for 10 consecutive days through February 23, 2024,
but retained its right to convert its Note. In addition, if the Company is unable to increase its market capitalization and is unable
to obtain a further waiver or amendment to the Lind Note, then the Company could experience an event of default under the Lind Note,
which could have a material adverse effect on the Company’s liquidity, financial condition, and results of operations. The Company
cannot make any assurances regarding the likelihood, certainty, or exact timing of the Company’s ability to increase its market
capitalization, as such metric is not within the immediate control of the Company and depends on a variety of factors outside the Company’s
control.
The foregoing description of the Transaction
Documents is qualified by reference to the full text of the forms of the Transaction Documents, which are filed as Exhibits hereto and
incorporated herein by reference.
2022 Financing
On May 11, 2022, the Company entered into
certain securities purchase agreement (the “May SPA”) with certain investors (the “Purchasers”). Pursuant to
the May SPA, the Company agreed to issue 2,000,000 shares of its Common Stock, at a price of $2.11 per share and 5-year warrants to purchase
up to 2,000,000 shares of Common Stock, exercisable at a price of $2.45 per share (the “May Warrants”) to the Purchasers.
The gross proceeds before deducting any estimated offering expenses are $4,220,000. The transaction contemplated by the May SPA was closed
on May 16, 2022.
The Company paid to the co-placement agents
an aggregate cash fee equal to 8% of the aggregate sales price of the securities sold and issued them warrants to purchase up to 160,000
shares of Common Stock, on the same terms as the May Warrants.
Strategy
Key elements of our business strategy include:
| ● | Advancing
to the pivotal trial phase of ABV-1701 Vitargus® for the treatments of Retinal Detachment
or Vitreous Hemorrhage, which we expect to generate revenues in the future. |
| ● | Focusing
on licensing ABV-1504 for the treatment of major depressive disorder, MDD, after the successful
completion of its Phase II clinical trials. |
| ● | Completing
Phase II, Part 2 clinical trial for ABV-1505 for the treatment of attention deficit hyperactivity
disorder, ADHD. |
| ● | Out
licensing drug candidates and medical device candidates to major pharmaceutical companies
for phase III and pivotal clinical trials, as applicable, and further marketing if approved
by the FDA. |
We plan to augment our core research and development
capability and assets by conducting Phase I and II clinical trials for investigational new drugs and medical devices in the fields of
CNS, Hematology/Oncology and Ophthalmology.
Our management team has extensive experiences
across a wide range of new drug and medical device development and we have in-licensed new drug and medical device candidates from large
research institutes and universities in both the U.S. and Taiwan. Through an assertive product development approach, we expect that we
will build a substantial portfolio of Oncology/ Hematology, CNS and Ophthalmology products. We primarily focus on Phase I and II research
of new drug candidates and out license the post-Phase-II products to pharmaceutical companies; we do not expect to devote substantial
efforts and resources to building the disease-specific distribution channels.
Business Objectives
The Company is operating its core business
based on collaborative activities that can generate current and future revenues through research, development and/or commercialization
joint venture agreements. The terms of these agreements typically include payment to the Company related to one or more of the following:
| ● | nonrefundable
upfront license fees, |
| ● | development
and commercial milestones, |
| ● | partial
or complete reimbursement of research and development costs and |
| ● | royalties
on net sales of licensed products. |
Each type of payments results in revenue except
for revenue from royalties on net sales of licensed products, which are classified as royalty revenues. To date, we have not received
any royalty revenues. Revenue is recognized upon satisfaction of a performance obligation by transferring control of a good or service
to the joint venture partner.
As part of the accounting for these arrangements,
the Company applies judgment to determine whether the performance obligations are distinct and develop assumptions in determining the
stand-alone selling price for each distinct performance obligation identified in the collaboration agreements. To determine the stand-alone
selling price, the Company relies on assumptions which may include forecasted revenues, development timelines, reimbursement rates for
R&D personnel costs, discount rates and probabilities of technical and regulatory success.
The Company had multiple deliverables under
the collaborative agreements, including deliverables relating to grants of technology licenses, regulatory and clinical development,
and marketing activities. Estimation of the performance periods of the Company’s deliverables requires the use of management’s
judgment. Significant factors considered in management’s evaluation of the estimated performance periods include, but are not limited
to, the Company’s experience in conducting clinical development, regulatory and manufacturing activities. The Company reviews the
estimated duration of its performance periods under its collaborative agreements on an annually basis, and makes any appropriate adjustments
on a prospective basis. Future changes in estimates of the performance period under its collaborative agreements could impact the timing
of future revenue recognition. For further details about these difference payment arrangements, see “Summary of Critical Accounting
Policies” below. Examples of recent collaborative agreements the Company has entered into are as follows:
Collaborative agreements with BHK, a
related party
| (i) |
In February and December
of 2015, BioLite, Inc. entered into a total of three joint venture agreements with BioHopeKing to jointly develop ABV-1501 for Triple
Negative Breast Cancer (TNBC), ABV-1504 for MDD and ABV-1505 for ADHD. The agreements granted marketing rights to BioHopeKing for
certain Asian countries in return for a series of milestone payments totaling $10 million in cash and equity of BioHopeKing or equity
securities owned by BioHopeKing. |
The milestone payments are determined by a
schedule of BioLite development achievements as shown below:
| Milestone | |
Payment | |
| Execution of BHK Co-Development Agreement | |
$ | 1,000,000 | |
| Investigational New Drug (IND) Submission | |
$ | 1,000,000 | |
| Phase II Clinical Trial Complete | |
$ | 1,000,000 | |
| Initiation of Phase III Clinical Trial | |
$ | 3,000,000 | |
| New Drug Application (NDA) Submission | |
$ | 4,000,000 | |
| Total | |
$ | 10,000,000 | |
| (ii) | In
December of 2015, BHK paid the initial cash payment of $1 million upon the execution of the
BHK Agreement. The Company concluded that certain deliverables are considered separate units
of accounting as the delivered items have value to the customer on a standalone basis and
recognized this cash payment as collaboration revenue when all research, technical, and development
data was delivered to BHK in 2015. The payment included compensation for past research efforts
and contributions made by BioLite Taiwan before the BHK agreement was signed and does not
relate to any future commitments made by BioLite Taiwan and BHK in the BHK Agreement. |
| (iii) | In
August 2016, the Company received the second milestone payment of $1 million, and recognized
collaboration revenue for the year ended December 31, 2016. As of December 31, 2022, the
Company had completed the phase II clinical trial for ABV-1504 MDD on October 31, 2019, but
has not yet completed the phase II clinical trial for ABV-1505 ADHD. |
| (iv) | In
addition to the milestone payments, BioLite Inc. is entitled to receive a royalty equal to
12% of BHK’s net sales related to ABV-1501, ABV-1504 and ABV-1505 Products. As of December
31, 2022, the Company has not earned royalties under the BHK Co-Development Agreement. |
| (v) | The
BHK Co-Development Agreement will remain in effect for fifteen years from the date of first
commercial sale of the Product in in Asia excluding Japan. |
Collaborative agreement with BioLite,
Inc., a related party
The Company entered into a collaborative agreement
with BioLite, Inc. on December 29, 2015, and then entered into two addendums to such agreement, as amended and revised, (the “BioLite
Agreement”). The majority shareholder of BioLite is one of the Company’s subsidiaries, Mr. Jiang, the Company’s Chairman
is a director of BioLite and Dr. Jiang, the Company’s Chief Strategy Officer and a director, is the Chairman of BioLite.
Pursuant to the BioLite Agreement, the Company
acquired the sole licensing rights to develop and commercialize for therapeutic purposes six compounds from BioLite. In accordance with
the terms of the Agreement, the Company shall pay BioLite (i) milestone payments of up to $100 million in cash and equity of the Company
or equity securities owned by it at various stages on a schedule dictated by BioLite’s achievements of certain milestones, as set
forth in the Agreement (the “Milestone Payments”) and (ii) a royalty payment equal to 5% of net sales of the drug products
when ABV-1501 is approved for sale in the licensed territories. If BioLite fails to reach any of the milestones in a timely manner, it
may not receive the rest of the payments from the Company.According to the BioLite Agreement, after Phase II clinical trials are completed,
15% of the Milestone Payment becomes due and shall be paid in two stages: (i) 5% no later than December 31, 2021 (the “December
2021 Payment”) and (ii) 10% no later than December 31, 2022.
On February 12, 2022, the Company’s
Board of Directors determined that the December 2021 Payment, which is equal to $5,000,000, shall be paid via the cancellation of certain
outstanding debt, in the amount of $5,000,000, that BioLite owes the Company as of December 31, 2021.
On February 22, 2022, the parties entered
into an amendment to the BioLite Agreement allowing the Company to make all payments due under the Agreement via the forgiveness of debt,
in equal value, owed by BioLite to the Company.
This was a related party transaction.
Co-Development agreement with Rgene
Corporation, a related party
On May 26, 2017, the Company entered into
a co-development agreement (the “Rgene Agreement”) with Rgene Corporation (the “Rgene”), a related party under
common control by the controlling beneficiary shareholder of YuanGene Corporation and the Company (See Note 12). Pursuant to the Rgene
Agreement, BriVision and Rgene agreed to co-develop and commercialize ABV-1507 HER2/neu Positive Breast Cancer Combination Therapy, ABV-1703
Pancreatic Cancer Combination Therapy and ABV-1527 Ovary Cancer Combination Therapy. Under the terms of the Rgene Agreement, Rgene is
required to pay the Company $3,000,000 in cash or stock of Rgene with equivalent value by August 15, 2017 as compensation of BriVision’s
past research efforts and contributions made by BriVision before the Rgene Agreement was executed. The payment does not relate to any
future milestones attained by BriVision. In addition to $3,000,000, the Company is entitled to receive 50% of the future net licensing
income or net sales profit earned by Rgene. All development costs shall be equally shared by both BriVision and Rgene.
On June 1, 2017, the Company delivered all
research, technical data and development data to Rgene pursuant to the Rgene Agreement in return for a cash payment of $450,000 and 1,530,000
common shares of Rgene stock valued at $2,550,000, which in 2018 was accounted for using the equity method long-term investment. On December
31, 2018, the Company determined to fully write off this investment based on the Company’s assessment of the severity and duration
of the impairment, and qualitative and quantitative analysis of the operating performance of the investee, adverse changes in market
conditions, the regulatory or economic environment, changes in operating structure of Rgene, additional funding requirements and Rgene’s
ability to remain in business. All research projects that were initiated will be managed and funded equally by the Company and Rgene.
The Company and Rgene signed an amendment
to the Rgene Agreement on November 10, 2020, pursuant to which both parties agreed to delete AB-1507 HER2/neu Positive Breast Cancer
Combination Therapy and AB-1527 Ovary Cancer Combination Therapy and add ABV-1519 EGFR Positive Non-Small Cell Lung Cancer Combination
Therapy and ABV-1526 Large Intestine / Colon / Rectal Cancer Combination Therapy to the products to be co-developed and commercialized.
Other provisions of the Rgene Agreement remain in full force and effect.
Clinical Development Service Agreement
with Rgene Corporation, a related party
On June 10, 2022, the Company expanded its
co-development partnership with Rgene. The Company’s subsidiary, BioKey, entered into a Clinical Development Service Agreement
with Rgene (“Service Agreement”) to guide certain Rgene drug products, RGC-1501 for the treatment of Non-Small Cell Lung
Cancer (NSCLC), RGC-1502 for the treatment of pancreatic cancer and RGC 1503 for the treatment of colorectal cancer patients, through
completion of Phase II clinical studies under U.S. FDA IND regulatory requirements (the “Rgene Studies”). Under the terms
of the Service Agreement, BioKey is eligible to receive payments totaling up to $3.0 million over a 3-year period with each payment amount
to be determined by certain regulatory milestones obtained during the agreement period.
Through a series of transactions over the
past 5 years, the Company and Rgene have co-developed the three drug products covered by the Service Agreement, which has resulted in
the Company owning 31.62% of Rgene.
As part of the Rgene Studies, the Company
agreed to loan $1.0 million to Rgene, for which Rgene has provided the Company with a 5% working capital convertible loan (the “Note”).
If the Note is fully converted, the Company will own an additional 6.4% of Rgene. The Company is expected to receive the outstanding
loan from the related party by the first half of 2024, either by cash or conversion of shares of Rgene. The Company may convert the Note
at any time into shares of Rgene’s common stock at either (i) a fixed conversion price equal to $1.00 per share or (ii) 20% discount
of the stock price of the then most recent offering, whichever is lower; the conversion price is subject to adjustment as set forth in
the Note. The Note includes standard events of default, as well as a cross default provision pursuant to which a breach of the Service
Agreement will trigger an event of default under the Note if not cured after 5 business days of written notice regarding the breach is
provided. Upon an event of default, the outstanding principal and any accrued and unpaid interest shall be immediately due and payable.
The Service Agreement shall remain in effect
until the expiration date of the last patent and automatically renew for 5 more years unless terminated earlier by either party with
six months written notice. Either party may terminate the Service Agreement for cause by providing 30 days written notice.
Rgene has further agreed, effective July 1,
2022, to provide the Company with a seat on Rgene’s Board of Directors until the loan is repaid in full. The Company has nominated
Dr. Jiang, its Chief Strategy Officer and Director to occupy that seat; Dr. Jiang is also one of the Company’s largest shareholders,
owning 12.8% of the Company.
The Rgene Studies is a related party transaction.
Collaborative agreement with BioFirst
Corporation, a related party
On July 24, 2017, the Company entered into
a collaborative agreement (the “BioFirst Agreement”) with BioFirst Corporation, a corporation incorporated under the laws
of Taiwan (“BioFirst”), pursuant to which BioFirst granted the Company global licensing rights to medical use of ABV-1701
Vitreous Substitute for Vitrectomy. BioFirst is a related party to the Company because a controlling beneficiary shareholder of YuanGene
Corporation and the Company is a Director and shareholders of BioFirst (See Note 12).
Pursuant to the BioFirst Agreement, the Company
and BioFirst will co-develop and commercialize BFC-1401. The Company will pay BioFirst a total amount of $3,000,000 in cash or stock
of the Company before September 30, 2018 as payment in full for BioFirst’s past research efforts and contributions made by BioFirst
before the BioFirst Agreement was executed. The Company is entitled to receive 50% of any future net licensing revenue or net profit
associated with Vitargus®. All development cost will be equally shared by both BriVision and BioFirst.
On September 25, 2017, BioFirst delivered
all research, technical, data and development data to the Company. For the year ended September 30, 2017, the Company determined to fully
expense the entire amount of $3,000,000 since the related licensing rights do not have alternative future uses. According to ASC 730-10-25-1,
absent alternative future uses the acquisition of product rights to be used in research and development activities must be charged to
research and development expenses immediately. Hence, the entire amount of $3,000,000 is fully expensed as research and development expense
during the year ended September 30, 2017.
On June 30, 2019, the Company entered into
a Stock Purchase Agreement (the “Purchase Agreement”) with BioFirst. Pursuant to the Purchase Agreement, the Company issued
428,571 shares of the Company’s common stock to BioFirst as payment for $3,000,000 owed by the Company to BioFirst in connection
with the BioFirst Agreement.
On August 5, 2019, the Company entered into
a second Stock Purchase Agreement with BioFirst whereby the Company issued 414,702 shares of the Company’s common stock to BioFirst
as repayment in full for a loan in the amount of $2,902,911 provided to BriVision from BioFirst.
On November 4, 2020, the Company executed
an amendment to the BioFirst Agreement with BioFirst to add ABV-2001 Intraocular Irrigation Solution and ABV-2002 Corneal Storage Solution
to the agreement. ABV-2002 is utilized during a corneal transplant procedure to replace a damaged or diseased cornea while ABV-2001 has
broader utilization during a variety of ocular procedures.
Initially the Company will focus on ABV-2002,
a solution utilized to store a donor cornea prior to either penetrating keratoplasty (full thickness cornea transplant) or endothelial
keratoplasty (back layer cornea transplant). ABV-2002 is a solution comprised of a specific poly amino acid that protects ocular tissue
from damage caused by external osmolarity exposure during pre-surgery storage. The specific polymer in ABV-2002 can adjust osmolarity
to maintain a range of 330 to 390 mOsM thereby permitting hydration within the corneal stroma during the storage period. Stromal hydration
results in (a) maintaining acceptable corneal transparency and (b) prevents donor cornea swelling. ABV-2002 also contains an abundant
phenolic phytochemical found in plant cell walls that provides antioxidant antibacterial properties and neuroprotection.
Early testing by BioFirst indicates that ABV-2002
may be more effective for protecting the cornea and retina during long-term storage than other storage media available today and can
be manufactured at lower cost. Further clinical development was put on hold due to the lack of funding.
In addition, BioFirst was incorporated on
November 7, 2006, focusing on the R&D, manufacturing, and sales of innovative patented pharmaceutical products. The technology of
BioFirst comes from the global exclusive licensing agreements BioFirst maintains with domestic R & D institutions. Currently, BioFirst’s
main research and development product is the vitreous substitute (Vitargus®), licensed by the National Health Research Institutes.
Vitargus is the world’s first bio-degradable vitreous substitute and offers a number of advantages over current vitreous substitutes
by minimizing medical complications and reducing the need for additional surgeries.
Vitargus has started the construction of a
GMP factory in Hsinchu Biomedical Science Park, Taiwan, with the aim at building a production base to supply the global market, and promote
the construction of bio-degradable vitreous substitute manufacturing centers in Taiwan. Completion of this factory would allow ABVC to
manufacture Vitargus with world-class technology in a GMP certified pharmaceutical factory. BioFirst is targeting to complete the construction
in 2024.
Co-Development agreement with BioLite
Japan K.K., a related party
On October 6, 2021 (the “Completion
Date”), the Company, Lucidaim Co., Ltd., a Japanese corporation (“Lucidaim,” together with the Company,
the “Shareholders”), and BioLite Japan K.K., a Japanese corporation (“Biolite JP”) entered into
a Joint Venture Agreement (the “Agreement”). Biolite JP is a private limited company (a Japanese Kabushiki Kaisha)
incorporated on December 18, 2018 and at the date of the Agreement has 10,000 ordinary shares authorized, with 3,049 ordinary shares
issued and outstanding (the “Ordinary Shares”). Immediately prior to the execution of the Agreement, Lucidaim owned
1,501 ordinary shares and the Company owned the 1,548 ordinary shares. The Shareholders entered into the joint venture to formally reduce
to writing their desire to invest in and operate Biolite as a joint venture. The business of the joint venture shall be the research
and development of drugs, medical device and digital media, investment, fund running and consulting, distribution and marketing of supplements
carried on by Biolite and its subsidiaries in Japan, or any other territory or businesses as may from time to time be agreed by an amendment
to the Agreement. The closing of the transaction is conditioned upon the approval and receipt of all necessary government approvals,
which have been received.
Pursuant to the Agreement and the related
share transfer agreement, the Company shall transfer 54 of its Ordinary Shares to Lucidaim for no consideration, such that following
the transfer, Lucidaim shall own 1,555 Ordinary Shares (51%) and the Company shall own 1,494 Ordinary Shares (49%). Also pursuant to
the Agreement, there shall be 3 directors of Biolite JP, consisting of 1 director appointed by the Company and 2 appointed by Lucidiam.
The Company shall appoint Eugene Jiang, the Company’s current Chairman and Chief Business Officer and Lucidaim shall appoint Michihito
Onishi; the current director of Biolite JP, Toru Seo (who is also a director of BioLite Japan’s other shareholder), is considered
the second Lucidaim director. The Agreement further provides that the Company and Biolite shall assign the research collaboration and
license agreement between them to Biolite or prepare the same (the “License Agreement”). The aforementioned transactions
occurred on the Completion Date.
As per the Agreement, the Shareholders shall
supervise and manage the business and operations of Biolite JP. The directors shall not be entitled to any renumeration for their services
as a director and each Shareholder can remove and replace the director he/she/it appointed. If a Shareholder sells or disposes of all
of its Ordinary Shares, the director such Shareholder appointed must tender his/her resignation. The Agreement also sets forth certain
corporate actions that must be pre-approved by all Shareholders (the “Reserved Matters”). If the Shareholders are
unable to make a decision on any Reserved Matter, then either Shareholder can submit a deadlock notice to the other shareholder, 5 days
after which they must refer the matter to each Shareholder’s chairman and use good faith to resolve the dispute. If such dispute
is not resolved within 10 days thereafter, then either Shareholder can offer to buy all of the other Shareholder’s Ordinary Shares
for cash at a specified price; if there is not affirmative acceptance of the sale, the sale shall proceed as set forth in the sale offer.
Each of the Shareholders maintains a pre-emptive
right to purchase such number of additional Ordinary Shares as would allow such Shareholder to maintain its ownership percentage in Biolite
JP if Biolite JP issues any new Ordinary Shares. However, the Agreement provides that the Company shall lose its pre-emptive rights under
certain conditions. The Shareholders also maintain a right of first refusal if the other Shareholder receives an offer to buy such shareholder’s
Ordinary Shares.
The Agreement also requires Biolite JP to
obtain a bank facility in the amount of JPY 30,460,000 (approximately USD272,000), for its initial working capital purposes. Pursuant
to the Agreement, each Shareholder agrees to guarantee such bank facility if the bank requires a guarantee. Accordingly, the Company
may be liable for the bank facility in an amount up to JPY 14,925,400 (approximately USD134,000), which represents 49% of the maximum
bank facility. The Agreement further provides that Biolite JP shall issue annual dividends at the rate of at least 1.5% of Biolite JP’s
profits, if it has sufficient cash to do so.
Pursuant to the Agreement, the Company and
Biolite JP agree to use their best efforts to execute the License Agreement by the end of December 2021. The Company agreed that any
negotiation on behalf of Biolite JP regarding the terms of the License Agreement shall be handled by the directors appointed by Lucidaim.
If the Company and such Lucidaim directors do not reach agreement on the terms, Biolite JP may at its sole discretion determine not to
execute the License Agreement without any liability to the Company.
The Agreement contains non-solicitation and
non-compete clauses for a period of 2 years after a Shareholder or its subsidiaries ceases to be a Shareholder, with such restrictive
covenants limited to business within the ophthalmologic filed or central neurological field. Any rights to intellectual property that
arise from Biolite JP’s activities, shall belong to Biolite JP.
The Agreement contains standard indemnification
terms, except that no indemnifying party shall have any liability for an individual liability unless it exceeds JPY 500,000 (approximately
USD4,500) and until the aggregate amount of all liabilities exceeds JPY 2,000,000 (approximately USD18,000) and then only to the extent
such liability exceed such limit.
The Company paid $150,000 towards the setup
of the joint venture; BioLite Japan’s other shareholder also paid $150,000 after the Letter of Intent was signed.
The Agreement shall continue for 10 years,
unless earlier terminated. The Agreement also allows a Shareholder to terminate the agreement upon certain defaults committed by another
Shareholder, as set forth in the Agreement.
This was a related party transaction.
In November 2021, the Company received $4,244,452
in gross proceeds from the exercise of warrants issued in the Company’s August 3, 2021, public offering of securities. Investors
exercised a total of 673,405 Series A warrants at a price of $6.30 per share, and 200 Series B warrants at a price of $10 per share.
BioKey Revenues
In addition to collaborative agreements, ABVC
earns revenue through its wholly owned BioKey subsidiary which provides a wide range of Contract Development & Manufacturing Organization
(“CDMO”) services including API characterization, pre-formulation studies, formulation development, analytical method development,
stability studies, IND/NDA/ANDA/510K submissions, and manufacturing clinical trial materials (from Phase I through Phase III) and commercial
manufacturing of pharmaceutical products.
In addition, BioKey provides a variety of
regulatory services tailored to the needs of its customers, which include proofreading and regulatory review of submission documents
related to formulation development, clinical trials, marketed products, generics, nutraceuticals and OTC products and training presentations.
In addition to supporting ABVC’s new drug development, BioKey submits INDs, NDAs, ANDAs, and DMFs to the FDA, on ABVC’s behalf
in compliance with new electronic submission guidelines of the FDA.
Impact of COVID-19 Outbreak
On January 30, 2020, the World Health Organization
declared the coronavirus outbreak a “Public Health Emergency of International Concern” and on March 10, 2020, declared it
to be a pandemic. Actions taken around the world to help mitigate the spread of the coronavirus include restrictions on travel, and quarantines
in certain areas, and forced closures for certain types of public places and businesses. The coronavirus and actions taken to mitigate
it have had and are expected to continue to have an adverse impact on the economies and financial markets of many countries, including
the geographical area in which the Company operates.
Due to the COVID-19 pandemic, our revenue
for the fiscal 2022 were significantly impacted. In 2023, our business started recovering from the COVID-19 impact. We have been working
on new contracts towards revenue generation and increase in sales of existing products and incorporating new products for sale.
The COVID-19 pandemic, including variants,
has adversely affected, and is expected to continue to adversely affect, elements of our CDMO business sector. The COVID-19 pandemic
government imposed restrictions constrained researcher access to labs globally. These constraints limited scientific discovery capacity
and we observed that demand in those labs fell well below historic levels. As constraints on social distancing were gradually lifted
around the world recently, labs have been able to increase research activity. While we believe that underlying demand is still not yet
at pre-COVID-19 levels since lab operations remain below their normal capacity, we are hopeful that the vaccination programs that are
underway combined with policy changes planned for the summer will further increase research activity and support a return to pre-COVID-19
demand levels worldwide.
The global pandemic of COVID-19 continues
to evolve rapidly, and we will continue to monitor the situation closely, including its potential effect on our plans and timelines.
Additionally, it is reasonably possible that
estimates made in the financial statements have been, or will be, materially and adversely impacted in the near term as a result of these
conditions, including losses on inventory; impairment losses related to goodwill and other long-lived assets and current obligations.
Summary of Critical Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements
have been prepared in accordance with the generally accepted accounting principles in the United States of America (the “U.S. GAAP”).
All significant intercompany transactions and account balances have been eliminated.
This basis of accounting involves the application
of accrual accounting and consequently, revenues and gains are recognized when earned, and expenses and losses are recognized when incurred.
The Company’s financial statements are expressed in U.S. dollars.
Fiscal Year
The Company changed its fiscal year from the
period beginning on October 1st and ending on September 30th to the period beginning on January 1st
and ending on December 31st, beginning January 1, 2018.
Use of Estimates
The preparation of financial statements in
conformity with generally accepted accounting principles in the United States of America requires management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated
financial statements and the amount of revenues and expenses during the reporting periods. Actual results could differ materially from
those results.
Stock Reverse Split
On July 25, 2023, the Company filed a Certificate
of Amendment to its Articles of Incorporation authorizing a 1-for-10 reverse stock split of the issued and outstanding shares of its
common stock. The Company’s stockholders previously approved the Reverse Stock Split at the Company’s Special Shareholder
Meeting held on July 7, 2023. The Reverse Stock Split was effected to reduce the number of issued and outstanding shares and to increase
the per share trading value of the Company’s common stock, although that outcome is not guaranteed. In turn, the Company believes
that the Reverse Stock Split will enable the Company to restore compliance with certain continued listing standards of NASDAQ Capital
Market. All shares and related financial information in this Form 10-K reflect this 1-for-10 reverse stock split.
Fair Value Measurements
FASB ASC 820, “Fair Value Measurements”
defines fair value for certain financial and nonfinancial assets and liabilities that are recorded at fair value, establishes a framework
for measuring fair value and expands disclosures about fair value measurements. It requires that an entity measure its financial instruments
to base fair value on exit price, maximize the use of observable units and minimize the use of unobservable inputs to determine the exit
price. It establishes a hierarchy which prioritizes the inputs to valuation techniques used to measure fair value. This hierarchy increases
the consistency and comparability of fair value measurements and related disclosures by maximizing the use of observable inputs and minimizing
the use of unobservable inputs by requiring that observable inputs be used when available. Observable inputs are inputs that reflect
the assumptions market participants would use in pricing the assets or liabilities based on market data obtained from sources independent
of the Company. Unobservable inputs are inputs that reflect the Company’s own assumptions about the assumptions market participants
would use in pricing the asset or liability developed based on the best information available in the circumstances. The hierarchy prioritizes
the inputs into three broad levels based on the reliability of the inputs as follows:
| ● | Level
1 - Inputs are quoted prices in active markets for identical assets or liabilities that the
Company has the ability to access at the measurement date. Valuation of these instruments
does not require a high degree of judgment as the valuations are based on quoted prices in
active markets that are readily and regularly available. |
| ● | Level
2 - Inputs other than quoted prices in active markets that are either directly or indirectly
observable as of the measurement date, such as quoted prices for similar assets or liabilities;
quoted prices in markets that are not active; or other inputs that are observable or can
be corroborated by observable market data for substantially the full term of the assets or
liabilities. |
| ● | Level
3 - Valuations based on inputs that are unobservable and not corroborated by market data.
The fair value for such assets and liabilities is generally determined using pricing models,
discounted cash flow methodologies, or similar techniques that incorporate the assumptions
a market participant would use in pricing the asset or liability. |
The carrying values of certain assets and
liabilities of the Company, such as cash and cash equivalents, restricted cash, accounts receivable, due from related parties, inventory,
prepaid expenses and other current assets, accounts payable, accrued liabilities, and due to related parties approximate fair value due
to their relatively short maturities. The carrying value of the Company’s short-term bank loan, convertible notes payable, and
accrued interest approximates their fair value as the terms of the borrowing are consistent with current market rates and the duration
to maturity is short. The carrying value of the Company’s long-term bank loan approximates fair value because the interest rates
approximate market rates that the Company could obtain for debt with similar terms and maturities.
Concentration of Clients
As of December 31, 2023, the most major client,
specializes in developing and commercializing of dietary supplements and therapeutics in dietary supplement industry, accounted for 87.24%
of the Company’s total account receivable.
As of December 31, 2022, the most major clients,
specializes in developing and commercializing of dietary supplements and therapeutics in dietary supplement industry, accounted for 71.89%
of the Company’s total account receivable; the second major client with its Chairman being the Board of Director of BioKey, accounted
for 16.62% of the Company’s total account receivable.
For the year ended December 31, 2023, the
most major client, distributing nutritional supplement in Asia Pacific, accounted for 80.04% of the Company’s total revenues. For
the year ended December 31, 2022, one major client, who is a Shareholder of the Company that works in development and commercialization
of new drugs in Taiwan, accounted for 93.22% of the Company’s total revenues.
Cash and Cash Equivalents
The Company considers highly liquid investments
with maturities of three months or less to be cash equivalents when purchased. As of December 31, 2023 and 2022, the Company’s
cash and cash equivalents amounted to $60,155 and $85,265, respectively. Some of the Company’s cash deposits are held in financial
institutions located in Taiwan where there is currently regulation mandated on obligatory insurance of bank accounts. The Company believes
this financial institution is of high credit quality.
Restricted Cash
Restricted cash primarily consist of cash
held in a reserve bank account in Taiwan. As of December 31, 2023 and 2022, the Company’s restricted cash amounted $656,625 and
$1,306,463, respectively.
Inventory
Inventory consists of raw materials, work-in-process,
finished goods, and merchandise. Inventories are stated at the lower of cost or market and valued on a moving weighted average cost basis.
Market is determined based on net realizable value. The Company periodically reviews the age and turnover of its inventory to determine
whether any inventory has become obsolete or has declined in value, and incurs a charge to operations for known and anticipated inventory
obsolescence.
Concentration of Credit Risk
The Company’s financial instruments
that are exposed to concentrations of credit risk consist primarily of cash and cash equivalents. The Company places its cash and temporary
cash investments in high quality credit institutions, but these investments may be in excess of Taiwan Central Deposit Insurance Corporation
and the U.S. Federal Deposit Insurance Corporation’s insurance limits. The Company does not enter into financial instruments for
hedging, trading or speculative purposes.
We perform ongoing credit evaluation of our
customers and requires no collateral. An allowance for doubtful accounts is provided based on a review of the collectability of accounts
receivable. We determine the amount of allowance for doubtful accounts by examining its historical collection experience and current
trends in the credit quality of its customers as well as its internal credit policies. Actual credit losses may differ from our estimates.
Accounts receivable and allowance for expected credit losses
accounts
Accounts receivable is recorded and carried
at the original invoiced amount less an allowance for any potential uncollectible amounts.
The Company make estimates of expected credit
and collectability trends for the allowance for credit losses and allowance for unbilled receivables based upon our assessment of various
factors, including historical experience, the age of the accounts receivable balances, credit quality of customers, current economic
conditions reasonable and supportable forecasts of future economic conditions, and other factors that may affect our ability to collect
from customers. The provision is recorded against accounts receivable balances, with a corresponding charge recorded in the consolidated
statements of income. Actual amounts received may differ from management’s estimate of credit worthiness and the economic environment.
Delinquent account balances are written-off against the allowance for doubtful accounts after management has determined that the likelihood
of collection is not probable.
Allowance for expected credit losses accounts
was $616,505 and $194,957 as of December 31, 2023 and 2022, respectively.
Revenue Recognition
During the fiscal year 2018, the Company adopted
Accounting Standards Codification (“ASC”), Topic 606 (ASC 606), Revenue from Contracts with Customers, using the modified
retrospective method to all contracts that were not completed as of January 1, 2018, and applying the new revenue standard as an adjustment
to the opening balance of accumulated deficit at the beginning of 2018 for the cumulative effect. The results for the Company’s
reporting periods beginning on and after January 1, 2018 are presented under ASC 606, while prior period amounts are not adjusted and
continue to be reported under the accounting standards in effect for the prior period. Based on the Company’s review of existing
collaborative agreements as of January 1, 2018, the Company concluded that the adoption of the new guidance did not have a significant
change on the Company’s revenue during all periods presented.
Pursuant to ASC 606, the Company recognizes
revenue when its customer obtains control of promised goods or services, in an amount that reflects the consideration that the Company
expects to receive in exchange for those goods or services. To determine revenue recognition for arrangements that the Company determines
is within the scope of ASC 606, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify
the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance
obligations in the contract; and (v) recognize revenue when (or as) the Company satisfies a performance obligation. The Company only
applies the five-step model to contracts when it is probable that the Company will collect the consideration the Company is entitled
to in exchange for the goods or services the Company transfers to the customers. At inception of the contract, once the contract is determined
to be within the scope of ASC 606, the Company assesses the goods or services promised within each contract, determines those that are
performance obligations, and assesses whether each promised good or service is distinct. The Company then recognizes as revenue the amount
of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied.
The following are examples of when the Company
recognizes revenue based on the types of payments the Company receives.
Collaborative Revenues - The Company
recognizes collaborative revenues generated through collaborative research, development and/or commercialization agreements. The terms
of these agreements typically include payment to the Company related to one or more of the following: non-refundable upfront license
fees, development and commercial milestones, partial or complete reimbursement of research and development costs, and royalties on net
sales of licensed products. Each type of payments results in collaborative revenues except for revenues from royalties on net sales of
licensed products, which are classified as royalty revenues. To date, the Company has not received any royalty revenues. Revenue is recognized
upon satisfaction of a performance obligation by transferring control of a good or service to the collaboration partners.
As part of the accounting for these arrangements,
the Company applies judgment to determine whether the performance obligations are distinct, and develop assumptions in determining the
stand-alone selling price for each distinct performance obligation identified in the collaboration agreements. To determine the stand-alone
selling price, the Company relies on assumptions which may include forecasted revenues, development timelines, reimbursement rates for
R&D personnel costs, discount rates and probabilities of technical and regulatory success.
The Company had multiple deliverables under
the collaborative agreements, including deliverables relating to grants of technology licenses, regulatory and clinical development,
and marketing activities. Estimation of the performance periods of the Company’s deliverables requires the use of management’s
judgment. Significant factors considered in management’s evaluation of the estimated performance periods include, but are not limited
to, the Company’s experience in conducting clinical development, regulatory and manufacturing activities. The Company reviews the
estimated duration of its performance periods under its collaborative agreements on an annually basis, and makes any appropriate adjustments
on a prospective basis. Future changes in estimates of the performance period under its collaborative agreements could impact the timing
of future revenue recognition.
(i) Non-refundable upfront payments
If a license to the Company’s intellectual
property is determined to be distinct from the other performance obligations identified in an arrangement, the Company recognizes revenue
from the related non-refundable upfront payments based on the relative standalone selling price prescribed to the license compared to
the total selling price of the arrangement. The revenue is recognized when the license is transferred to the collaboration partners and
the collaboration partners are able to use and benefit from the license. To date, the receipt of non-refundable upfront fees was solely
for the compensation of past research efforts and contributions made by the Company before the collaborative agreements entered into
and it does not relate to any future obligations and commitments made between the Company and the collaboration partners in the collaborative
agreements.
(ii) Milestone payments
The Company is eligible to receive milestone
payments under the collaborative agreement with collaboration partners based on achievement of specified development, regulatory and
commercial events. Management evaluated the nature of the events triggering these contingent payments, and concluded that these events
fall into two categories: (a) events which involve the performance of the Company’s obligations under the collaborative agreement
with collaboration partners, and (b) events which do not involve the performance of the Company’s obligations under the collaborative
agreement with collaboration partners.
The former category of milestone payments
consists of those triggered by development and regulatory activities in the territories specified in the collaborative agreements. Management
concluded that each of these payments constitute substantive milestone payments. This conclusion was based primarily on the facts that
(i) each triggering event represents a specific outcome that can be achieved only through successful performance by the Company of one
or more of its deliverables, (ii) achievement of each triggering event was subject to inherent risk and uncertainty and would result
in additional payments becoming due to the Company, (iii) each of the milestone payments is non-refundable, (iv) substantial effort is
required to complete each milestone, (v) the amount of each milestone payment is reasonable in relation to the value created in achieving
the milestone, (vi) a substantial amount of time is expected to pass between the upfront payment and the potential milestone payments,
and (vii) the milestone payments relate solely to past performance. Based on the foregoing, the Company recognizes any revenue from these
milestone payments in the period in which the underlying triggering event occurs.
(iii) Multiple Element Arrangements
The Company evaluates multiple element arrangements
to determine (1) the deliverables included in the arrangement and (2) whether the individual deliverables represent separate units of
accounting or whether they must be accounted for as a combined unit of accounting. This evaluation involves subjective determinations
and requires management to make judgments about the individual deliverables and whether such deliverables are separate from other aspects
of the contractual relationship. Deliverables are considered separate units of accounting provided that: (i) the delivered item(s) has
value to the customer on a standalone basis and (ii) if the arrangement includes a general right of return relative to the delivered
item(s), delivery or performance of the undelivered item(s) is considered probable and substantially within its control. In assessing
whether an item under a collaboration has standalone value, the Company considers factors such as the research, manufacturing, and commercialization
capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. The Company also
considers whether its collaboration partners can use the other deliverable(s) for their intended purpose without the receipt of the remaining
element(s), whether the value of the deliverable is dependent on the undelivered item(s), and whether there are other vendors that can
provide the undelivered element(s).
The Company recognizes arrangement consideration
allocated to each unit of accounting when all of the revenue recognition criteria in ASC 606 are satisfied for that particular unit of
accounting. In the event that a deliverable does not represent a separate unit of accounting, the Company recognizes revenue from the
combined unit of accounting over the Company’s contractual or estimated performance period for the undelivered elements, which
is typically the term of the Company’s research and development obligations. If there is no discernible pattern of performance
or objectively measurable performance measures do not exist, then the Company recognizes revenue under the arrangement on a straight-line
basis over the period the Company is expected to complete its performance obligations. Conversely, if the pattern of performance in which
the service is provided to the customer can be determined and objectively measurable performance measures exist, then the Company recognizes
revenue under the arrangement using the proportional performance method. Revenue recognized is limited to the lesser of the cumulative
amount of payments received or the cumulative amount of revenue earned, as determined using the straight-line method or proportional
performance method, as applicable, as of the period ending date.
At the inception of an arrangement that includes
milestone payments, the Company evaluates whether each milestone is substantive and at risk to both parties on the basis of the contingent
nature of the milestone. This evaluation includes an assessment of whether: (1) the consideration is commensurate with either the Company’s
performance to achieve the milestone or the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting
from its performance to achieve the milestone, (2) the consideration relates solely to past performance and (3) the consideration is
reasonable relative to all of the deliverables and payment terms within the arrangement. The Company evaluates factors such as the scientific,
clinical, regulatory, commercial, and other risks that must be overcome to achieve the particular milestone and the level of effort and
investment required to achieve the particular milestone in making this assessment. There is considerable judgment involved in determining
whether a milestone satisfies all of the criteria required to conclude that a milestone is substantive. Milestones that are not considered
substantive are recognized as earned if there are no remaining performance obligations or over the remaining period of performance, assuming
all other revenue recognition criteria are met.
(iv) Royalties and Profit Sharing Payments
Under the collaborative agreement with the
collaboration partners, the Company is entitled to receive royalties on sales of products, which is at certain percentage of the net
sales. The Company recognizes revenue from these events based on the revenue recognition criteria set forth in ASC 606. Based on those
criteria, the Company considers these payments to be contingent revenues, and recognizes them as revenue in the period in which the applicable
contingency is resolved.
Revenues Derived from Research and Development
Activities Services - Revenues related to research and development and regulatory activities are recognized when the related services
or activities are performed, in accordance with the contract terms. The Company typically has only one performance obligation at the
inception of a contract, which is to perform research and development services. The Company may also provide its customers with an option
to request that the Company provides additional goods or services in the future, such as active pharmaceutical ingredient, API, or IND/NDA/ANDA/510K
submissions. The Company evaluates whether these options are material rights at the inception of the contract. If the Company determines
an option is a material right, the Company will consider the option a separate performance obligation.
If the Company is entitled to reimbursement
from its customers for specified research and development expenses, the Company accounts for the related services that it provides as
separate performance obligations if it determines that these services represent a material right. The Company also determines whether
the reimbursement of research and development expenses should be accounted for as revenues or an offset to research and development expenses
in accordance with provisions of gross or net revenue presentation. The Company recognizes the corresponding revenues or records the
corresponding offset to research and development expenses as it satisfies the related performance obligations.
The Company then determines the transaction
price by reviewing the amount of consideration the Company is eligible to earn under the contracts, including any variable consideration.
Under the outstanding contracts, consideration typically includes fixed consideration and variable consideration in the form of potential
milestone payments. At the start of an agreement, the Company’s transaction price usually consists of the payments made to or by
the Company based on the number of full-time equivalent researchers assigned to the project and the related research and development
expenses incurred. The Company does not typically include any payments that the Company may receive in the future in its initial transaction
price because the payments are not probable. The Company would reassess the total transaction price at each reporting period to determine
if the Company should include additional payments in the transaction price.
The Company receives payments from its customers
based on billing schedules established in each contract. Upfront payments and fees may be recorded as advance from customers upon receipt
or when due, and may require deferral of revenue recognition to a future period until the Company performs its obligations under these
arrangements. Amounts are recorded as accounts receivable when the right of the Company to consideration is unconditional. The Company
does not assess whether a contract has a significant financing component if the expectation at contract inception is such that the period
between payment by the customers and the transfer of the promised goods or services to the customers will be one year or less.
Property and Equipment
Property and equipment is carried at cost
net of accumulated depreciation. Repairs and maintenance are expensed as incurred. Expenditures that improve the functionality of the
related asset or extend the useful life are capitalized. When property and equipment is retired or otherwise disposed of, the related
gain or loss is included in operating income. Leasehold improvements are depreciated on the straight-line method over the shorter of
the remaining lease term or estimated useful life of the asset. Depreciation is calculated on the straight-line method, including property
and equipment under capital leases, generally based on the following useful lives:
| | |
Estimated Life
in Years |
| Buildings and leasehold improvements | |
5 ~ 50 |
| Machinery and equipment | |
5 ~ 10 |
| Office equipment | |
3 ~ 6 |
Construction-in-Progress
The Company acquires constructions that constructs
certain of its fixed assets. All direct and indirect costs that are related to the construction of fixed assets and incurred before the
assets are ready for their intended use are capitalized as construction-in-progress. No depreciation is provided in respect of construction-in-progress.
Construction in progress is transferred to specific fixed asset items and depreciation of these assets commences when they are ready
for their intended use. The Company acquired 20% of the ownership of a certain property and parcel of land owned by Zhonghui, with a
view to jointly develop the property into a healthcare center for senior living, long-term care, and medical care in the areas of ABVC’s
special interests, such as Ophthalmology, Oncology, and Central Nervous Systems. The plan is to establish a base for the China market
and global development of these interests. The Company is a party to a related cooperation agreement with Zhonghui, but is awaiting final
asset ownership certification from the Chinese government.
Impairment of Long-Lived Assets
The Company has adopted Accounting Standards
Codification subtopic 360-10, Property, Plant and Equipment (“ASC 360-10”). ASC 360-10 requires that long-lived assets and
certain identifiable intangibles held and used by the Company be reviewed for impairment whenever events or changes in circumstances
indicate that the carrying amount of an asset may not be recoverable. The Company evaluates its long-lived assets for impairment annually
or more often if events and circumstances warrant. Events relating to recoverability may include significant unfavorable changes in business
conditions, recurring losses, or a forecasted inability to achieve break-even operating results over an extended period. Should impairment
in value be indicated, the carrying value of intangible assets will be adjusted, based on estimates of future discounted cash flows resulting
from the use and ultimate disposition of the asset. ASC 360-10 also requires assets to be disposed of be reported at the lower of the
carrying amount or the fair value less costs to sell.
Long-term Equity Investment
The Company acquires the equity investments
to promote business and strategic objectives. The Company accounts for non-marketable equity and other equity investments for which the
Company does not have control over the investees as:
| ● | Equity
method investments when the Company has the ability to exercise significant influence, but
not control, over the investee. Its proportionate share of the income or loss is recognized
monthly and is recorded in gains (losses) on equity investments. |
| ● | Non-marketable
cost method investments when the equity method does not apply. |
Significant judgment is required to identify
whether an impairment exists in the valuation of the Company’s non-marketable equity investments, and therefore the Company considers
this a critical accounting estimate. Its yearly analysis considers both qualitative and quantitative factors that may have a significant
impact on the investee’s fair value. Qualitative analysis of its investments involves understanding the financial performance and
near-term prospects of the investee, changes in general market conditions in the investee’s industry or geographic area, and the
management and governance structure of the investee. Quantitative assessments of the fair value of its investments are developed using
the market and income approaches. The market approach includes the use of comparable financial metrics of private and public companies
and recent financing rounds. The income approach includes the use of a discounted cash flow model, which requires significant estimates
regarding the investees’ revenue, costs, and discount rates. The Company’s assessment of these factors in determining whether
an impairment exists could change in the future due to new developments or changes in applied assumptions.
Other-Than-Temporary Impairment
The Company’s long-term equity investments
are subject to a periodic impairment review. Impairments affect earnings as follows:
| ● | Marketable
equity securities include the consideration of general market conditions, the duration and
extent to which the fair value is below cost, and our ability and intent to hold the investment
for a sufficient period of time to allow for recovery of value in the foreseeable future.
The Company also considers specific adverse conditions related to the financial health of,
and the business outlook for, the investee, which may include industry and sector performance,
changes in technology, operational and financing cash flow factors, and changes in the investee’s
credit rating. The Company records other-than-temporary impairments on marketable equity
securities and marketable equity method investments in gains (losses) on equity investments. |
| ● | Non-marketable
equity investments based on the Company’s assessment of the severity and duration of
the impairment, and qualitative and quantitative analysis of the operating performance of
the investee; adverse changes in market conditions and the regulatory or economic environment;
changes in operating structure or management of the investee; additional funding requirements;
and the investee’s ability to remain in business. A series of operating losses of an
investee or other factors may indicate that a decrease in value of the investment has occurred
that is other than temporary and that shall be recognized even though the decrease in value
is in excess of what would otherwise be recognized by application of the equity method. A
loss in value of an investment that is other than a temporary decline shall be recognized.
Evidence of a loss in value might include, but would not necessarily be limited to, absence
of an ability to recover the carrying amount of the investment or inability of the investee
to sustain an earnings capacity that would justify the carrying amount of the investment.
The Company records other-than-temporary impairments for non-marketable cost method investments
and equity method investments in gains (losses) on equity investments. Other-than-temporary
impairments of equity investments were $0 and $0 for the years ended December 31, 2023 and
2022, respectively. |
Goodwill
The Company evaluates goodwill for impairment
annually or more frequently when an event occurs or circumstances change that indicate the carrying value may not be recoverable. In
testing goodwill for impairment, the Company may elect to utilize a qualitative assessment to evaluate whether it is more likely than
not that the fair value of a reporting unit is less than its carrying amount. If the qualitative assessment indicates that goodwill impairment
is more likely than not, the Company performs a two-step impairment test. The Company tests goodwill for impairment under the two-step
impairment test by first comparing the book value of net assets to the fair value of the reporting units. If the fair value is determined
to be less than the book value or qualitative factors indicate that it is more likely than not that goodwill is impaired, a second step
is performed to compute the amount of impairment as the difference between the estimated fair value of goodwill and the carrying value.
The Company estimates the fair value of the reporting units using discounted cash flows. Forecasts of future cash flows are based on
our best estimate of future net sales and operating expenses, based primarily on expected category expansion, pricing, market segment
share, and general economic conditions.
The Company completed the required testing
of goodwill for impairment as of December 31, 2023, and determined that goodwill was impaired because of the current financial condition
of the Company and the Company’s inability to generate future operating income without substantial sales volume increases, which
are highly uncertain. Furthermore, the Company anticipates future cash flows indicate that the recoverability of goodwill is not reasonably
assured.
Research and Development Expenses
The Company accounts for the cost of using
licensing rights in research and development cost according to ASC Topic 730-10-25-1. This guidance provides that absent alternative
future uses the acquisition of product rights to be used in research and development activities must be charged to research and development
expenses when incurred.
The Company accounts for R&D costs in
accordance with Accounting Standards Codification (“ASC”) 730, Research and Development (“ASC 730”). Research
and development expenses are charged to expense as incurred unless there is an alternative future use in other research and development
projects or otherwise. Research and development expenses are comprised of costs incurred in performing research and development activities,
including personnel-related costs, facilities-related overhead, and outside contracted services including clinical trial costs, manufacturing
and process development costs for both clinical and preclinical materials, research costs, and other consulting services. Non-refundable
advance payment for goods and services that will be used in future research and development activities are expensed when the activity
has been performed or when the goods have been received rather than when the payment is made. In instances where the Company enters into
agreements with third parties to provide research and development services, costs are expensed as services are performed.
Post-retirement and post-employment benefits
The Company’s subsidiaries in Taiwan
adopted the government mandated defined contribution plan pursuant to the Labor Pension Act (the “Act”) in Taiwan. Such labor
regulations require that the rate of contribution made by an employer to the Labor Pension Fund per month shall not be less than 6% of
the worker’s monthly salaries. Pursuant to the Act, the Company makes monthly contribution equal to 6% of employees’ salaries
to the employees’ pension fund. The Company has no legal obligation for the benefits beyond the contributions made. The total amounts
for such employee benefits, which were expensed as incurred, were $10,314 and $13,031 for the years ended December 31, 2023 and 2022,
respectively. Other than the above, the Company does not provide any other post-retirement or post-employment benefits.
Stock-based Compensation
The Company measures expense associated with
all employee stock-based compensation awards using a fair value method and recognizes such expense in the consolidated financial statements
on a straight-line basis over the requisite service period in accordance with FASB ASC Topic 718 “Compensation-Stock Compensation”.
Total employee stock-based compensation expenses were $0 and $1,241,930 for the years ended December 31, 2023 and 2022, respectively.
The Company accounted for stock-based compensation
to non-employees in accordance with FASB ASC Topic 718 “Compensation-Stock Compensation” and FASB ASC Topic 505-50 “Equity-Based
Payments to Non-Employees” which requires that the cost of services received from non-employees is measured at fair value at the
earlier of the performance commitment date or the date service is completed and recognized over the period the service is provided. Total
non-employee stock-based compensation expenses were $1,635,708 and $5,794,848 for the years ended December 31, 2023 and 2022, respectively.
Beneficial Conversion Feature
From time to time, the Company may issue convertible
notes that may contain an imbedded beneficial conversion feature. A beneficial conversion feature exists on the date a convertible note
is issued when the fair value of the underlying common stock to which the note is convertible into is in excess of the remaining unallocated
proceeds of the note after first considering the allocation of a portion of the note proceeds to the fair value of the warrants, if related
warrants have been granted. The intrinsic value of the beneficial conversion feature is recorded as a debt discount with a corresponding
amount to additional paid in capital. The debt discount is amortized to interest expense over the life of the note using the effective
interest method.
Income Taxes
The Company accounts for income taxes using
the asset and liability approach which allows the recognition and measurement of deferred tax assets to be based upon the likelihood
of realization of tax benefits in future years. Under the asset and liability approach, deferred taxes are provided for the net tax effects
of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used
for income tax purposes. A valuation allowance is provided for deferred tax assets if it is more likely than not these items will expire
before the Company is able to realize their benefits, or future deductibility is uncertain.
Under ASC 740, a tax position is recognized
as a benefit only if it is “more likely than not” that the tax position would be sustained in a tax examination, with a tax
examination being presumed to occur. The evaluation of a tax position is a two-step process. The first step is to determine whether it
is more-likely-than-not that a tax position will be sustained upon examination, including the resolution of any related appeals or litigations
based on the technical merits of that position. The second step is to measure a tax position that meets the more-likely-than-not threshold
to determine the amount of benefits recognized in the financial statements. A tax position is measured at the largest amount of benefit
that is greater than 50 percent likely of being realized upon ultimate settlement. Tax positions that previously failed to meet the more-likely-than-not
recognition threshold should be recognized in the first subsequent period in which the threshold is met. Previously recognized tax positions
that no longer meet the more-likely-than-not criteria should be de-recognized in the first subsequent financial reporting period in which
the threshold is no longer satisfied. Penalties and interest incurred related to underpayment of income tax are classified as income
tax expense in the year incurred. No significant penalty or interest relating to income taxes has been incurred for the years ended December
31, 2023 and 2022. GAAP also provides guidance on de-recognition, classification, interest and penalties, accounting in interim periods,
disclosures and transition.
Valuation of Deferred Tax Assets
A valuation allowance is recorded to reduce
the Company’s deferred tax assets to the amount that is more likely than not to be realized. In assessing the need for the valuation
allowance, management considers, among other things, projections of future taxable income and ongoing prudent and feasible tax planning
strategies. If the Company determines that sufficient negative evidence exists, then it will consider recording a valuation allowance
against a portion or all of the deferred tax assets in that jurisdiction. If, after recording a valuation allowance, the Company’s
projections of future taxable income and other positive evidence considered in evaluating the need for a valuation allowance prove, with
the benefit of hindsight, to be inaccurate, it could prove to be more difficult to support the realization of its deferred tax assets.
As a result, an additional valuation allowance could be required, which would have an adverse impact on its effective income tax rate
and results. Conversely, if, after recording a valuation allowance, the Company determines that sufficient positive evidence exists in
the jurisdiction in which the valuation allowance was recorded, it may reverse a portion or all of the valuation allowance in that jurisdiction.
In such situations, the adjustment made to the deferred tax asset would have a favorable impact on its effective income tax rate and
results in the period such determination was made.
Loss Per Share of Common Stock
The Company calculates net loss per share
in accordance with ASC Topic 260, “Earnings per Share”. Basic loss per share is computed by dividing the net loss by the
weighted average number of common shares outstanding during the period. Diluted loss per share is computed similar to basic loss per
share except that the denominator is increased to include the number of additional common shares that would have been outstanding if
the potential common stock equivalents had been issued and if the additional common shares were dilutive. Diluted earnings per share
excludes all dilutive potential shares if their effect is anti-dilutive.Commitments and Contingencies
The Company has adopted ASC Topic 450 “Contingencies”
subtopic 20, in determining its accruals and disclosures with respect to loss contingencies. Accordingly, estimated losses from loss
contingencies are accrued by a charge to income when information available before financial statements are issued or are available to
be issued indicates that it is probable that an assets had been impaired or a liability had been incurred at the date of the financial
statements and the amount of the loss can be reasonably estimated. Legal expenses associated with the contingency are expensed as incurred.
If a loss contingency is not probable or reasonably estimable, disclosure of the loss contingency is made in the financial statements
when it is at least reasonably possible that a material loss could be incurred.
Foreign-currency Transactions
For the Company’s subsidiaries in Taiwan,
the foreign-currency transactions are recorded in New Taiwan dollars (“NTD”) at the rates of exchange in effect when the
transactions occur. Gains or losses resulting from the application of different foreign exchange rates when cash in foreign currency
is converted into New Taiwan dollars, or when foreign-currency receivables or payables are settled, are credited or charged to income
in the year of conversion or settlement. On the balance sheet dates, the balances of foreign-currency assets and liabilities are restated
at the prevailing exchange rates and the resulting differences are charged to current income except for those foreign currencies denominated
investments in shares of stock where such differences are accounted for as translation adjustments under the Statements of Stockholders’
Equity (Deficit).
Translation Adjustment
The accounts of the Company’s subsidiaries
in Taiwan were maintained, and their financial statements were expressed, in New Taiwan Dollar (“NT$”). Such financial statements
were translated into U.S. Dollars (“$” or “USD”) in accordance ASC 830, “Foreign Currency Matters”,
with the NT$ as the functional currency. According to the Statement, all assets and liabilities are translated at the current exchange
rate, shareholder’s deficit are translated at the historical rates and income statement items are translated at an average exchange
rate for the period. The resulting translation adjustments are reported under other comprehensive income (loss) as a component of shareholders’
equity (deficit).
Recent Accounting Pronouncements
In August 2020, the FASB issued ASU 2020-06,
Debt - Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging-Contracts in Entity’s Own Equity (Subtopic
815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity (“ASU 2020-06”). ASU 2020-06
simplifies the accounting for convertible debt by eliminating the beneficial conversion and cash conversion accounting models. Upon adoption
of ASU 2020-06, convertible debt, unless issued with a substantial premium or an embedded conversion feature that is not clearly and
closely related to the host contract, will no longer be allocated between debt and equity components. This modification will reduce the
issue discount and result in less non-cash interest expense in financial statements. ASU 2020-06 also updates the earnings per share
calculation and requires entities to assume share settlement when the convertible debt can be settled in cash or shares. For contracts
in an entity’s own equity, the type of contracts primarily affected by ASU 2020-06 are freestanding and embedded features that
are accounted for as derivatives under the current guidance due to a failure to meet the settlement assessment by removing the requirements
to (i) consider whether the contract would be settled in registered shares, (ii) consider whether collateral is required to be posted,
and (iii) assess shareholder rights. ASU 2020-06 is effective for fiscal years beginning after December 15, 2023. Early adoption is permitted,
but no earlier than fiscal years beginning after December 15, 2020, and only if adopted as of the beginning of such fiscal year. The
Company is currently evaluating the impact that the standard will have on its consolidated financial statements.
In May 2021, the FASB issued ASU 2021-04,
Earnings Per Share (Topic 260), Debt - Modifications and Extinguishments (Subtopic 470-50), Compensation - Stock Compensation (Topic
718), and Derivatives and Hedging - Contracts in Entity’s Own Equity (Subtopic 815-40): Issuer’s Accounting for Certain Modifications
or Exchanges of Freestanding Equity-Classified Written Call Options (“ASU 2021-04”). ASU 2021-04 provides guidance as to
how an issuer should account for a modification of the terms or conditions or an exchange of a freestanding equity-classified written
call option (i.e., a warrant) that remains classified after modification or exchange as an exchange of the original instrument for a
new instrument. An issuer should measure the effect of a modification or exchange as the difference between the fair value of the modified
or exchanged warrant and the fair value of that warrant immediately before modification or exchange and then apply a recognition model
that comprises four categories of transactions and the corresponding accounting treatment for each category (equity issuance, debt origination,
debt modification, and modifications unrelated to equity issuance and debt origination or modification). ASU 2021-04 is effective for
all entities for fiscal years beginning after December 15, 2021, including interim periods within those fiscal years. An entity should
apply the guidance provided in ASU 2021-04 prospectively to modifications or exchanges occurring on or after the effective date. Early
adoption is permitted for all entities, including adoption in an interim period. If an entity elects to early adopt ASU 2021-04 in an
interim period, the guidance should be applied as of the beginning of the fiscal year that includes that interim period. The Company
is currently evaluating the impact that the standard will have on its consolidated financial statements.
Estimates and Assumptions
In preparing our consolidated financial statements,
we use estimates and assumptions that affect the reported amounts and disclosures. Our estimates are often based on complex judgments,
probabilities and assumptions that we believe to be reasonable, but that are inherently uncertain and unpredictable. We are also subject
to other risks and uncertainties that may cause actual results to differ from estimated amounts.
Results of Operations - Year Ended December
31, 2023 Compared to Year Ended December 31, 2022.
Revenues. We generated $152,430
and $969,783 in revenues for the years ended December 31, 2023 and 2022, respectively. The decrease of $817,353, or approximately 84%,
was primarily caused by the completion of ongoing projects and waiting for new approval.
Operating Expenses. Our operating
expenses were $8,066,902 in the year ended December 31, 2023, compared to $15,797,780 in December 31, 2022. Such decrease in operating
expenses was mainly attributable to the decreased stock-based compensation and selling, general and administrative expenses, by $6,100,337,
and decreasing research and development expenses of $1,630,541.
Other Income (expense). The other expense was $2,437,773
in the year ending December 31, 2023, compared to other income of $400,184 on December 31, 2022. The change was principally caused by
the increase in interest expense, mainly from the convertible notes payable, while being offset by the increase in foreign exchange for
the year ended December 31, 2023, loss on investment in equity securities and decrease in impairment loss and investment loss for the
year ended December 31, 2023.
Interest income (expense), net, was $(2,307,859)
for the year ended December 31, 2023, compared to $(106,151) for the year ended December 31, 2022. The increase of $(2,201,708), or approximately
2,074%, was primarily due to the increase in interest expense due to recognition of interest expense for the converted notes for proper
accounting purpose.
Net Loss. The net loss was $10,910,288
for the year ended December 31, 2023, compared to $16,312,374 for the year ended December 31, 2022. The Company reduce its net loss by
$5,061,086 or approximately 31% during the year ended December 31, 2023 from 2022, through more effective usage of funding and discontinuing
certain consulting services.
Liquidity and Capital Resources
Working Capital
| | |
As of
December 31,
2023 | | |
As of
December 31,
2022 | |
| | |
| | |
| |
| Current Assets | |
$ | 1,656,709 | | |
$ | 2,987,247 | |
| Current Liabilities | |
$ | 5,932,490 | | |
$ | 5,543,628 | |
| Working (Deficit) Capital | |
$ | (4,275,781 | ) | |
$ | (2,556,381 | ) |
Cash Flow from Operating Activities
During the years ended December 31, 2023 and
2022, the net cash used in operating activities were ($4,235,845) and $7,398,391, respectively. The decrease in the amount of $3,162,546
was primarily due to the increased account receivables, loss on investment in equity securities, loss and sales of treasury stock, accrued
expenses and other current liabilities, partially offset by the decreased net loss, gain on sales of investment in equity securities,
due from related parties, prepaid expenses, impairment loss, and stock-based compensation; and by the decrease of deferred tax during
the year ended December 31, 2023.
Cash Flow from Investing Activities
During the years ended December 31, 2023 and
2022, the net cash used in investing activities were $360,186 and $1,721,684, respectively. The decrease in the amount of $1,361,498
was primarily due to the decrease in prepayment for equity investment and purchase of equipment, while being offset by the increase in
prepayment for long-term investments during the year ended December 31, 2023.
Cash Flow from Financing Activities
During the years ended December 31, 2023 and
2022, the net cash provided by financing activities were $3,918,960 and $4,013,925, respectively. The net cash provided by financing
activities decreased by $94,965, due to the increase in proceeds from convertible notes and issuance of warrants, partially offset by
the decrease in issuance of common stock, as well as decrease in proceeds from short-term loans, and repayment of short-term notes during
the year ended December 31, 2023.
Off-Balance Sheet Arrangements
As of December 31, 2023, we did not have any
off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes
in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures, or capital resources that is material
to investors.
BUSINESS
Industry Overview
The biotechnology industry focuses on developing
breakthrough products and technologies to combat various types of diseases through efficient industrial manufacturing process. Biotechnology
is an important business sector in the world’s economies and plays a key role in human health. Companies engaged in biotechnology
generally require large amounts of capital investment for their research & development activities and it may take up to tens of years
to develop and commercialize a new drug or a new medical device. ABVC (“we” or the “Company”) is an early stage
biotechnology company with a pipeline of seven new drugs and one medical device under development, all of which are licensed from related
parties of the Company.
Our Mission
We devote our resources to building a sophisticated
biotech company and becoming a pioneer in the biopharmaceutical industry. Dr. Uttam Patil, our Chief Executive Officer, and Dr. Tsung-Shann
Jiang, the founder and majority shareholder of the Company, understand the challenges and opportunities of the biotech industry and intend
to provide therapeutic solutions to significant unmet medical needs and to improve health and quality of human life by developing innovative
botanical drugs to treat central nervous system (“CNS”) and oncology/ hematology diseases.
Business Overview
As of the date hereof, the Company’s minimal
revenue has come from the sale of CDMO services through BioKey. However, the Company’s focus is on developing a pipeline of products
by carefully tracking new medical discoveries or medical device technologies in research institutions in the Asia-Pacific region. Pre-clinical,
disease animal model and Phase I safety studies are examined closely by the Company’s scientists and other specialists known to
the Company to identify drugs or medical devices that it believes demonstrate efficacy and safety based on the Company’s internal
qualifications. Once a drug or medical device is shown to be a good candidate for further development and ultimately commercialization,
ABVC licenses the drug or medical device from the original researchers and introduces the drug or medical device clinical trial plan
to highly respected principal investigators in the United States, Australia and Taiwan. In almost all cases, ABVC has found that research
institutions in each of those countries are eager to work with the Company to move forward with Phase II clinical trials.
Institutions that have or are now conducting
phase II clinical trials in partnership with ABVC include:
| |
● |
Drug:
ABV-1504, Major Depressive Disorder (MDD), Phase II completed. NCE drug Principal Investigators: Charles DeBattista M.D. and Alan
F. Schatzberg, MD, Stanford University Medical Center, Cheng-Ta Li, MD, Ph.D – Taipei Veterans General Hospital |
| |
● |
Drug:
ABV-1505, Adult Attention-Deficit Hyperactivity Disorder (ADHD), Phase II Part 1 completed. Principal Investigators: Keith McBurnett,
Ph.D. and Linda Pfiffner, Ph.D., University of California San Francisco (UCSF), School of Medicine. Phase II, Part 2 clinical study
sites includes UCSF and 5 locations in Taiwan.The Principal Investigators are Keith McBurnett, Ph.D. and Linda Pfiffner, Ph.D., University
of California San Francisco (UCSF), School of Medicine; Susan Shur-Fen Gau, M.D., National Taiwan University Hospital; Xinzhang Ni,
M.D. Linkou Chang Gung Memorial Hospital; Wenjun Xhou, M.D., Kaohsiung Chang Gung Memorial Hospital; Ton-Ping Su, M.D., Cheng Hsin
General Hospital, Cheng-Ta Li, M.D., Taipei Veterans General Hospital. The Phase II, Part 2 began in the 1st quarter of
2022 at the 5 Taiwan sites. The UCSF site joined the study in the 2nd quarter of 2023. The subjects enrolled in the study
has reached the number for interim analysis in 2023 December, and the interim analysis of the study is in progress. |
| |
● |
Drug:
ABV-1601, Major Depression in Cancer Patients, Phase I/II, NCE drug Principal Investigator: Scott Irwin, MD, Ph.D. – Cedars
Sinai Medical Center (CSMC). The Phase I clinical study will be initiated in the 1st quarter of 2024. |
| |
● |
Medical
Device: ABV-1701, Vitargus® in vitrectomy surgery, Phase II Study has been initiated in Australia and Thailand, Principal Investigator:
Duangnate Rojanaporn, M.D., Ramathibodi Hospital; Thuss Sanguansak, M.D., Srinagarind Hospital of the two Thailand Sites and Professor/Dr.
Matthew Simunovic, Sydney Eye Hospital; Dr. Elvis Ojaimi, East Melbourne Eye Group & East Melbourne Retina. The Phase II study
started in the 2nd quarter of 2023, and the company is working on improvements to the Vitargus Product through the new batch of investigational
product. |
The following trials are expected to begin
in the third quarter of 2024:
| |
● |
Drug:
ABV-1519, Non-Small Cell Lung Cancer treatment, Phase I/II Study in Taiwan, Principal Investigator: Dr. Yung-Hung Luo, M.D., Taipei
Veterans General Hospital (TVGH) |
| |
● |
Drug:
ABV-1703, Advanced Inoperable or Metastatic Pancreatic Cancer, Phase II, Principal Investigator: Andrew E. Hendifar, MD – Cedars
Sinai Medical Center (CSMC) |
Upon completing a Phase II trial, ABVC will
seek a partner, typically a large pharmaceutical company, to complete a Phase III study and commercialize the drug or medical device
upon approval by the US FDA, Taiwan TFDA and other country regulatory authorities.
GMP Manufacturing
ABVC owns a certified GMP manufacturing facility,
through BioKey, that is qualified to deliver small quantities of drugs for use by its clients in clinical trials from Phase I to Phase
III. The GMP facility can manufacture direct API or blend fill-in capsules, manual and automated encapsulation, wet granulation or tray
drying process, tablet compression and coating process, packaging solid dosage forms for ANDA and IND submission.
The BioKey facility consists of a GMP suite,
product development area, analytical laboratory, food processing area, caged GMP storage area, receiving area and two warehouses. The
facility was remodeled in December 2008 and received its first drug manufacturing license in June 2009. ABVC’s current drug manufacturing
license allows it to manufacture drug products under IND for human clinical trials until the expiration of the license on December 2,
2024.
In 2022, BioKey began manufacturing a dietary
supplement based on the maitake mushroom. The mushrooms, supplied by Shogun Maitake Canada, Co. Ltd., are grown in a controlled temperature
and humid environment free of pesticides and chemicals. Initially, sales of the new supplement in the US and Canada will be targeted
to high end grocery stores and worldwide via online distribution. While there are many mushroom-based supplements currently available
to customers, BioKey believes its new line has a significant competitive advantage since the purity and consistency of the mushrooms
themselves exceeds any maitake mushrooms currently available and the extraction process employed by BioKey delivers a particularly strong
dose. The maitake mushroom is rich in bioactive polysaccharides, especially beta-glucans. These polysaccharides have well-documented
immune-protecting and antitumor properties. BioKey has developed both a tablet and a liquid version of the supplement. GMP manufacturing
of bulk quantities Maitake mushroom tablets and Maitake mushroom drinks were completed in 2 and 1 batches respectively for commercial
launches in Taiwan and Canada in 2022.
Beta-glucans in maitake mushrooms has been shown
to reduce cholesterol, resulting in improved artery functionality and overall better cardiovascular health that lowers the risk of heart
disease. Further, studies have shown that the beta-glucans in maitake mushroom have the effect of strengthening the immune system1.
In a trial of postmenopausal breast cancer patients, oral administration of a maitake extract was shown to have immunomodulatory effects.
In a different trial done at Memorial Sloan Kettering Cancer Center, maitake extracts were shown to enhance neutrophil and monocyte function
in patients with myelodysplastic syndrome. It boosts production of lymphokines (protein mediators) and interleukins (secreted proteins)
resulting in improved immune response. Further, beta-glucans, has been shown in clinical trials to lower blood glucose levels thereby
helping to activate insulin receptors, while reducing insulin resistance in diabetes management.
BioKey has entered into a three-year distribution
agreement with Define Biotech Co. Ltd., a Taiwan-based pharmaceutical marketing company that focuses on sales of drugs, dietary supplements
and medical products in the Asia-Pacific region. The agreement grants Define Biotech the exclusive right to distribute this new dietary
supplement in China and Taiwan in exchange for the commitment to purchase $3.0 million worth of the new product over the three-year period.
NASDAQ Listing
On August 5, 2021, we closed a public offering
(the “Offering”) of 1,100,000 units (the “Units”), with each Unit consisting of one share of our common stock
(the “Common Stock”), one Series A warrant (the “Series A Warrants”) to purchase one share of common stock at
an exercise price equal to $6.30 per share, exercisable until the fifth anniversary of the issuance date, and one Series B warrant (the
“Series B Warrants,” and together with the Series A Warrants, the “Public Warrants”) to purchase one share of
common stock at an exercise price equal to $10.00 per share, exercisable until the fifth anniversary of the issuance date; the exercise
price of the Public Warrants are subject to certain adjustment and cashless exercise provisions as described therein. The Company completed
the Offering pursuant to its registration statement on Form S-1 (File No. 333-255112), originally filed with the Securities and Exchange
Commission (the “SEC”) on April 8, 2021 (as amended, the “Original Registration Statement”), that the SEC declared
effective on August 2, 2021 and the registration statement on Form S-1 (File No. 333-258404) that was filed and automatically effective
on August 4, 2021 (the “S-1MEF,” together with the Original Registration Statement, the “Registration Statement”).
The Units were priced at $6.25 per Unit, before underwriting discounts and offering expenses, resulting in gross proceeds of $6,875,000.
The Offering was conducted on a firm commitment basis. The Common Stock was approved for listing on The Nasdaq Capital Market and commenced
trading under the ticker symbol “ABVC” on August 3, 2021.
On August 19, 2022, we received a deficiency
letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq Stock Market LLC (“Nasdaq”)
notifying us that, for the last 30 consecutive business days, the closing bid price for our common stock was below the minimum $1.00
per share required for continued listing on The Nasdaq Capital Market pursuant to Nasdaq Listing Rule 5550(a)(2) (“Rule 5550(a)(2)”).
In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were initially given until February 14, 2023 to regain compliance with Rule
5550(a)(2). Since we did not regain compliance by such date, we requested and received an additional 180 days, until August 14, 2023,
to comply with Rule 5550(a)(2).
On May 24, 2023, we received a deficiency
letter from the Nasdaq Listing Qualifications Department (the “Staff”) of the Nasdaq Stock Market LLC (“Nasdaq”)
notifying the Company that it is not currently in compliance with the minimum stockholders’ equity requirement, or the alternatives
of market value of listed securities or net income from continuing operations, for continued listing on the Nasdaq Capital Market. Nasdaq
Listing Rule 5550(b)(1) requires listed companies to maintain stockholders’ equity of at least $2,500,000, and the Company’s
stockholders’ equity was $1,734,507 as of March 31, 2023. In accordance with Nasdaq rules, the Company had 45 calendar days, or
until July 10, 2023, to submit a plan to regain compliance. After submitting a plan to regain compliance, on July 10, 2023,Nasdaq granted
the Company an extension until August 30, 20203, to comply with Listing Rule 5550(b)(1). On July 31, 2023, the Company issued 300,000
shares of Common Stock and 200,000 pre-funded warrants, at an exercise price of $0.01 per share, in a registered direct offering. Pursuant
to this transaction, the stockholders’ equity was increased by $1.75M. On August 1, 2023, $500,000 of Notes were converted at $3.50
per share and the holder received 142,857 shares of Common Stock. As a result of this conversion, the stockholders’ equity was
increased by $0.5M. Additionally, on August 14, 2023, the Company entered into a cooperation agreement with Zhonghui United Technology
(Chengdu) Group Co., Ltd., pursuant to which the Company acquired a 20% ownership of certain property and a parcel of the land owned
by Zhonghui in exchange for an aggregate of 370,000 shares of Common Stock. Accordingly, stockholders’ equity increased by $7.4M.
On February 23, 2023, the Company entered into a securities purchase agreement with Lind, pursuant to which the Company issued Lind a
secured, convertible note in the principal amount of $3,704,167 (the “Lind Offering”), for a purchase price of $3,175,000
(the “Lind Note”), that is convertible into shares of Common Stock at an initial conversion price of $1.05 per share, subject
to adjustment. On August 24, 2023, the Company started repaying Lind the monthly installments due under the Lind Notes; $308,000 was
repaid via the issuance of 176,678 shares of Common Stock (the “Monthly Shares”) at the Redemption Share Price (as defined
in the Lind Note) of $1.698 per share. Pursuant to the terms of the Lind Note, Lind increased the amount of the next monthly payment
to one million dollars, such that as of September and together with the Monthly Shares, the Company repaid Lind a total of $1M by September
2023. As a result, the stockholders’ equity increased by an additional $1M. As a result of the four transactions referenced above,
the Company’ estimated that its stockholders’ equity would increase by approximately $10.65M. On September 6, 2023, Nasdaq
issued a letter that the Company is in compliance with Rule 5550(b)(1), but noted that if at the time of the Company’s next periodic
report the Company does not evidence compliance, it may be subject to delisting.
Name Change and Cusip Number
The Company’s shareholders approved
an amendment to the Company’s Articles of Incorporation to change the Company’s corporate name to “ABVC BioPharma,
Inc.” and approved and adopted the Certificate of Amendment to affect same at the 2020 annual meeting of shareholders (the “Annual
Meeting”). Nevada’s Secretary of State approved the name change on March 8, 2021, and FINRA processed our request for
such name change on April 30, 2021. The new name was effective on May 3, 2021. Stock certificates issued before the name change remain
valid and stockholders are not required to submit their stock certificates for exchange as a result of the name change. New stock
certificates issued by the Company after the name change will be printed with the Company’s new name, ABVC BioPharma, Inc.; existing
stock certificates remain valid.
The Company’s cusip number is 0091F106. The Company’s
stock symbol remains ABVC.
Our Pipeline
| I. |
Central Nervous System |
| 1. | ABV-1504
to treat Major Depressive Disorder (“MDD”) |
We are developing and researching
ABV-1504, a botanical reuptake inhibitor that targets norepinephrine. Prior to clinical trials, we conducted radioligand-binding assay
tests on ABV-1504. Radioligand-binding assays are used to characterize the binding effects of a drug to its target receptor. In the case
of ABV-1504, the receptors of radioligand-binding assays are norepinephrine, dopamine and serotonin. The radioligand-binding assay test
on norepinephrine was conducted from May 3 to May 8, 2007 and the radioligand-binding assay test on dopamine and serotonin was administered
from November 26 to December 5, 2007. The result of radioligand-binding assay to norepinephrine of ABV-1504was 2.102 μg/ml of IC50,
which indicated ABV-1504’s high inhibitory efficiency on norepinephrine. The results of radioligand-binding assay to dopamine and
serotonin were not as good as to norepinephrine, which indicated lower inhibitory efficiency. Because research has shown that norepinephrine
inhibitors can alleviate the level of depression, our research team saw ABV-1504’s potential to treat depression and decided to
commence the clinical trial process of ABV-1504.
In 2013, ABVC successfully completed
the Phase I clinical trial of ABV-1504. The primary objective of the Phase I study was to assess the safety profile of ABV-1504. The
safety endpoint was assessed based on the results of physical examinations, vital signs, laboratory data, electrocardiograms (“ECG”),
Columbia-Suicide Severity Rating Scale evaluation and a number of adverse events during the study period. We began recruiting healthy
people as subjects for the Phase I trial in Taiwan on October 30, 2012. For the Phase I trial, we screened 85 healthy volunteers at the
Taipei Veterans General Hospital and eventually enrolled 30 people as trial subjects. We divided the subjects into four cohort groups
and administered ABV-1504oral capsules of 380 mg, 1140 mg, 2280 mg, and 3800 mg to the subjects in each cohort group, respectively. BioLite
visited the first subject the first time on November 13, 2012 and the last subject the last time on July 5, 2013. During the said period,
no subject had a serious adverse event nor discontinued the trial due to any adverse events. ABVC did not observe any clinically significant
findings in physical examinations, vital signs, electrocardiogram, laboratory measurements, and C-SSRS throughout the treatment period.
However, ABVC observed the following mild adverse events: two subjects with flatulence and one subject with constipation in the single-dose
380mg cohort of seven subjects; one subject with somnolence and one subject with stomatitis ulcer in the single-dose 2,280 mg cohort.
Comparatively, two subjects with somnolence and one subject with stomatitis ulcer were observed in the placebo group of seven subjects.
ABVC did not observe any suicidal ideation or behavior throughout the trial period. ABV-1504’s Phase I clinical trial results reflected
that the oral administration of ABV-1504 to healthy volunteers was safe and well-tolerated at the dose levels of from 380 mg to 3,800
mg.
ABVC received an IND approval to proceed
with the Phase II clinical trial of ABV-1504 from the F.D.A. in March 2014 and an IND approval of its Phase II trial from the Taiwan
F.D.A. in June 2014. For the Phase II trial, BioLite administered oral capsules to 72 MDD patients (the trial subjects) in a randomized,
double-blind study with a placebo control group to assess ABV-1504’s efficacy and safety profile, primarily in accordance with
the Montgomery-Åsberg Depression Rating Scale (“MADRS”). ABVC via BioLite began recruiting Phase II subjects in March
2015 at the following study sites, Taipei Veterans General Hospital, Linkou Chang Gung Memorial Hospital, Taipei City Hospital-Songde
Branch, Tri-Service General Hospital, Wan Fang Hospital and started recruiting MDD patients at Stanford Depression Research Clinic. The
first five sites are in Taiwan and the last one is in the United States. The primary endpoint of the Phase II trial is to see changes
of the subjects’ MADRS total scores from the baseline scores of the placebo subjects within the first six weeks. The secondary
objectives of the Phase II trial are to evaluate the efficacy and safety profile of ABV-1504 on other rating scales with secondary endpoints
of (i) demonstrating changes in MADRS total scores from baseline scores within the second to seventh weeks and (ii) showing changes in
the total scores on Hamilton Rating Scale for Depression (HAM-D-17), Hamilton Rating Scale for Anxiety (HAM-A), Depression and Somatic
Symptoms Scale (DSSS), Clinical Global Impression Scale (CGI) from the baseline scores in the second, fourth, sixth and seventh week.
ABVC plans to measure the percentages of partial responders (subjects with a 25% to 50% decrease of total MADRS scores from the baseline
score) and responders (subjects with 50% or more decrease of total MADRS scores from the baseline score) by the second, fourth, sixth
and seventh week. Additionally, ABVC intends to monitor the subjects’ performance in accordance with the Safety Assessments and
Columbia-Suicide Severity Rating Scale from the screening stage to each subject’s last visit as well as to analyze the differences
in the mean changes of MADRS, HAM-D-17, HAM-A, DSSS, CGI and Columbia-Suicide Severity Rating Scale scores of the subjects administered
with ABV-1504 and the placebo group in the second, fourth, sixth and seventh week.
On May 23, 2019, the Company announced
the Phase II clinical study results of ABV-1504. The clinical study results showed that PDC-1421, the active pharmaceutical ingredient
of ABV-1504, met the pre-specified primary endpoint of the Phase II clinical trial and significantly improved the symptoms of MDD. The
Phase II clinical study was a randomized, double-blind, placebo-controlled, multi-center trial, in which sixty (60) adult patients with
confirmed moderate to severe MDD were treated with PDC-1421 in either low dose (380 mg) or high dose (2 x 380 mg) compared with placebo
administration, three times a day for six weeks. PDC-1421 high dose (2 x 380 mg) met the pre-specified primary endpoint by demonstrating
a highly significant 13.2-point reduction in the Montgomery-Åsberg Depression Rating Scale (MADRS) total score by Intention-To-Treat
(ITT) analysis, averaged over the 6-week treatment period (overall treatment effect) from baseline, as compared to 9.2-point reduction
of the placebo group. By Per-Protocol (PP) analysis, PDC-1421 showed a dose dependent efficacy toward MDD in which high dose (2 x 380
mg) gave 13.4-point reduction in MADRS total score from baseline and low dose (380 mg) gave 10.4-point reduction as compared to a 8.6-point
in the placebo group. Based on the trial results as set forth above, the Company has decided to use the high dose formula for ABV-1504’s
Phase III clinical trial.
| |
2. |
ABV-1505 to treat Attention
Deficit Hyperactivity Disorder (“ADHD”) |
We developed the ADHD indication from
the same API of ABV-1504. Also, ABV-1505 shares similar pharmaceutical mechanism of action as ABV-1504 in as much as ABV-1505 shows the
potential of increasing the level of norepinephrine in the human’s nervous system by inhibiting its reabsorption. Because of ABV-1505’s
sufficient similarity with ABV-1504, in January 2016 the FDA approved our IND application to conduct ABV-1505’s Phase II clinical
trial based on its preclinical research and the Phase I trial results of ABV-1504.
For the ADHD Phase II trial, ABVC
plans to recruit a maximum of 105 ADHD patients as trial subjects in the United States and Taiwan, to whom ABVC intends to administer
ABV-1505 oral capsules. ABVC has designed a randomized, double-blind dose escalation study with a placebo-controlled group to assess
the efficacy and safety profile of ABV-1505, primarily against the ADHD Rating Scale-IV (“ADHD-RS-IV”). The primary endpoint
of the Phase II trial is a 40% or higher improvement on the ADHD-RS-IV from the respective baseline scores within a period of up to eight
weeks. The secondary objective is to determine the efficacy and safety profile of ABV-1505 on other rating scales with secondary endpoints
of (i) improvements of the total ADHD symptom scores from the respective baseline scores on the Conners’ Adult ADHD Rating Scale-Self
Report: Short Version (“CAARS-S:S”) 18-Item for a treatment period of eight weeks at maximum; and (ii) achievement of scores
of two or lower on both the Clinical Global Impression-ADHD- Severity (“CGI-ADHD-S”) and Clinical Global Impression-ADHD-Improvement
(“CGI-ADHD-I”) from the subjects’ respective baseline scores. The University of California San Francisco (“UCSF”)
initiated the Phase II, Part 1 clinical trial entitled “A Phase II Tolerability and Efficacy Study of PDC-1421 Treatment in Adult
Patients with Attention-Deficit Hyperactivity Disorder (ADHD). Part I, on January 14, 2020. The Part 1 trial is a single center, open
label, dose escalation evaluation with two dosage levels in six subjects. Six subjects were initially evaluated for safety and efficacy
assessments at low-dose (1 capsule of PDC-1421, three times a day (TID)) for 28 days. A safety checkpoint was evaluated at day-28 for
entering the high-dose (2 capsules TID). The subjects who passed the checkpoint were evaluated for safety and efficacy assessments at
high-dose (2 capsules of PDC-1421 TID) for 28 days. On July 15, 2020, the last patient last visit (LPLV) marked the final step toward
the completion of the ABV-1505 Phase II Part I clinical trial for the treatment of adult ADHD. On October 24, 2020, a full clinical study
report (CSR) of ABV-1505 Phase II Part I clinical trial was issued. The study results showed that the PDC-1421 Capsule was safe, well
tolerated and efficacious during its treatment and the follow-up period with six adult patients. For the primary endpoints, the percentages
of improvement in ADHD-RS-IV score from baseline to 8 weeks treatment were 83.3% (N=5) in the ITT population and 80.0% (N=4) in the PP
population. Both low and high doses of PDC-1421 Capsule met the primary end points by passing the required 40% population in ADHD-RS-IV
test scores. Overall, the results from this study, which demonstrate the therapeutic value of PDC-1421, support further Phase II Part
II clinical development of ABV-1505 for the treatment of adult ADHD.
The Phase II Part II study with
its clinical protocol entitled “A Phase II Tolerability and Efficacy Study of PDC-1421 Treatment in Adult Patients with Attention-Deficit
Hyperactivity Disorder (ADHD), Part II” is a randomized, double-blind, placebo-controlled, parallel three-groups with a maximum
99 subjects to be enrolled. This study was started at five Taiwan medical centers beginning in April 2022. The University of California,
San Francisco site was initiated in the 2nd quarter of 2023. The subjects enrolled in the study has reached the number for
interim analysis (69 subjects) in 2023 December, and the interim analysis of the study is now in progress.
| |
3. |
ABV-1601 to treat Depression
in Cancer Patients |
We developed a treatment for depression
in cancer patient from the same active pharmaceutical ingredients as ABV-1504. ABV-1601 shares similar pharmaceutical mechanisms of action
as ABV-1504 in that ABV-1601 shows the potential of increasing the level of norepinephrine in the human nervous system by inhibiting
its reabsorption. Due to ABV-1601’s similarity with ABV-1504, the FDA approved our ABV-1601-001 clinical protocol under the same
IND as for ABV-1504 (IND 112567) in December 2018.
For the Phase II trial of ABV-1601,
ABVC plans to recruit a maximum number of 54 cancer patients with depression, to whom ABVC intends to administer ABV-1601 oral capsules.
ABVC is engaging the Principal Investigator at Cedars-Sinai Medical Center in the U.S. which designed a randomized, double-blind dose
escalation study with a comparator-controlled group to assess the efficacy and safety profile of ABV-1601, primarily against Montgomery-Åsberg
Depression Rating Scale (MADRS) total score. The primary endpoint of the Phase II trial is a change in MADRS, Hospital Anxiety and Depression
Scale (HADS), subscales (HADS-A and HADS-D), and Clinical Global Impression Scale (CGI) total scores from baseline in patients taking
PDC-1421 compared to the comparator. As of the date hereof, the Part I of Phase II clinical protocol, which is an open trial, has been
approved by Cedars-Sinai Medical Center IRB Committee. This study will be initiated on March 31, 2023.
| |
1. |
ABV-1702 to
treat Myelodysplastic Syndrome (“MDS”) |
ABVC started the preparation for ABV-1702’s
Phase II clinical trials after receiving its IND approval from the FDA in July 2016. ABVC plans to recruit fifty-two subjects in the
United States who are diagnosed with either IPSS int-1, IPSS int-2 or high risk MDS or CMML and may take azacitidine as part of the subjects’
prescription. Azacitidine is an FDA-approved drug used to treat MDS. ABVC intends to administer ABV-1702 in the oral liquid form along
with azacitidine. The Phase II trial is divided into two parts, where Part 1 is to determine the safety and recommended dose level (“RDL”)
of ABV-1702 in combination with azacitidine and Part 2 is to determine whether ABV-1702 under the established RDL reduces bactericidal
and fungicidal infection in the subjects’ respiratory systems. The primary endpoint of Part 1 Phase II trial is to assess the safety
and RDL profile of ABV-1702 administered with azacitidine by measuring ABV-1702’s prohibited toxicity. The secondary endpoints
of Phase II Part 1 are to determine the safety, time-to-first infection after first dose (Day 1) of the first azacitidine treatment cycle,
reduction in treatment requirements and duration of infections, enhancement of immune responses, improvements of response rates, progression,
and survival rates of the subjects under such ABV-1702 - azacitidine combination treatment. The primary endpoint of Part 2 of Phase II
is to determine whether ABV-1702 under the established RDL reduces bactericidal and fungicidal infection risks in the subjects’
respiratory systems in combination with azacitidine as compared to the control group with incidence of infections and incidence/frequency
of inpatient hospitalization due to infections. The secondary endpoints of Part 2 of Phase II are to determine the safety, time-to-first
infection after first dose (Day 1) of the first azacitidine treatment cycle, reduction in required dosage and duration of infection,
enhancement of immune responses, improvement of response rate, progression, and survival rates of the subjects under the trial conditions.
In April 2016, BioLite submitted a letter to the FDA in response to its queries with additional information about the proposed Phase
II trial.
The Company expects to begin Phase
II clinical trials of ABV-1702 in the fourth quarter of 2024 and is actively looking for qualified principal investigators and an appropriate
site for the study and therefore the timing cannot be guaranteed.
| |
2. |
ABV-1703 to
treat Pancreatic Cancer |
ABVC developed a new indication for
Pancreatic Cancer from Maitake Extract, which is named as ABV-1703 and out licensed it to Rgene for the preparation of its IND application
with the FDA. On August 25, 2017, ABV-1703’s Phase II trial was approved by FDA. Pursuant to the ABVC-Rgene Co-development Agreement,
ABVC is responsible for coordinating and conducting the clinical trials of ABV-1703 globally and Rgene is responsible for preparing the
related FDA applications. As of the date hereof, we are engaging Cedars-Sinai Medical Center in the U.S. to conduct the Phase II clinical
trial and plan to initiate the Phase II trial in the third quarter of 2023. We plan to submit ABV-1703’s Phase II clinical trial
IND to the Taiwan FDA after we commence the clinical trials in the United States.
| |
3. |
ABV- 1501 Triple Negative
Breast Cancer - Combination therapy for Triple Negative Breast Cancer (“TNBC”) |
| |
● |
ABV-
1501 is developed from BLI-1401-2 whose active pharmaceutical ingredient is Yukiguni Maitake Extract 404. Memorial Sloan Kettering
Cancer Center (“MSKCC”) conducted the Phase I clinical trial of a polysaccharide extract from Grifola frondosa (Maitake
mushroom), which is very similar to Yukiguni Maitake Extract 404. The Phase I trial focused on Grifola frondosa extract’s immunological
effects on breast cancer patients. The results of the Phase I trial showed that oral administration of a polysaccharide extract from
Maitake mushroom is associated with both immunologically stimulatory and inhibitory measurable effects in peripheral blood. |
| |
● |
Our ABV-1501 Investigational
New Drug (“IND”) application to the US FDA for the Phase II clinical trials referencing the MSKCC maitake research resulted
in a Phase II IND approval in March of 2016 by the U.S. FDA. |
| |
|
|
| |
● |
The collaboration
with BHK to file clinical trial application to the Taiwan FDA (“TFDA”) for conducting this combination therapy trial
in Taiwan was temporarily put on hold due to the lack of funding. |
Our Collaborative Agreements
| I. |
ABV-1701 Vitreous Substitute
for Vitrectomy and Collaboration Agreement with BioFirst |
On July 24, 2017, BriVision, one of
our wholly-owned subsidiaries entered into a collaboration agreement (the “BioFirst Agreement”) with BioFirst, pursuant to
which BioFirst granted BriVision the global license to co-develop BFC-1401 Vitreous Substitute for Vitrectomy (“BFC-1401”)
for medical purposes. BioFirst is a related party to the Company because BioFirst and YuanGene Corporation (“YuanGene”),
the Company’s controlling shareholder, are under common control, being both controlled by the controlling beneficiary shareholder
of YuanGene.
According to the BioFirst Agreement,
we are to co-develop and commercialize BFC-1401 or ABV-1701 with BioFirst and are obligated to pay BioFirst $3,000,000 (the “Total
Payment”) in cash or common stock of BriVision on or before September 30, 2018 in two installments. An upfront payment of $300,000,
representing 10% of the Total Payment due under the Collaboration Agreement, was to be paid upon execution of the BioFirst Agreement.
BriVision is entitled to receive 50% of the future net licensing income or net sales profit when ABV-1701 is sublicensed or commercialized.
On June 30, 2019, the Company and BioFirst entered into a Stock Purchase Agreement (the “Purchase Agreement”), pursuant to
which the Company will issue 428,571 shares of the Company’s common stock to BioFirst in consideration for $3,000,000 owed by the
Company to BioFirst in connection with the BioFirst Collaborative Agreement. For more information about the BioFirst Agreement and Purchase
Agreement, please refer to the current reports on Form 8-K filed on July 24, 2017 and July 12, 2019.
On November 7, 2016, the application
of Phase I clinical trial prepared and submitted by BioFirst was approved by the Human Research Ethics Committee, Australia (“HREC”),
and on November 14, 2016, it was approved by the Therapeutic Goods Administration, Australia (“TGA”).
We successfully finished the Phase
I clinical trial of ABV-1701 at Sydney Retina Clinic and Day Surgery, a clinic located in Sydney, Australia. This was the only site for
this Phase I clinical trial. The trial started on November 17, 2016, and was completed with positive results in July 2018. The Protocol
Title is “A Phase I, single center, safety and tolerability study of Vitargus in the treatment of Retinal Detachment.”
The primary endpoint of this Phase
I clinical trial was to evaluate the safety and tolerability of a single intravitreal dose of Vitargus in patients as a vitreous substitute
during vitrectomy surgery for retinal detachment. Intravitreal is a route of administration of a drug or other substance, in which the
substance is delivered into the eyes. The secondary endpoint of this Phase I clinical trial is to assess retinal attachment and Virtagus
degradation at day 90 and to assess best corrected visual acuity (“BVCA”) after vitrectomy surgery. BVCA refers to the best
possible vision a person can achieve. The primary and second endpoints are required by HREC for the purpose of evaluation of our Phase
I clinical trial application. We enrolled an aggregate number of 10 patient subjects in this trial. On November 17, 2016, we received
the approval from the Data and Safety Monitoring Board for the first subject, and nine more subjects were enrolled thereafter. In this
trial, Vitargus was injected into the vitreous cavity of vitrectomised eyes, whose vitreous gel was removed from the vitreous cavity
after a vitrectomy surgery. On August 24, 2020, a full clinical study report (CSR) of ABV-1701 Phase I clinical trial was issued. The
study results showed that ABV-1701 (Vitargus) was well-tolerated as a vitreous substitute without any apparent toxicity to ocular tissues.
Further, there was no indication of an increased overall safety risk with Vitargus. For efficacy, participants showed significant improvement
in visual acuity. The optical properties of Vitargus allowed the patients to see well and facilitated visualisation of the fundus immediately
following surgery. In addition, since Vitargus set as a stable semisolid gel adhering to the retina, it maintained its position without
requiring the patient to remain face-down following surgery.
ABV-1701, Vitargus® in vitrectomy
surgery, Phase II Study will be started in the 2nd quarter of 2023. A total of four (4) study sites in Australia and Thailand
join this multi-nation and multi-site clinical study. The Company is working on improvements to the Vitargus product through the new
batch of investigational product.
| II. | Co-development
Agreement with Rgene |
On May 26, 2017, American BriVision
Corporation entered into a co-development agreement (the “Co-Dev Agreement”) with Rgene Corporation (the “Rgene”),
a related party under common control by controlling beneficiary shareholder of YuanGene Corporation and the Company. Pursuant to Co-Dev
Agreement, BriVision and Rgene agreed to co-develop and commercialize ABV-1507 HER2/neu Positive Breast Cancer Combination Therapy, ABV-17
Pancreatic Cancer Combination Therapy and ABV-1527 Ovary Cancer Combination Therapy. Under the terms of the Co-Dev Agreement, Rgene is
required to pay the Company $3,000,000 in cash or stock of Rgene with equivalent value by August 15, 2017. The payment is for the compensation
of BriVision’s past research efforts and contributions made by BriVision before the Co-Dev Agreement was signed and it does not
relate to any future commitments made by BriVision and Rgene in this Co-Dev Agreement. In addition to the $3,000,000, the Company is
entitled to receive 50% of the future net licensing income or net sales profit earned by Rgene, if any, and any development costs shall
be equally shared by both BriVision and Rgene.
By June 1, 2017, the Company had delivered
all research, technical, data and development data to Rgene. Since both Rgene and the Company are related parties and under common control
by a controlling beneficiary shareholder of YuanGene Corporation and the Company, the Company has recorded the full amount of $3,000,000
in connection with the Co-Dev Agreement as additional paid-in capital during the year ended September 30, 2017. During the year ended
December 31, 2017, the Company received $450,000 in cash. On December 24, 2018, the Company received the remaining balance of $2,550,000
in the form of newly issued shares of Rgene’s Common Stock, at the price of NT$50 (approximately equivalent to $1.60 per share),
for an aggregate number of 1,530,000 shares, which accounted for equity method long-term investment as of December 31, 2018. During the
year ended December 31, 2018, the Company has recognized investment loss of $549. On December 31, 2018, the Company determined to fully
write off this investment based on the Company’s assessment of the severity and duration of the impairment, and qualitative and
quantitative analysis of the operating performance of the investee, adverse changes in market conditions and the regulatory or economic
environment, changes in operating structure of Rgene, additional funding requirements, and Rgene’s ability to remain in business.
All projects that have been initiated will be managed and supported by the Company and Rgene.
The Company and Rgene signed an amendment
to the Co-Dev Agreement on November 10, 2020, pursuant to which both parties agreed to delete AB-1507 HER2/neu Positive Breast Cancer
Combination Therapy and AB 1527 Ovary Cancer Combination Therapy and add ABV-1519 EGFR Positive Non-Small Cell Lung Cancer Combination
Therapy and ABV-1526 Large Intestine / Colon / Rectal Cancer Combination Therapy to the products to be co-developed and commercialized.
Other provisions of the Co-Dev Agreement remain in full force and effect.
III. Clinical Development Service Agreement with Rgene
On June 10, 2022, the Company expanded
its co-development partnership with Rgene. BioKey entered into a Clinical Development Service Agreement with Rgene (“Service Agreement”)
to guide certain Rgene drug products, RGC-1501 for the treatment of Non-Small Cell Lung Cancer (NSCLC), RGC-1502 for the treatment of
pancreatic cancer and RGC 1503 for the treatment of colorectal cancer patients, through completion of Phase II clinical studies under
U.S. FDA IND regulatory requirements (the “Rgene Studies”).
Under the terms of the Service Agreement,
BioKey is eligible to receive payments totaling up to $3.0 million over a 3-year period with each payment amount to be determined by
certain regulatory milestones obtained during the agreement period. Through a series of transactions over the past 5 years, the Company
and Rgene have co-developed the three drug products covered by the Service Agreement, which has resulted in the Company owning 31.62%
of Rgene.
As part of the Rgene Studies, the
Company agreed to loan $1.0 million to Rgene, for which Rgene has provided the Company with a 5% working capital convertible loan (the
“Note”). If the Note is fully converted, the Company will own an additional 6.4% of Rgene. The Company is expected to receive
the outstanding loan from the related party by the 2023 Q1, either by cash or conversion of shares of Rgene. The Company may convert
the Note at any time into shares of Rgene’s common stock at either (i) a fixed conversion price equal to $1.00 per share or (ii)
20% discount of the stock price of the then most recent offering, whichever is lower; the conversion price is subject to adjustment as
set forth in the Note. The Service Agreement shall remain in effect until the expiration date of the last patent and automatically renew
for 5 more years unless terminated earlier by either party with six months written notice. Either party may terminate the Service Agreement
for cause by providing 30 days written notice.
Rgene has further agreed, effective
July 1, 2022, to provide the Company with a seat on Rgene’s Board of Directors until the loan is repaid in full. The Company has
nominated Dr. Jiang, its Chief Strategy Officer and Director to occupy that seat; Dr. Jiang is also one of the Company’s largest
shareholders, owning 12.8% of the Company. For more information about the Service Agreement and Note, please refer to the current reports
on Form 8-K filed on June 21, 2022.
BLEX 404, a new drug under clinical
development covered by the Service Agreement, is extracted from Maitake mushroom (Grifola frondosa), an edible mushroom. Its immunological
effects and the safety have been demonstrated in two Phase I/II clinical studies performed at Memorial Sloan Kettering Cancer Center
(MSKCC) with breast cancer and myelodysplastic syndromes (MDS) patients.
Market Distribution Strategy
We focus primarily on developing botanical drugs,
which are intended for use in the diagnosis, cure, mitigation or treatment of disease in humans. Together with our strategic partners,
we plan to market, distribute and sell our drug products internationally once those drug candidates comply with the local authorities
regulating drugs and foods. Currently, many countries follow the International Council for Harmonization of Technical Requirements for
Registration of Pharmaceuticals for Human Use (the “ICH”) guidelines that are published by European Medicines to provide
guidance on quality and safety of pharmaceutical development and new drug commercialization in Japan, the United States and Europe. All
of our drug candidates first go through the United States FDA process for new drug development first and then seek regulatory approval
from regulators equivalent to the FDA in the jurisdictions where we plan to distribute those candidates.
Intellectual Property
The new drug candidates are dependent on or are the subject of
the following patents and patent applications.
| No. |
|
Status |
|
Patent
No. |
|
Patent
Starting
Date |
|
Patent
Expiration
Date |
|
Patent
Name |
|
Territory |
|
Patent
Owner (1) (2) |
| 1 |
|
granted |
|
DE202007003503 U1 |
|
8/23/2007 |
|
9/20/2026 |
|
Novel Polygalatenosides
and use thereof as an antidepressant agent |
|
Germany |
|
MPITDC |
| 2 |
|
granted |
|
7531519 |
|
5/12/2009 |
|
9/20/2026 |
|
Novel Polygalatenosides
and use thereof as an antidepressant agent |
|
The U.S. |
|
MPITDC |
| 3 |
|
granted |
|
4620652 |
|
11/20/2006 |
|
11/19/2026 |
|
Novel Polygalatenosides
and use thereof as an antidepressant agent |
|
Japan |
|
MPITDC |
| 4 |
|
granted |
|
I 314453 |
|
9/21/2006 |
|
9/20/2026 |
|
Novel Polygalatenosides
and use thereof as an antidepressant agent |
|
Taiwan |
|
MPITDC |
| 5 |
|
granted |
|
I389713 |
|
3/21/2013 |
|
10/13/2030 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute (3) |
|
Taiwan |
|
NHRI |
| 6 |
|
granted |
|
US 8197849 B2 |
|
6/12/2012 |
|
8/30/2030 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
The U.S. |
|
NHRI |
| 7 |
|
granted |
|
AU 2011/215775 B2 |
|
4/17/2014 |
|
2/9/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
Australia |
|
NHRI |
| 8 |
|
granted |
|
KR 10-1428898 |
|
8/4/2014 |
|
2/9/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
Korea |
|
NHRI |
| 9 |
|
granted |
|
CA 2786911 (C) |
|
10/6/2015 |
|
2/10/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
Canada |
|
NHRI |
| 10 |
|
granted |
|
WO2011100469 A1 |
|
N/A(4) |
|
N/A(4) |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
PCT |
|
NHRI |
| 11 |
|
granted |
|
EP 2534200 |
|
4/8/2015 |
|
2/9/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
European Union (Germany,
United Kingdom, France, Switzerland, Spain, Italy) |
|
NHRI |
| 12 |
|
granted |
|
特許第
5885349號 |
|
2/9/2011 |
|
2/9/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute |
|
Japan |
|
NHRI |
| 13 |
|
granted |
|
ZL 201180005494.7 |
|
12/24/2014 |
|
2/9/2031 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute(3) |
|
China |
|
NHRI |
| 14 |
|
granted |
|
HK1178188 |
|
3/6/2015 |
|
6/21/2030 |
|
Cross-linked oxidized
hyaluronic acid for use as a vitreous substitute(3) |
|
Hong Kong (5) |
|
NHRI |
| 15 |
|
granted |
|
US 16/936,032 |
|
9/4/2020 |
|
9/4/2040 |
|
Polygala extract for
the treatment of major depressive disorder |
|
US |
|
BioLite |
| 16 |
|
granted |
|
TW I821593 |
|
11/1/2023 |
|
7/22/2040 |
|
Polygala extract for
the treatment of major depressive disorder |
|
Taiwan |
|
BioLite |
| 17 |
|
granted |
|
US17/120,965 |
|
12/20/2020 |
|
12/20/2040 |
|
Polygala Extract for
the Treatment of Attention Deficit Hyperactive Disorder |
|
U.S. |
|
BioLite |
| 18 |
|
granted |
|
TW 110106546 |
|
2/24/2021 |
|
2/24/2041 |
|
Polygala Extract for
the Treatment of Attention Deficit Hyperactive Disorder |
|
Taiwan |
|
BioLite |
| 19 |
|
granted |
|
TW I792427 |
|
02/11/2023 |
|
07/19/2041 |
|
Storage Media For Preservation
of Corneal Tissue |
|
Taiwan |
|
NHRI |
| 20 |
|
granted |
|
AU2021314052B2 |
|
04/09/2024 |
|
04/09/2041 |
|
Polygala Extract for
the Treatment of Major Depressive Disorder |
|
Australia |
|
BioLite |
| 21 |
|
applied |
|
202180001626. 2 |
|
|
|
|
|
Polygala Extract
for the Treatment of Major Depressive Disorder |
|
China |
|
|
| 22 |
|
applied |
|
特願 2023502736 |
|
|
|
|
|
Polygala Extract for the Treatment
of Major Depressive Disorder |
|
Japan |
|
|
| 23 |
|
applied |
|
21 846 424.6 |
|
|
|
|
|
Polygala Extract for the Treatment of Major Depressive Disorder |
|
Europe |
|
|
| 24 |
|
applied |
|
110106546 |
|
|
|
|
|
Polygala Extract for the Treatment of Attention-Deficient and Hyperactivity Disorder |
|
Taiwan |
|
|
| 25 |
|
applied |
|
202180001615. 4 |
|
|
|
|
|
Polygala Extract for the Treatment of Attention-Deficient and Hyperactivity Disorder |
|
China |
|
|
| 26 |
|
applied |
|
特願 2023536203 |
|
|
|
|
|
Polygala Extract for the Treatment of Attention-Deficient and Hyperactivity Disorder |
|
Japan |
|
|
| 27 |
|
applied |
|
21 907 345.9 |
|
|
|
|
|
Polygala Extract for the Treatment of Attention-Deficient and Hyperactivity Disorder |
|
Europe |
|
|
| 28 |
|
applied |
|
2021403197 |
|
|
|
|
|
Polygala Extract for the Treatment of Attention-Deficient and Hyperactivity Disorder |
|
Australia |
|
|
| (1) | “MPITDC”
stands for Medical and Pharmaceutical Industry Technology and Development Center, Taiwan. |
| (2) | “NHRI”
stands for National Health Research Institutes, Taiwan. |
| (3) | The
patent name is translated into English and the original patent name is written as “交联氧化透明质酸作为眼球玻璃体之替代物.” |
| (4) | The
starting date and expiration date of patents under PTC are subject to the laws of the specific participating jurisdiction where the patent
application is filed. We have subsequently submitted such patent to the jurisdictions listed in No.22 herein above. |
| (5) | NHRI
has obtained standard patent in Hong Kong based on the registration of the patent (listed as No.24 herein) granted by the State Intellectual
Property Office, People’s Republic of China. |
Corporate History and Structure
ABVC was incorporated under the laws of the State
of Nevada on February 6, 2002 and has three wholly-owned Subsidiaries: BriVision, BioLite Holding, Inc. and BioKey, Inc. BriVision was
incorporated in July 2015 in the State of Delaware and is in the business of developing pharmaceutical products in North America.
BioLite Holding was incorporated under the laws
of the State of Nevada on July 27, 2016, with 500,000,000 shares authorized, par value $0.0001. Its key Subsidiaries include BioLite
BVI, Inc. (“BioLite BVI”) that was incorporated in the British Virgin Islands on September 13, 2016 and BioLite Inc. (“BioLite
Taiwan”), a Taiwanese corporation that was founded in February 2006. BioLite Taiwan has been in the business of developing new
drugs for over twelve years. Certain shareholders of BioLite Taiwan exchanged approximately 73% of equity securities in BioLite Taiwan
for the Common Stock in BioLite Holding in accordance with a share purchase/ exchange agreement (the “Share Purchase/ Exchange
Agreement”). As a result, BioLite Holding owns via BioLite BVI approximately 73% of BioLite Taiwan. The other shareholders who
did not enter this Share Purchase/ Exchange Agreement retain their equity ownership in BioLite Taiwan.
Incorporated in California on November 20, 2000,
BioKey has chosen to initially focus on developing generic drugs to ride the opportunity of the booming industry.
Upon closing of the Mergers on February 8, 2019,
BioLite and BioKey became two wholly-owned subsidiaries of ABVC.
In November 2023, the Company and one of its
subsidiaries, BioLite, Inc. (“BioLite”) each entered into a multi-year, global licensing agreement with AiBtl BioPharma Inc.
(“AIBL”) for the Company and BioLite’s CNS drugs with the indications of MDD (Major Depressive Disorder) and ADHD (Attention
Deficit Hyperactivity Disorder) (the “Licensed Products”). The license covers the Licensed Products’ clinical trial,
registration, manufacturing, supply, and distribution rights. The parties are determined to collaborate on the global development of
the Licensed Products. The parties are also working to strengthen new drug development and business collaboration, including technology,
interoperability, and standards development. As per each of the respective agreements, each of ABVC and BioLite received 23 million shares
of AIBL stock at $10 per share, and if certain milestones are met, each may receive $3,500,000 and royalties equaling 5% of net sales,
up to $100 million. Upon the issuance of the shares, AIBL became a subsidiary of ABVC.
The following chart illustrates the corporate
structure of ABVC:

Effective March 5, 2022, the Company’s
Board for Directors approved amending the Company’s Bylaws to remove Section 2.8, which permitted cumulative voting for directors
since cumulative voting is specifically prohibited by our Articles of Incorporation. Since it is not otherwise stated in our Articles
of Incorporation or Bylaws, directors shall be elected by a plurality of the votes cast at the election, as provided in the Nevada Revised
Statutes.
Effective March 14, 2024, the Company’s
Board for Directors approved amending the Company’s Bylaws to amend Section 2.8 of the Company’s Bylaws to revise the number
of shares needed to establish a quorum at shareholder meetings. The Amendment changes the quorum requirement from a majority to 33-1/3%
of the votes entitled to be cast on a matter.
Competition
The healthcare industry is highly competitive
and subject to significant and rapid technological change as researchers learn more about diseases and develop new technologies and treatments.
Significant competitive factors in our industry include product efficacy and safety; quality and breadth of an organization’s technology;
skill of an organization’s employees and its ability to recruit and retain key employees; timing and scope of regulatory approvals;
the average selling price of products; the availability of raw materials and qualified manufacturing capacity; manufacturing costs; intellectual
property and patent rights and their protection; and our capabilities of securing competent collaborators. Market acceptance of our current
products and product candidates will depend on a number of factors, including: (i) potential advantages over existing or alternative
therapies or tests, (ii) the actual or perceived safety of similar classes of products, (iii) the effectiveness of sales, marketing,
and distribution capabilities, and (iv) the scope of any approval provided by the FDA or foreign regulatory authorities.
Since we are a small biopharmaceutical company
compared to other companies that we may compete against, it is our intention to license our products to much larger pharmaceutical, specialty
pharmaceutical and generic drug companies with the financial, technical and human resources to compete effectively in the markets we
address.
We anticipate that our license partners will
face intense and increasing competition when and as our new drug candidates enter the markets, as advanced technologies become available
and as generic forms of currently branded products become available. Finally, the development of new treatment methods for the diseases
we are targeting could render our products non-competitive or obsolete. There can be no assurance that any of our new drug candidates
will be clinically superior or scientifically preferable to products developed or introduced by our competitors.
The following chart lists some, not all, of the
biopharmaceutical companies that research, develop, commercialize, distribute or sell drugs that are in competition with our drug candidates.
| Disease |
|
Drug
Name |
|
Pharmaceutical
Companies |
|
Headquarters |
| Major Depressive Disorder |
|
Cymbalta oral |
|
Eli Lilly and Co., Inc. |
|
IN |
| |
|
Lexapro oral |
|
Forest Laboratories, Inc. |
|
NJ |
| |
|
|
|
Pfizer Pharmaceuticals, Inc. |
|
CT |
| |
|
|
|
|
|
|
| Attention-Deficit |
|
Adderall XR |
|
Shire Development LLC |
|
MA |
| Hyperactivity Disease |
|
Ritalin |
|
Novartis Pharmaceuticals Corporation |
|
NJ |
| |
|
Dexedrine |
|
Amedra Pharmaceuticals LLC |
|
PA |
| |
|
|
|
|
|
|
| Myelodysplastic |
|
Vidaza |
|
Celgene Corporation |
|
NJ |
| Syndromes |
|
Dacogen |
|
Astex Pharmaceuticals, Inc. |
|
CA |
| |
|
|
|
|
|
|
| Triple Negative Breast
Cancer |
|
Avastin |
|
Genentech, Inc. |
|
CA |
| |
|
Erbitux (Cetuximab) |
|
ImClone Systems Incorporated |
|
NY |
| |
|
|
|
|
|
|
| Pancreatic Cancer |
|
Abraxane, Abraxis BioScience
LLC |
|
Los Angeles |
|
CA |
| |
|
Novartis Pharma Stein AG |
|
Stein |
|
Switzerland |
| |
|
|
|
|
|
|
| Vitargus for the treatments |
|
Alcon Laboratories, Inc. |
|
Fort Worth |
|
TX |
of
Retinal Detachment or
Vitreous Hemorrhage |
|
Arcadophta |
|
Toulouse |
|
France |
Government Regulations
Currently, we are focusing on the research and
development of six therapeutic candidates in the fields of CNS, oncology/hematology and autoimmune, for which regulatory approval must
be received before we can commence marketing. In addition, our cGMP facility is subject to review by the FDA. Regulatory approval processes
and FDA regulations for ABVC’s current and any future product candidates are discussed below.
Approval Process for Pharmaceutical Products
FDA Approval Process for Pharmaceutical Products
In the U.S., pharmaceutical products are subject
to extensive regulation by the FDA. The Federal Food, Drug and Cosmetic Act (the “FDC Act”), and other federal and state
statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval,
labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical
products. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions,
such as FDA refusal to approve pending NDAs, warning letters, product recalls, product seizures, total or partial suspension of production
or distribution, injunctions, fines, civil penalties, and criminal prosecution. Pharmaceutical product development in the U.S. typically
involves the performance of satisfactory nonclinical, also referred to as pre-clinical, laboratory and animal studies under the FDA’s
Good Laboratory Practice, or GLP, regulation, the development and demonstration of manufacturing processes, which conform to FDA mandated
current good manufacturing requirements, or cGMPs, including a quality system regulating manufacturing, the submission and acceptance
of an IND application, which must become effective before human clinical trials may begin in the U.S., obtaining the approval of Institutional
Review Boards, or IRBs, at each site where we plan to conduct a clinical trial to protect the welfare and rights of human subjects in
clinical trials, adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication
for which FDA approval is sought, and the submission to the FDA for review and approval of an NDA. Satisfaction of FDA requirements typically
takes many years and the actual time required may vary substantially based upon the type, complexity, and novelty of the product or disease.
Pre-clinical tests generally include laboratory
evaluation of a product candidate, its chemistry, formulation, stability and toxicity, as well as certain animal studies to assess its
potential safety and efficacy. Results of these pre-clinical tests, together with chemistry, manufacturing controls and analytical data
and the clinical trial protocol, which details the objectives of the trial, the parameters to be used in monitoring safety, and the effectiveness
criteria to be evaluated, along with other requirements must be submitted to the FDA as part of an IND, which must become effective before
human clinical trials can begin. The entire clinical trial and its protocol must be in compliance with what are referred to as good clinical
practice, or GCP, requirements. The term, GCP, is used to refer to various FDA laws and regulations, as well as international scientific
standards intended to protect the rights, health and safety of patients, define the roles of clinical trial sponsors and assure the integrity
of clinical trial data.
An IND automatically becomes effective 30 days
after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions about the intended conduct of the
trials and imposes what is referred to as a clinical hold. Pre-clinical studies generally take several years to complete, and there is
no guarantee that an IND based on those studies will become effective, allowing clinical testing to begin. In addition to FDA review
of an IND, each medical site that desires to participate in a proposed clinical trial must have the protocol reviewed and approved by
an independent IRB or Ethics Committee, or EC. The IRB considers, among other things, ethical factors, and the selection and safety of
human subjects. Clinical trials must be conducted in accordance with the FDA’s GCP requirements. The FDA and/or IRB may order the
temporary, or permanent, discontinuation of a clinical trial or that a specific clinical trial site be halted at any time, or impose
other sanctions for failure to comply with requirements under the appropriate entity jurisdiction.
Clinical trials to support NDAs for marketing
approval are typically conducted in three sequential phases, but the phases may overlap.
In Phase I clinical trials, a product candidate
is typically introduced either into healthy human subjects or patients with the medical condition for which the new drug is intended
to be used. The main purpose of the trial is to assess a product candidate’s safety and the ability of the human body to tolerate
the product candidate. Phase I clinical trials generally include less than 50 subjects or patients.
During Phase 2 trials, a product candidate is
studied in an exploratory trial or trials in a limited number of patients with the disease or medical condition for which it is intended
to be used in order to: (i) further identify any possible adverse side effects and safety risks, (ii) assess the preliminary or potential
efficacy of the product candidate for specific target diseases or medical conditions, and (iii) assess dosage tolerance and determine
the optimal dose for Phase III trials.
Phase III trials are generally undertaken to
demonstrate clinical efficacy and to further test for safety in an expanded patient population with the goal of evaluating the overall
risk-benefit relationship of the product candidate. Phase III trials are generally designed to reach a specific goal or endpoint, the
achievement of which is intended to demonstrate the candidate product’s clinical efficacy and adequate information for labeling
of the approved drug.
The FDA has 60 days from its receipt of an NDA
to determine whether the application will be accepted for filing based on the FDA’s threshold determination that it is sufficiently
complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. The FDA has agreed
to certain performance goals in the review of NDAs. Most applications for standard review drug products are reviewed within ten months;
most applications for priority review drugs are reviewed within six months. Priority review can be applied to drugs that the FDA determines
offer major advances in treatment, or provide a treatment where no adequate therapy exists. The review process for both standard and
priority review may be extended by the FDA for three additional months to consider certain late-submitted information, or information
intended to clarify information already provided in the submission. The FDA may also refer applications for novel drug products, or drug
products which present difficult questions of safety or efficacy, to an advisory committee — typically a panel that includes clinicians
and other experts — for review, evaluation, and a recommendation as to whether the application should be approved. The FDA is not
bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA
will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the
facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with cGMPs is satisfactory and the
NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing
facilities, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies
in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application. If
and when those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval
letter. The FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included. An
approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition
of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug
outweigh the potential risks.
REMS can include medication guides, communication
plans for healthcare professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training
or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient
registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval
may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product
approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
Post-Approval Regulations
Even if a product candidate receives regulatory
approval, the approval is typically limited to specific clinical indications. Further, even after regulatory approval is obtained, subsequent
discovery of previously unknown problems with a product may result in restrictions on its use or even complete withdrawal of the product
from the market. Any FDA-approved products manufactured or distributed by us are subject to continuing regulation by the FDA, including
record-keeping requirements and reporting of adverse events or experiences. Further, drug manufacturers and their subcontractors are
required to register their establishments with the FDA and state agencies, and are subject to periodic inspections by the FDA and state
agencies for compliance with cGMPs, which impose rigorous procedural and documentation requirements upon us and our contract manufacturers.
ABVC cannot be certain that ABVC or its present or future contract manufacturers or suppliers will be able to comply with cGMPs regulations
and other FDA regulatory requirements. Failure to comply with these requirements may result in, among other things, total or partial
suspension of production activities, failure of the FDA to grant approval for marketing, and withdrawal, suspension, or revocation of
marketing approvals.
If the FDA approves one or more of our product
candidates, ABVC must provide certain updated safety and efficacy information. Product changes, as well as certain changes in the manufacturing
process or facilities where the manufacturing occurs or other post-approval changes may necessitate additional FDA review and approval.
The labeling, advertising, promotion, marketing and distribution of a drug must be in compliance with FDA and Federal Trade Commission,
or FTC, requirements which include, among others, standards and regulations for direct-to-consumer advertising, off-label promotion,
industry sponsored scientific and educational activities, and promotional activities involving the Internet. The FDA and FTC have very
broad enforcement authority, and failure to abide by these regulations can result in penalties, including the issuance of a warning letter
directing us to correct deviations from regulatory standards and enforcement actions that can include seizures, fines, injunctions and
criminal prosecution.
Foreign Regulatory Approval
Outside of the U.S., ABVC’s ability to
market our product candidates will be contingent also upon its receiving marketing authorizations from the appropriate foreign regulatory
authorities, whether or not FDA approval has been obtained. The foreign regulatory approval process in most industrialized countries
generally encompasses risks similar to those ABVC will encounter in the FDA approval process. The requirements governing conduct of clinical
trials and marketing authorizations, and the time required to obtain requisite approvals, may vary widely from country to country and
differ from those required for FDA approval.
ABVC will be subject to additional regulations
in other countries in which we market, sell and import our products, including Canada. ABVC or its distributors must receive all necessary
approvals or clearance prior to marketing and/or importing our products in those markets.
Other Regulatory Matters
Manufacturing, sales, promotion and other activities
following product approval are also subject to regulation by numerous regulatory authorities in addition to the FDA, including, in the
U.S., the Centers for Medicare & Medicaid Services, other divisions of the Department of Health and Human Services, the Drug Enforcement
Administration, the Consumer Product Safety Commission, the Federal Trade Commission, the Occupational Safety &Health Administration,
the Environmental Protection Agency and state and local governments. In the U.S., sales, marketing and scientific/educational programs
must also comply with state and federal fraud and abuse laws. Pricing and rebate programs must comply with the Medicaid rebate requirements
of the U.S. Omnibus Budget Reconciliation Act of 1990 and more recent requirements in the Health Care Reform Law, as amended by the Health
Care and Education Affordability Reconciliation Act, or ACA. If products are made available to authorized users of the Federal Supply
Schedule of the General Services Administration, additional laws and requirements apply. The handling of any controlled substances must
comply with the U.S. Controlled Substances Act and Controlled Substances Import and Export Act. Products must meet applicable child-resistant
packaging requirements under the U.S. Poison Prevention Packaging Act. Manufacturing, sales, promotion and other activities are also
potentially subject to federal and state consumer protection and unfair competition laws.
The distribution of pharmaceutical products is
subject to additional requirements and regulations, including extensive recordkeeping, licensing, storage and security requirements intended
to prevent the unauthorized sale of pharmaceutical products.
The failure to comply with regulatory requirements
subjects firms to possible legal or regulatory action. Depending on the circumstances, failure to meet applicable regulatory requirements
can result in criminal prosecution, fines, imprisonment or other penalties, injunctions, recall or seizure of products, total or partial
suspension of production, denial or withdrawal of product approvals, or refusal to allow a firm to enter into supply contracts, including
government contracts. In addition, even if a firm complies with FDA and other requirements, new information regarding the safety or effectiveness
of a product could lead the FDA to modify or withdraw product approval. Prohibitions or restrictions on sales or withdrawal of future
products marketed by us could materially affect our business in an adverse way.
Changes in regulations, statutes or the interpretation
of existing regulations could impact our business in the future by requiring, for example: (i) changes to our manufacturing arrangements;
(ii) additions or modifications to product labeling; (iii) the recall or discontinuation of our products; or (iv) additional record-keeping
requirements. If any such changes were to be imposed, they could adversely affect the operation of our business.
Employees
As of December 31, 2023, we, including the
subsidiaries, have 19 employees, 16 of which are full-time, located in the U.S. and Taiwan.
| Functional Area | |
Number of
Employees | |
| Senior management | |
| 4 | |
| Research and development | |
| 9 | |
| Administration | |
| 2 | |
| Accounting | |
| 4 | |
| Total | |
| 19 | |
ABVC believes that it maintains a good working
relationship with its employees. ABVC offers its employees competitive benefits, including a pleasant and rewarding work environment,
career-oriented training, and career growth opportunities. ABVC believes its employees are devoted to delivering superb services. ABVC
did not experience any significant labor disputes.
Legal Proceedings
From time to time ABVC and its Subsidiaries may
become involved in legal proceedings and claims, or be threatened with other legal actions and claims, arising in the ordinary course
of business relating to its intellectual property, product liability, regulatory compliance and/or marketing and advertising of its products.
As of the date of this prospectus, ABVC and its Subsidiaries were not involved or threatened with any legal actions and regulatory proceedings.
Environment
ABVC seeks to comply with all applicable statutory
and administrative requirements concerning environmental quality. Expenditures for compliance with federal state and local environmental
laws have not had, and are not expected to have, a material effect on ABVC’s capital expenditures, results of operations or competitive
position.
Properties
Our Subsidiary BioLite has its laboratories
located in Hsinchu Biomedical Science Park, with an address of 20, Sec. 2, Shengyi Rd., 2nd Floor, Zhubei City, Hsinchu County 302, Taiwan
(R.O.C.). On January 1, 2015, BioLite Taiwan entered into a lease agreement with the National Science Park Administrative Office (Hsinchu
City) under which it rents two dormitory buildings in Hsinchu County, Taiwan for a period of five years. The aggregate leasing area amounts
to approximately 678 square meters (equivalent to approximately 7,298 square feet) on the second floor of the building. The leased space
counts for approximately 1.9% of the total space of the building. On January 1, 2020, BioLite Taiwan extended the contract for another
five years. The new expiration date is on December 31, 2024. The rent increases by a small percentage each year during the term of the
lease agreement. BioLite paid $50,572 and $60,104 in rental expense for the laboratory space for the years ended December 31, 2023 and
2022, respectively.
Another subsidiary BioKey is headquartered
in Fremont, California. BioKey’s office lease will end on February 28, 2026 and the office occupies approximately 28,186 square
feet. BioKey’s space consists of offices, research and production laboratories, and manufacturing facilities, which are GMP certified.
BioKey has an option to extend the lease for its offices in Fremont for a period of five years commencing February 28, 2026, and BioKey
may exercise this option for 5 more years. The total BioKey’s rental expenses were $353,466 and $328,051 for the years ended December
31, 2023 and 2022, respectively.
MANAGEMENT
The following table sets forth as of the date
of this prospectus, the name, age, and position of each executive officer and director and the term of office of each such person.
| Name |
|
Age |
|
Title |
| Eugene Jiang |
|
37 |
|
Chairman of the Board and Chief Business Officer
(“CBO”) |
| Dr. Uttam Patil |
|
38 |
|
Chief Executive Officer (“CEO”) |
| Leeds Chow |
|
34 |
|
Chief Financial Officer (“CFO”) |
| Dr. Tsung-Shann (T.S.) Jiang |
|
69 |
|
Chief Strategy Officer (“CSTRO”) and
Director |
| Dr. Tsang Ming Jiang |
|
62 |
|
Director |
| Dr. Chang-Jen Jiang |
|
67 |
|
Director |
| Norimi Sakamoto |
|
52 |
|
Independent Director |
| Yen-Hsin Chou |
|
42 |
|
Independent Director |
| Hsin-Hui Miao |
|
56 |
|
Independent Director |
| Yoshinobu Odaira |
|
75 |
|
Independent Director |
| Che-Wei Hsu |
|
42 |
|
Independent Director |
| Shuling Jiang |
|
67 |
|
Director |
| Yu-Min (Francis) Chung |
|
58 |
|
Independent Director |
| Dr. Chi-Hsin (Richard) King |
|
74 |
|
Chief Scientific Officer (“CSO”) |
Set forth below is certain biographical information
regarding each of our directors and executive officers as of the date of this prospectus.
Eugene Jiang, Chairman, has served as
our CEO and President since the Company’s inception in July 2015 until he resigned on September 15, 2017. He remains the Chairman
of the Board. He also serves as our CBO since September 2019 and serves as the CBO of BioKey, Inc. since 2019. Mr. Jiang also serves
as Director for BioLite Incorporation since June 2015 and as Director for BioFirst Corp. since 2012. He also serves as CEO for Genepro
Investment Company since March 2010. Mr. Jiang obtained a PMBA degree from National Taiwan University in 2017 and an EMBA degree from
the University of Texas in Arrington in 2010. And in 2009, Mr. Jiang received a bachelor’s degree in Physical Education from Fu-Jen
Catholic University.
Dr. Uttam Patil, CEO, was appointed
as the Company’s Chief Executive Officer on June 21, 2023. Dr. Patil has served as the Chief Operating and Scientific Officer of
the Company’s subsidiary, BioKey, Inc. since May 2023; he also works for Rgene Corporation (a related party), as the R&D Manager
since May 2023, after being promoted from Project Manager, to which he serves from August 2022 to May 2023. Prior to that, Dr. Patil
was a Post-Doctoral Research Fellow at NTNU from March 2020 to July 2022. In 2019, Dr. Patil received the “Platinum Award”
for an Oral Presentation on the topic, “Nucleobase Functionalized Single-Walled Carbon Nanotubes Hybridization with Single-Stranded
DNA” at a Workshop on Organic Chemistry for Junior Chemists held in South Korea. Dr. Patil received his Ph.D. in Chemistry from
National Tsing Hua University and a Masters in Analytical Chemistry from Pune University, as well as a Bachelors in industrial chemistry
from Pune University.
Leeds Chow, CFO, was appointed as the
Company’s Chief Financial Officer and Principal Accounting Officer on September 4, 2022. He has served as a Financial Controller
of the Company from March 2021 to August 2022. Mr. Chow has over 12 years of experience in Audit and Financing Industry. He has served
as the finance manager in a family office, in charge of managing investment portfolios, handling financial and operating aspects. He
has also worked in a local investment company in Hong Kong, serving as a financial advisor during the Hong Kong Initial Public Offering
process, as well as preparing opinion letters as an independent financial advisor for transactions for Hong Kong listed companies. Mr.
Chow graduated in University of California, Santa Barbara, with a Bachelor of Arts degree, majoring in Business Economics with Accounting
Emphasis.
Dr. T.S. Jiang, Chief Strategy Officer and
Director, has served as the Company’s Chief Strategy Officer since September 2019. Dr. Jiang serves as the CEO of Biokey, Inc.
since December 2021, as a director of BioFirst Corp. since 2013, and has been the CEO and chairman of BioLite, Inc., a subsidiary of
BioLite BVI, Inc., since January 2010. Prior to BioLite, Dr. Jiang served as the president and/or chairman of multiple biotech companies
in Taiwan, including PhytoHealth Corporation from 1998 to 2009 and AmCad BioMed Corporation from 2008 to 2009. In addition, Dr. Jiang
is a director on various biotech associations, such as the Taiwan Bio Industry Organization (Taiwan) from 2006 to 2008 and the Chinese
Herbs and Biotech Development Association in Taiwan from 2003 to 2006. Dr. Jiang was an assistant professor at University of Illinois
from 1981 to 1987 and an associate professor at Rutgers, the State University of New Jersey from 1987 to 1990 and served as a professor
at a few Taiwanese universities during a period from 1990 to 1993, such as National Taiwan University, National Cheng Kung University
and Tunghai University. Dr. Jiang obtained his bachelor degree in Engineering and Chemical Engineering from National Taiwan University
in Taiwan in 1976, masters and Ph.D. from Northwestern University in the U.S. in 1981 and Executive Master of Business Administration
(“EMBA”) from National Taiwan University in Taiwan in 2007. As a successful entrepreneur, Dr. Jiang has developed and commercialized
PG2 Lyo Injection, a new drug to treat cancer related fatigue. From 1998 to 2009, Dr. T. S. Jiang served as President of Phyto Health
Corporation where he led a project team to develop PG2 Injectable. This product was extracted, isolated and purified from a type of Traditional
Chinese Medicine. PG2 Injection was intended for cancer patients who had trouble recovering from severe fatigue. Dr. Jiang oversaw and
managed the R&D department, daily corporate operations and business of Phyto Health Corporation when he was the President. PG2 Lyo
Injection received approval on its NDA from Taiwan Food and Drug Administration in 2010 and later was launched into the Taiwan market
in 2012. We believe that Dr. Jiang provides leadership and technological guidance on our strategic development and operations.
Dr. Tsang Ming Jiang, Director, has served
as a director of BioFirst Corp. since 2017 and as a technical director at Supermicro Computer, Inc. since August 2022. Dr. Jiang served
as a technical director at the Industrial Technology Research Institute in Taiwan from February 2017 to July 2021. Prior to joining the
Industrial Technology Research Institute as a technical director, Dr. Jiang worked at the Company as chief information officer from November
2016 to January 2017, Ericsson as engineering manager from 2013 to 2016 and the Industrial Technology Research Institute as deputy director
from October 2011 to February 2013. In addition, Dr. Jiang worked at several other research institutes, including University of Alaska
Fairbanks, National Taiwan University and Chung Cheng University, with his research interest in cloud computing and Internet security,
especially in the areas of virtualization, software-defined data centers, SDN enabled networks and big data analytics. Dr. Jiang received
his Bachelor of Science in electrical engineering in 1983 and Master of Science in electrical engineering in 1984, both from National
Taiwan University, and his Ph.D. in electrical engineering and computer science from University of Illinois at Chicago in 1988. Dr. Tsang
Ming Jiang is a brother of Dr. Tsung-Shann Jiang, who together with his wife collectively owns 80% of Lion Arts Promotion, Inc. which
has approximately 69.3% of ownership interest in the Company through YuanGene Corporation, a wholly-owned subsidiary of Lion Arts Promotion,
Inc.
Dr. Chang-Jen Jiang, Director, has served
as a director of BioLite Inc. since 2013 and as a director of BioFirst Corp. since 2015. Dr. Jiang has been a pediatrician at the department
of pediatrics of Eugene Women and Children Clinic since 2016. Previously, Dr. Chang-Jen worked as an attending doctor at the department
of pediatrics of Keelung Hospital, the Ministry of Health and Welfare in Taiwan from 1994 to 2009. Before his position at Keelung Hospital,
he was a chief doctor at the department of pediatrics, hematology and oncology of Mackay Memorial Hospital in Taiwan for three years
until 1994. Dr. Chang-Jen Jiang obtained his doctor of medicine degree (the Taiwanese equivalent degree of MD) from Taipei Medical University
in Taiwan in 1982 and started his career in Mackay Memorial Hospital. We believe that the Company will benefit from Dr. Jiang’s
knowledge in biology and experiences in medical practice.
Norimi Sakamoto, Director, currently serves
a director at Shogun Maitake Canada Co., Ltd. from June 2016. Ms. Sakamoto served as the chief executive officer of MyLife Co., Ltd.
from June 2013 to March 2020. Ms. Sakamoto started her career in 1997 from Sumitomo Corporation Hokkaido Co., Ltd. in Japan. Ms. Sakamoto
received her Bachelor Degree of Arts in travel and tourism from Davis and Elkins College in 1993 and Master of Science in urban studies
from the University of New Orleans in 1995.
Yen-Hsin Chou, Director, has served as
a financial specialist at Mega Bank since 2011. Ms. Chou’s responsibilities primarily include customer services and financial consultations.
Ms. Chou received a Bachelor Degree in finance and economics from Yuan Ze University School of Economics in 2010.
Hsin-Hui Miao, Director, served as counter
manager at Yueh Shan Chi Cram School from August 2021 to May 2022. From August 1988 to July 2021, Ms. Miao was a kindergarten teacher
and also severed as the leader of general affairs team at the affiliated high school of Tunghai University, Kindergarten Division. Ms.
Miao received her Bachelor Degree of Education from Taichung University of Education in 1998.
Yoshinobu Odaira, Director, was elected
as a director on our Board of Directors on February 8, 2019. He is an entrepreneur and has founded a number of Japanese agricultural
companies, including Yukiguni Maitake, our licensing partner. In 1983, Mr. Odaira established Yukiguni Maitake, which became a public
company in Japan in 1994. In 2015, Bain Capital Private Equity purchased Yukiguni Maitake through a tender offer. In addition to his
success with Yukiguni Maitake, Mr. Odaira served as the CEO of Yukiguni Shoji Co., Ltd. since 1988, as the CEO of Odaira Shoji Co., Ltd.
from 1989 and as a director of Shogun Maitake Japan Co., Ltd. since June 1989. In 2015, Mr. Odaira founded two new companies, Shogun
Maitake Canada Co., Ltd. in Canada and Odaira Kinoko Research Co., Ltd. in Japan. Mr. Odaira has served as the CEO and director of Shogun
Maitake Canada Co., Ltd. since June 2016. Mr. Odaira served as a director of BioLite Inc. from February 2019 to April 2019. Yoshinobu
Odaira graduated from the Ikazawa Junior High School in 1963. We believe that we will benefit from Mr. Odaira’s successful business
experience.
Che Wei Hsu , Director, is currently
employed as a clerk by Chunghwa Post Co., Ltd. since August 2016; previously she was a teacher in a Junior High School. Ms. Hsu received
a Bachelor Degree from Tunghai University School of Chinese Literature in 2004.
Shuling Jiang , Director, has served
as a director for various companies, including BioLite, Inc. and BioFirst Corp, since 2017 and started to serve as Managing Director
for Biokey, Inc. in 2022. Ms. Jiang received a Bachelor Degree from National Taiwan Normal University School of Music in 1978 and a Master
Degree from Northwestern University School of Music in 1983.
Yu-Min (Francis) Chung, Director, was
a Partner at Maxpro Ventures, an investment firm in Taiwan focused on breakthrough biomedical technology companies, from July 2018 to
May 2022. Prior to that, he served as Vice President at TaiAn Technology, which is a biotechnology service company and a management company
for biotechnology venture capital funds in Taiwan, from June 2016 to June 2018. Mr. Chung received his Bachelor’s Degree of Science
in Chemistry from National Taiwan University in 1987, Master’s Degree in Business Administration from National Taiwan University
in 2006, and Ph.D. in Pharmacy from University of Iowa in 1995.
Significant Employees
The following are employees who are not executive
officers, but who are expected to make significant contributions to our business:
Dr. Chi-Hsin Richard King, CSO. Effective
September 15, 2017, the Board appointed Dr. Chi-Hsin Richard King as the CSO of the Company. Dr. Chi-Hsin Richard King, 71, retired since
July 2017. He served as the consultant at TaiGen Biotechnology Co. Ltd (“TaiGen”), a Taiwan company in the biotechnology
business, from August 2016 to July 2017, the Senior Vice President at TaiGen from July 2008 to August 2016 and as the Vice President
at Research and Development of TaiGen from June 2005 to July 2008. Dr. King served as the Director at Albany Molecular Research Inc.
(“AMRI”), a New York corporation, from January 2003 to June 2005, the Assistant Director at Medicinal Chemistry Department
of AMRI from January 2000 to December 2002 and the Assistant Director at Chemical Development Department of AMRI from August 1997 to
January 2000. Dr. King received the Ph.D. degree of bio-organic chemistry from University of Utah in 1980, and B.S. degree of chemistry
from National Taiwan Normal University in 1972.
Family Relationships
There are no family relationships among the executive
officers and directors of the Company, except that Dr. Tsang Ming Jiang, Dr. Tsung-Shann Jiang and Dr. Chang-Jen Jiang are brothers,
Mr. Eugene Jiang is Dr. Tsung-Shann Jiang’s son, and the marital relationship between Yoshinobu Odaira and Norimi Sakamoto and
between Shuling Jiang and Dr. Jiang.
Legal Proceedings
Involvement in Certain Legal Proceedings
During the past ten years, none of our current
directors, executive officers, promoters, control persons, or nominees has been:
| |
● |
the subject of any bankruptcy
petition filed by or against any business of which such person was a general partner or executive officer either at the time of the
bankruptcy or within two years prior to that time; |
| |
● |
convicted in a criminal
proceeding or is subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); |
| |
● |
subject to any order, judgment,
or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction or any Federal or State authority,
permanently or temporarily enjoining, barring, suspending or otherwise limiting his involvement in any type of business, securities
or banking activities; |
| |
● |
found by a court of competent
jurisdiction (in a civil action), the Commission or the Commodity Futures Trading Commission to have violated a federal or state
securities or commodities law. |
| |
● |
the subject of, or a party
to, any Federal or State judicial or administrative order, judgment, decree, or finding, not subsequently reversed, suspended or
vacated, relating to an alleged violation of (a) any Federal or State securities or commodities law or regulation; (b) any law or
regulation respecting financial institutions or insurance companies including, but not limited to, a temporary or permanent injunction,
order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition
order; or (c) any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or |
| |
● |
the subject of, or a party
to, any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization (as defined in Section
3(a)(26) of the Exchange Act (15 U.S.C. 78c(a)(26))), any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange
Act (7 U.S.C. 1(a)(29))), or any equivalent exchange, association, entity or organization that has disciplinary authority over its
members or persons associated with a member. |
Unless disclosed otherwise, we are currently
not a party to any material legal or administrative proceedings and are not aware of any pending legal or administrative proceedings
against us. We may from time to time become a party to various legal or administrative proceedings arising in the ordinary course of
our business.
Director Independence
The NASDAQ Rules require that a majority of the
Board be independent. The Board consists of 11 directors, of which nine are non-management directors. Each year the Board reviews the
materiality of any relationship that each of our directors has with the Company, either directly or indirectly. No member of the Board
has any relationship or arrangement that would require disclosure under Item 404 of Regulation S-K. For additional information see “Certain
Relationships and Related-Party Transactions” in this report. Based on this review, the Board has determined that the following
current directors are “independent directors” as defined by the NASDAQ Rules: Messrs. Odaira and Chung and Mses. Sakamoto,
Chou and Miao.
Each director who is a member of the Audit and
Finance Committee, Compensation Committee and Nominating and Corporate Governance Committee is an independent director.
Board Committees
Audit Committee. The Audit Committee of
the Board of Directors currently consists of Ms. Chou, Yen-Hsin (Chair), Ms. Miao, Hsin-Hui, and Ms. Hsu, Che-Wei. The functions of the
Audit Committee include the retention of our independent registered public accounting firm, reviewing and approving the planned scope,
proposed fee arrangements and results of the Company’s annual audit, reviewing the adequacy of the Company’s accounting and
financial controls and reviewing the independence of the Company’s independent registered public accounting firm. The Board has
determined that Ms. Chou, Ms. Miao and Ms. Hsu are each an “independent director” under the listing standards of The NASDAQ
Stock Market. The Board of Directors has also determined Ms. Chou is an “audit committee financial expert” within the applicable
definition of the SEC. The Audit Committee is governed by a written charter approved by the Board of Directors, a copy of which is available
on our website at www.abvcpharma.com. Information contained on our website are not incorporated by reference into and do not form any
part of this reports. We have included the website address as a factual reference and do not intend it to be an active link to the website.
Compensation Committee. The Compensation
Committee of the Board of Directors currently consists of Ms. Norimi Sakamoto (Chair), Ms. Miao, Hsin-Hui, and Ms. Hsu, Che-Wei. The
functions of the Compensation Committee include the approval of the compensation offered to our executive officers and recommending to
the full Board of Directors the compensation to be offered to our directors, including our Chairman. The Board has determined that Ms.
Sakamoto, Ms. Miao and Ms. Hsu are each an “independent director” under the listing standards of The NASDAQ Stock Market
LLC. In addition, the members of the Compensation Committee qualify as “non-employee directors” for purposes of Rule 16b-3
under the Exchange Act and as “outside directors” for purposes of Section 162(m) of the Internal Revenue Code of 1986, as
amended. The Compensation Committee is governed by a written charter approved by the Board of Directors, a copy of which is available
on our website at www.abvcpharma.com. Information contained on our website are not incorporated by reference into and do not form any
part of this report. We have included the website address as a factual reference and do not intend it to be an active link to the website.
Corporate Governance and Nominating Committee.
The Corporate Governance and Nominating Committee of the Board of Directors consists of Mr. Yoshinobu Odaira (Chair), Ms. Miao, Hsin-Hui,
and Ms. Hsu, Che-Wei, each of whom is an independent director under Nasdaq’s listing standards. The corporate governance and nominating
committee is responsible for overseeing the selection of persons to be nominated to serve on our board of directors. The corporate governance
and nominating committee considers persons identified by its members, management, shareholders, investment bankers and others.
Guidelines for Selecting Director Nominees
The guidelines for selecting nominees, which
are specified in the Corporate Governance and Nominating Committee Charter, generally provide that persons to be nominated:
| |
● |
should have demonstrated
notable or significant achievements in business, education or public service; |
| |
● |
should possess the requisite
intelligence, education and experience to make a significant contribution to the board of directors and bring a range of skills,
diverse perspectives and backgrounds to its deliberations; and |
| |
● |
should have the highest
ethical standards, a strong sense of professionalism and intense dedication to serving the interests of the shareholders. |
The corporate governance and nominating committee
will consider a number of qualifications relating to management and leadership experience, background and integrity and professionalism
in evaluating a person’s candidacy for membership on the board of directors. The nominating committee may require certain skills
or attributes, such as financial or accounting experience, to meet specific board needs that arise from time to time and will also consider
the overall experience and makeup of its members to obtain a broad and diverse mix of board members. The board of directors will also
consider director candidates recommended for nomination by our shareholders during such times as they are seeking proposed nominees to
stand for election at the next annual meeting of shareholders (or, if applicable, a special meeting of shareholders). Our shareholders
that wish to nominate a director for election to the Board should follow the procedures set forth in our bylaws. The nominating committee
does not distinguish among nominees recommended by shareholders and other persons.
Board Leadership Structure and Role in Risk
Oversight
We have two separate individuals serving as our
CEO and Chairman. Our Board of Directors, or the Board, is primarily responsible for overseeing our risk management processes on behalf
of our company. The Board receives and reviews periodic reports from management, auditors, legal counsel, and others, as considered appropriate
regarding our company’s assessment of risks. In addition, the Board focuses on the most significant risks facing our company and
our company’s general risk management strategy, and also ensures that risks undertaken by our company are consistent with the board’s
appetite for risk. While the Board oversees our company’s risk management, management is responsible for day-to-day risk management
processes. We believe this division of responsibilities is the most effective approach for addressing the risks facing our company and
that our board leadership structure supports this approach.
Code of Ethics
We adopted a code of ethics, a copy of which
is attached herein as Exhibit 14.1. The Code of Ethics applies to all of our employees, officers and directors. This Code constitutes
a “code of ethics” as defined by the rules of the SEC. Copies of the code may be obtained free of charge from our website,
www.abvcpharma.com. Any amendments to, or waivers from, a provision of our code of ethics that applies to any of our executive officers
will be posted on our website in accordance with the rules of the SEC.
Indemnification
Neither our Articles of Incorporation nor Bylaws
prevent us from indemnifying our officers, directors and agents to the extent permitted under the Nevada Revised Statute (“NRS”).
NRS Section 78.7502 provides that a corporation shall indemnify any director, officer, employee or agent of a corporation against expenses,
including attorneys’ fees, actually and reasonably incurred by him in connection with any the defense to the extent that a director,
officer, employee or agent of a corporation has been successful on the merits or otherwise in defense of any action, suit or proceeding
referred to Section 78.7502(1) or 78.7502(2), or in defense of any claim, issue or matter therein.
Insofar as indemnification for liabilities arising
under the Securities Act of 1933 may be permitted to directors, officers or persons controlling the Company pursuant to Nevada law, we
are informed that in the opinion of the Securities and Exchange Commission, such indemnification is against public policy as expressed
in the Securities Act and is therefore unenforceable.
EXECUTIVE COMPENSATION
The following tables set forth, for each of the
last two completed fiscal years of us, the total compensation awarded to, earned by or paid to any person who was a principal executive
officer during the preceding fiscal year and every other highest compensated executive officers earning more than $100,000 during the
last fiscal year (together, the “Named Executive Officers”). The tables set forth below reflect the compensation of the Named
Executive Officers.
Summary Compensation Table
| Name and Principal Position |
|
Year |
|
|
Salary
($) |
|
|
Bonus
($) |
|
|
Stock
Awards
($) |
|
|
Option
Awards
($)(7) |
|
|
Non-Equity
Incentive Plan Compensation
($) |
|
|
Change
in Pension
Value and
Nonqualified Deferred Compensation Earnings
($) |
|
|
All
Other Compensation
($) |
|
|
Total
($) |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Howard Doong (1) |
|
|
2022 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
248,386 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
448,386 |
|
| |
|
|
2023 |
|
|
|
95,000 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
95,000 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Leeds Chow (2) |
|
|
2022 |
|
|
|
130,000 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
130,000 |
|
| |
|
|
2023 |
|
|
|
180,000 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
180,000 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Tsung-Shann Jiang (3) |
|
|
2022 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
248,386 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
448,386 |
|
| |
|
|
2023 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
200,000 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Richard Chi-Hsin King (4) |
|
|
2022 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
248,386 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
448,386 |
|
| |
|
|
2023 |
|
|
|
90,556 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
90,556 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eugene Jiang (5) |
|
|
2022 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
248,386 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
448,386 |
|
| |
|
|
2023 |
|
|
|
200,000 |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
200,000 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Chihliang An (6) |
|
|
2022 |
|
|
|
133,333 |
|
|
|
|
|
|
|
|
|
|
|
248,386 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
381,719 |
|
| |
|
|
2023 |
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Uttam Patil (1) |
|
|
2022 |
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
| |
|
|
2023 |
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
(1) |
Dr. Doong was appointed
as the CEO on September 15, 2017. Dr. Doong later resigned from his position as the Company’s CEO on June 21, 2023. The Company’s
board of directors appointed Dr. Uttam Patil to replace Dr. Doong as the Company’s CEO. |
| (2) |
Mr. Chow was appointed
as the CFO on September 4, 2022. |
| |
|
| (3) |
Dr. Jiang was appointed
as the CSTRO on September 1, 2019. Dr. Jiang was also appointed as the Company’s
CSO on June 15, 2023, to replace Dr. King, who resigned from his position as CSO. |
| |
|
| (4) |
Dr. King was appointed
as the CSO on September 15, 2017. Dr. King later resigned from his position as the
Company’s CSO on June 15, 2023. The Company’s board of directors appointed Dr. Jiang to replace Dr. King as the Company’s
CSO. |
| |
|
| (5) |
Eugene Jiang was appointed
as CBO on September 1, 2019. |
| |
|
| (6) |
Mr. An resigned from
his positions as the Company’s CFO on September 4, 2022. |
| |
|
| (7) |
The
weighted average grant date fair value of options granted during 2023 was $2.79, using the Black-Scholes option-pricing model. Accordingly,
the Company recognized stock-based compensation expense of $1,635,709 for the year ended December 31, 2023. There were no options
granted during 2023 |
Narrative Disclosure to Summary Compensation
Table
Other than set out below, there are no arrangements
or plans in which we provide pension, retirement or similar benefits for directors or executive officers. Our directors and executive
officers may receive share options at the discretion of our board of directors in the future. We do not have any material bonus or profit
sharing plans pursuant to which cash or non-cash compensation is or may be paid to our directors or executive officers, except that share
options may be granted at the discretion of our board of directors.
Stock Option Plan
Our board approved and adopted the Amended
and Restated 2016 Equity Incentive Plan on September 12, 2020 (the “Plan”), a copy of which is attached hereto as exhibit
10.17.
Grants of Plan-Based Awards
On April 16, 2022,
the Company entered into stock option agreements with 5 directors, pursuant to which the Company granted options to purchase an aggregate
of 761,920 shares of common stock under the Plan, as amended, at an exercise price of $3 per share. The options were vested at the grant
date and become exercisable for 10 years from the grant date.
As of the date of
this report, we have granted options under the Plan that can be exercised for an aggregate of 2,587,104 shares of Common Stock.
Outstanding Equity Awards at Fiscal Year End
The following table summarizes outstanding
unexercised options, unvested stocks and equity incentive plan awards held by each of our named executive officers, as of December 31,
2023:
OUTSTANDING EQUITY AWARDS AT FISCAL YEAR-END
| OPTION
AWARDS |
|
STOCK
AWARDS |
| Name |
|
Number
of Securities Underlying Unexercised Options (#) Exercisable |
|
|
Number
of Securities Underlying Unexercised Options (#) Unexercisable |
|
|
Equity
Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) |
|
|
Options
Exercise Prices ($) |
|
|
Option
Expiration Date |
|
Number
of Shares or Units of Stock That Have Not Vested (#) |
|
Market
Value of Shares or Units of Stock That Have Not Vested ($) |
|
|
Equity
Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights That Have Not Been Issued (#) |
|
|
Equity
Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Other Rights That Have Not Been Issued ($) |
|
| Howard Doong |
|
|
85,715 |
|
|
|
10,715 |
|
|
|
- |
|
|
|
2.00 |
|
|
Nov 20, 2031 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
|
|
400,001 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Oct 15, 2032 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
152,384 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Apr 16, 2033 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Chihliang An |
|
|
54,762 |
|
|
|
9,524 |
|
|
|
- |
|
|
|
2.00 |
|
|
Nov 20, 2031 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
233,334 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Oct 15, 2032 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
152,384 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Apr 16, 2033 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Tsung-Shann Jiang |
|
|
34,105 |
|
|
|
- |
|
|
|
- |
|
|
|
2.00 |
|
|
Nov 20, 2031 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
30,000 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Oct 15, 2032 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
152,384 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Apr 16, 2033 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Richard Chi-Hsin King |
|
|
82,144 |
|
|
|
14,286 |
|
|
|
- |
|
|
|
2.00 |
|
|
Nov 20, 2031 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
316,667 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Oct 15, 2032 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
152,384 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Apr 16, 2033 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Eugene Jiang |
|
|
72,418 |
|
|
|
12,193 |
|
|
|
- |
|
|
|
2.00 |
|
|
Nov 20, 2031 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
30,000 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Oct 15, 2032 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
152,384 |
|
|
|
- |
|
|
|
- |
|
|
|
3.00 |
|
|
Apr 16, 2033 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Uttam Patil |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Compensation of Directors
We did not pay stock options to directors
in fiscal year 2023.
Pension, Retirement or Similar Benefit Plans
There are no arrangements or plans in which we
provide pension, retirement or similar benefits for directors or executive officers. We have no material bonus or profit sharing plans
pursuant to which cash or non-cash compensation is or may be paid to our directors or executive officers, except that stock options may
be granted at the discretion of the board of directors or a committee thereof.
Employment Contracts
Dr. Uttam Patil has entered into an
employment agreement (“Patil Employment Agreement”) with the Company on June 23, 2023, pursuant to which he shall receive
the initial base salary by stock options in accordance with Company’s standard payroll practice. As of the date of this prospectus,
Dr. Patil has yet to receive any stock options.
On September 4, 2022, the Board appointed
Mr. Leeds Chow as the Company’s Chief Financial Officer (“CFO”) and Principal Accounting Officer effective from September
4, 2022 for a term of 3 years.
Dr. Chi-Hsin Richard King has entered into
an employment agreements (“King Employment Agreement”) with the Company, pursuant to which he shall receive an annual base
salary of $50,000. As of December 31, 2017, we paid Mr. King 10,416 shares of the Company’s common stock at a per share price of
$1.60 as opposed to cash compensation. Under King Employment Agreement, Dr. King is employed as the CSO of the Company. We may terminate
the employment for cause, at any time, without notice or remuneration, for certain acts of the executive officer, such as conviction
or plea of guilty to a felony or grossly negligent or dishonest acts to our detriment, or misconduct or a failure to perform agreed duties.
In such case, the executive officer will not be entitled to receive payment of any severance benefits or other amounts by reason of the
termination, and the executive officer’s right to all other benefits will terminate, except as required by any applicable law.
We may also terminate an executive officer’s employment without cause upon one-month advance written notice. In such case of termination
by us, we are required to provide compensation to the executive officer, including severance pay equal to 12 months of base salary. The
executive officer may terminate the employment at any time with a one-month advance written notice if there is any significant change
in the executive officer’s duties and responsibilities or a material reduction in the executive officer’s annual salary.
In such case, the executive officer will be entitled to receive compensation equivalent to 12 months of the executive officer’s
base salary. On August 21, 2019, all of the Board members present at the Meeting, unanimously reelected Dr. Richard King as the Chief
Scientific Officer (“CSO”), which became effective on September 1, 2019 for a term of three years. On
June 13, 2023, Dr. Richard King resigned from his position as the CSO. The Company’s board of directors appointed Dr. Jiang to
replace Dr. Richard King as the CSO.
On August 21, 2019, all of the Board members
present at the Meeting, except Eugene Jiang, appointed Mr. Eugene Jiang, the current Chairman of the Board, as the Chief Business Officer,
effective since September 1, 2019 for a term of three years. Mr. Eugene Jiang excused himself from the discussion regarding his appointment
as the Chief Business Officer of the Company during the Board meeting. The contract was renewed for another three years.
On August 21, 2019, all of the Board members
present at the Meeting, except Dr. Tsung-Shann Jiang, reelected Dr. Tsung-Shann Jiang as the Chief Strategy Officer, effective since
September 1, 2019 for a term of three years. Dr. Tsung-Shann Jiang excused himself from the discussion regarding his appointment as the
Chief Strategy Officer of the Company during the Board meeting. The contract was renewed for another three years.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information regarding beneficial ownership of our common stock as of the date hereof (i) each person (or group of affiliated persons) who is known by us to own more than five percent (5%) of the outstanding shares of our Common Stock, (ii) each director, executive officer and director nominee, and (iii) all of our directors, executive officers and director nominees as a group.
Beneficial ownership is determined in accordance with SEC rules and generally includes voting or investment power with respect to securities. For purposes of this table, a person or group of persons is deemed to have “beneficial ownership” of any shares of common stock that such person has the right to acquire within 60 days of the date of the respective table. For purposes of computing the percentage of outstanding shares of our common stock held by each person or group of persons named above, any shares that such person or persons has the right to acquire within 60 days of the date of the respective table is deemed to be outstanding for such person, but is not deemed to be outstanding for the purpose of computing the percentage ownership of any other person. The inclusion herein of any shares listed as beneficially owned does not constitute an admission of beneficial ownership.
Unless otherwise noted, the business address of each beneficial owner listed is 44370 Old Warm Springs Blvd., Fremont, CA 94538. Except as otherwise indicated, the persons listed below have sole voting and investment power with respect to all shares of our common stock owned by them, except to the extent that power may be shared with a spouse.
As of March 12, 2024, we had 10,560,421 shares
of common stock issued and outstanding.
| Name of Beneficial Owner | |
Amount and
Nature of
Beneficial
Ownership | | |
Percent of
Class | |
| Dr. Uttam Patil | |
| 72,428 | | |
| * | |
| Eugene Jiang (1) | |
| 147,373 | | |
| 1.4 | % |
| Leeds Chow | |
| 52,007 | | |
| * | |
| Yen-Hsin Chou | |
| 41,956 | | |
| * | |
| Hsin-Hui Miao | |
| 48,072 | | |
| * | |
| Dr. Tsang-Ming Jiang | |
| 41,994 | | |
| * | |
| Norimi Sakamoto | |
| 41,854 | | |
| * | |
| Dr. Tsung-Shann Jiang (2)(4) | |
| 590,843 | | |
| 5.6 | % |
| Dr. Chang-Jen Jiang (3) | |
| 42,082 | | |
| * | |
| Yoshinobu Odaira | |
| 57,758 | | |
| * | |
| Che -Wei Hsu | |
| 41,723 | | |
| * | |
| Shuling Jiang | |
| 1,628,464 | | |
| 15.4 | % |
| Yu-Min Chung | |
| 41,943 | | |
| * | |
| All officers and directors as a group (Fourteen (14) persons) | |
| 2,848,497 | | |
| 27.0 | % |
| YuanGene Corporation (4) | |
| 829,699 | | |
| 7.9 | % |
|
(1) |
Eugene Jiang held 147,373
shares through direct ownership. |
|
(2) |
Dr. Tsung-Shann Jiang
held 167,599 shares of common stock through his ownership in YuanGene Corporation, 722 shares through Rgene Corporation, 608 shares
through BioFirst, 45 shares through BioLite, 3,227 shares through Lion Arts, and the rest of 418,642 shares through direct ownership. |
|
(3) |
Dr. Chang-Jen Jiang
held 234 shares of common stock in the Company through his ownership in BioFirst, 1 share through Rgene, and the rest of 41,847 shares
through direct ownership. |
| (4) |
Ms. Shuling Jiang held
662,100 shares of common stock through her ownership in YuanGene Corporation, 964 shares through Rgene Corporation, 8,833 shares
through BioFirst, 182 shares through BioLite, 48,761 shares through Liongene, 21,313 shares through Keypoint, 1,012 shares through
Genepro, 12,747 shares through Lion Arts, and the rest of 872,552 shares through direct ownership. |
|
(5) |
YuanGene Corporation
is a company wholly-owned by Lion Arts, which is owned by Shu-Ling Chiang (80%) and Dr. Tsung-Shann Jiang (20%); however, YuanGene
appointed Eugene Jiang to have sole voting control over the shares held by YuanGene, the principal office address of which is 2nd
floor, Building B, SNPF Plaza, Savalalo, Apia, Samoa. |
RELATED PARTY TRANSACTIONS OF DIRECTORS AND EXECUTIVE OFFICERS
Except as disclosed
herein, no director, executive officer, shareholder holding at least 5% of shares of our common stock, or any family member thereof,
had any material interest, direct or indirect, in any transaction, or proposed transaction since January 1, 2022, in which the amount
involved in the transaction exceeds the lesser of $120,000 or one percent of the average of our total assets at the year-end for the
last two completed fiscal years.
Co-Development agreement with Rgene Corporation
On November 10, 2020, the Company and Rgene signed an amendment to the Co-Dev Agreement dated May 26, 2017, pursuant to which both parties agreed to delete AB-1507 HER2/neu Positive Breast Cancer Combination Therapy and AB 1527 Ovary Cancer Combination Therapy and add ABV-1519 EGFR Positive Non-Small Cell Lung Cancer Combination Therapy and ABV-1526 Large Intestine / Colon / Rectal Cancer Combination Therapy to the products to be co-developed and commercialized. Other provisions of the Co-Dev Agreement remain in full force and effect.
Clinical Development Service Agreement
with Rgene Corporation
On June 10, 2022, the Company expanded its
co-development partnership with Rgene. BioKey, Inc. entered into a Clinical Development Service Agreement with Rgene (“Service
Agreement”) to guide certain Rgene drug products, RGC-1501 for the treatment of Non-Small Cell Lung Cancer (NSCLC), RGC-1502 for
the treatment of pancreatic cancer and RGC 1503 for the treatment of colorectal cancer patients, through completion of Phase II clinical
studies under U.S. FDA IND regulatory requirements (the “Rgene Studies”). The Service Agreement shall remain in effect until
the expiration date of the last patent and automatically renew for 5 more years unless terminated earlier by either party with six months
written notice. Under the terms of the Service Agreement, BioKey is eligible to receive payments totaling up to $3.0 million over a 3-year
period with each payment amount to be determined by certain regulatory milestones obtained during the agreement period.
Collaborative agreement with BioFirst Corporation
On November 4, 2020, we executed an amendment
to our collaboration agreement with BioFirst dated July 24, 2017, to add ABV-2001 Intraocular Irrigation Solution and ABV-2002 Corneal
Storage Solution to our agreement. ABV-2002 is intended to be utilized during a corneal transplant procedure to replace a damaged or
diseased cornea while ABV-2001 has broader utilization during a variety of ocular procedures.
Initially ABVC will focus on ABV-2002, a solution
utilized to store a donor cornea prior to either penetrating keratoplasty (full thickness cornea transplant) or endothelial keratoplasty
(back layer cornea transplant). Designated ABV-2002 under ABVC’s product identification system, the solution is comprised of a
specific poly amino acid that protects ocular tissue from damage caused by external osmolarity exposure during pre-surgery storage. The
specific polymer in ABV 2002 can adjust osmolarity to maintain a range of 330 to 390 mOsM thereby permitting hydration within the corneal
stroma during the storage period. Stromal hydration results in (a) maintaining acceptable corneal transparency and (b) prevents donor
cornea swelling. ABV-2002 also contains an abundant phenolic phytochemical found in plant cell walls that provides antioxidant antibacterial
properties and neuroprotection.
Early testing by BioFirst indicates that ABV-2002
may be more effective for protecting the cornea and retina during long-term storage than other storage media available today and can
be manufactured at lower cost. ABV-2002 is categorized as a Class I Medical Device that has the lowest risk to patients; however, further
clinical development was put on hold due to the lack of funding.
On May 11, 2018, the Company and BioFirst
(Australia) entered into a loan agreement for a total amount of $40,000 to meet its working capital needs. The advances bear 0% interest
rate and are due on demand prior to September 30, 2020. Afterwards, all outstanding load will bear interest rate at 12% per annum. On
July 1, 2020, the Company entered into a loan agreement with BioFirst (Australia) for $361,487 to properly record R&D cost and tax
refund allocation based on co-development contract executed on July 24, 2017. The loan was originally set to mature on September 30,
2021 with an interest rate of 6.5% per annum, however, on September 7, 2021, the Company entered into a loan agreement with BioFirst
(Australia) for $67,873 to meet its new project needs. On December 1, 2021, the Company entered into a loan agreement with BioFirst (Australia)
for $250,000 to increase the cost for upcoming projects. The loan has an interest rate of 6.5% per annum and matured on November 30,
2022. As of December 31, 2022 and 2021, the aggregate amount of outstanding loans and accrued interest was $1,028,556 and $491,816, respectively.
Joint Venture Agreement
On October 6, 2021 (the “Completion
Date”), the Company, Lucidaim Co., Ltd., a Japanese corporation (“Lucidaim,” together with the Company,
the “Shareholders”), and BioLite Japan K.K., a Japanese corporation (“Biolite JP”) entered into
a Joint Venture Agreement (the “Agreement”). Biolite JP is a private limited company (a Japanese Kabushiki Kaisha)
incorporated on December 18, 2018 and at the date of the Agreement has 10,000 ordinary shares authorized, with 3,049 ordinary shares
issued and outstanding (the “Ordinary Shares”). Immediately prior to the execution of the Agreement, Lucidaim owned
1,501 Ordinary Shares and the Company owned 1,548 Ordinary Shares. The Shareholders entered into the joint venture to formally reduce
to writing their desire to invest in and operate Biolite JP as a joint venture. The business of the joint venture shall be the research
and development of drugs, medical device and digital media, investment, fund running and consulting, distribution and marketing of supplements
carried on by Biolite JP and its subsidiaries in Japan, or any other territory or businesses as may from time to time be agreed by an
amendment to the Agreement. The closing of the transaction is conditioned upon the approval and receipt of all necessary government approvals,
which have been received.
Pursuant to the Agreement and the related share transfer agreement, the Company shall transfer 54 of its Ordinary Shares to Lucidaim for no consideration, such that following the transfer, Lucidaim shall own 1,555 Ordinary Shares (51%) and the Company shall own 1,494 Ordinary Shares (49%). Also pursuant to the Agreement, there shall be 3 directors of Biolite JP, consisting of 1 director appointed by the Company and 2 appointed by Lucidiam. The Company shall appoint Eugene Jiang, the Company’s current Chairman and Chief Business Officer and Lucidaim shall appoint Michihito Onishi; the current director of Biolite JP, Toru Seo (who is also a director of BioLite Japan’s other shareholder), is considered the second Lucidaim director. The Agreement further provides that the Company and Biolite JP shall assign the research collaboration and license agreement between them to Biolite JP or prepare the same (the “License Agreement”). The aforementioned transactions occurred on the Completion Date.
As per the Agreement, the Shareholders shall
supervise and manage the business and operations of Biolite JP. The directors shall not be entitled to any renumeration for their services
as a director and each Shareholder can remove and replace the director he/she/it appointed. If a Shareholder sells or disposes of all
of its Ordinary Shares, the director such Shareholder appointed must tender his/her resignation. The Agreement also sets forth certain
corporate actions that must be pre-approved by all Shareholders (the “Reserved Matters”). If the Shareholders are
unable to make a decision on any Reserved Matter, then either Shareholder can submit a deadlock notice to the other shareholder, 5 days
after which they must refer the matter to each Shareholder’s chairman and use good faith to resolve the dispute. If such dispute
is not resolved within 10 days thereafter, then either Shareholder can offer to buy all of the other Shareholder’s Ordinary Shares
for cash at a specified price; if there is not affirmative acceptance of the sale, the sale shall proceed as set forth in the sale offer.
Each of the Shareholders maintains a pre-emptive right to purchase such number of additional Ordinary Shares as would allow such Shareholder to maintain its ownership percentage in Biolite JP if Biolite JP issues any new Ordinary Shares. However, the Agreement provides that the Company shall lose its pre-emptive rights under certain conditions. The Shareholders also maintain a right of first refusal if the other Shareholder receives an offer to buy such shareholder’s Ordinary Shares.
The Agreement also requires Biolite JP to obtain a bank facility in the amount of JPY 30,460,000 (approximately USD272,000), for its initial working capital purposes. Pursuant to the Agreement, each Shareholder agrees to guarantee such bank facility if the bank requires a guarantee. Accordingly, the Company may be liable for the bank facility in an amount up to JPY 14,925,400 (approximately USD134,000), which represents 49% of the maximum bank facility. The Agreement further provides that Biolite JP shall issue annual dividends at the rate of at least 1.5% of Biolite JP’s profits, if it has sufficient cash to do so.
Pursuant to the Agreement, the Company and Biolite JP agree to use their best efforts to execute the License Agreement by the end of December 2021, but since it was not yet executed, the parties continue such efforts. The Company agreed that any negotiation on behalf of Biolite JP regarding the terms of the License Agreement shall be handled by the directors appointed by Lucidaim. If the Company and such Lucidaim directors do not reach agreement on the terms, Biolite JP may at its sole discretion determine not to execute the License Agreement without any liability to the Company.
The Agreement contains non-solicitation and non-compete clauses for a period of 2 years after a Shareholder or its subsidiaries ceases to be a Shareholder, with such restrictive covenants limited to business within the ophthalmologic filed or central neurological field. Any rights to intellectual property that arise from Biolite JP’s activities, shall belong to Biolite JP.
The Agreement contains standard indemnification terms, except that no indemnifying party shall have any liability for an individual liability unless it exceeds JPY 500,000 (approximately USD4,500) and until the aggregate amount of all liabilities exceeds JPY 2,000,000 (approximately USD18,000) and then only to the extent such liability exceed such limit.
The Company paid $150,000 towards the setup of the joint venture; BioLite Japan’s other shareholder also paid $150,000 after the Letter of Intent was signed.
The Agreement shall continue for 10 years, unless earlier terminated. The Agreement also allows a Shareholder to terminate the agreement upon certain defaults committed by another Shareholder, as set forth in the Agreement.
Agreement with BioLite, Inc.
We entered into a Collaborative Agreement with BioLite, Inc., a company incorporated under the laws of Taiwan, and a subsidiary of the Company, (“BioLite”) on December 29, 2015, and then entered into two addendums to such agreement (as amended and revised, (the “Agreement”). The majority shareholder of BioLite is one of the Company’s subsidiaries, the Company’s Chairman is a director of BioLite and Dr. Jiang, the Company’s Chief Strategy Officer and a director, is the Chairman of BioLite.
Pursuant
to the Agreement, the Company acquired the sole licensing rights to develop and commercialize for therapeutic purposes six compounds
from BioLite. In accordance with the terms of the Agreement, the Company shall pay BioLite (i) milestone payments of up to $100 million
in cash and equity of the Company or equity securities owned by it at various stages on a schedule dictated by BioLite’s achievements
of certain milestones, as set forth in the Agreement (the “Milestone Payments”) and (ii) a royalty payment equal to 5% of
net sales of the drug products when ABV-1501 is approved for sale in the licensed territories. If BioLite fails to reach any of the milestones
in a timely manner, it may not receive the rest of the payments from the Company. According to the Agreement, after Phase II clinical
trials are completed, 15% of the Milestone Payment becomes due and shall be paid in two stages: (i) 5% no later than December 31, 2021
(the “December 2021 Payment”) and (ii) 10% no later than December 31, 2022. On February 12, 2022, the Company’s Board
of Directors determined that the December 2021 Payment, which is equal to $5,000,000, shall be paid via the cancellation of certain outstanding
debt, in the amount of $5,000,000, that BioLite owes the Company as of December 31, 2021. On February 22, 2022, the parties entered into
an amendment to the Agreement allowing the Company to make all payments due under the Agreement via the forgiveness of debt, in equal
value, owed by BioLite to the Company.
This was a related party transaction and was
conducted at arm’s length. In addition to the Company’s board of directors approving the modification of terms of the Agreement,
the Company’s audit committee approved them too. The Board believes it is in the Company’s best interest to cancel outstanding
debt and apply it to the December 2021 Payment.
Following such approval, the Company and BioLite
entered into an amendment to the Agreement reflecting the modified payment method.
Real Estate Purchase
On February 6, 2024, the Company entered into
a definitive agreement with Shuling Jiang (“Shuling”), pursuant to which Shuling shall transfer the ownership of certain
land she owns located at Taoyuan City, Taiwan (the “Land”) to the Company (the “Agreement”). Shuling
is a director of the Company, is married to TS Jiang, the Company’s Chief Strategic Officer and owns approximately 15.4% of the
Company’s issued and outstanding shares of common stock.
In consideration for the Land, the Company
shall pay Shuling (i) 703,495 restricted shares of the Company’s common stock (the “Shares”) at a price of $3.50
per share and (ii) five-year warrants to purchase up to 1,000,000 shares of the Company’s common stock, with an exercise price
of $2.00 per share. Under the Agreement, Shuling will also transfer outstanding liability owed on the Land (approximately $500,000) to
the Company. Thus, the parties value the exchange at approximately $2,962,232.
Other related party transactions
Due from related parties:
| (1) |
On June 16, 2022, the Company entered
into a one-year convertible loan agreement with Rgene, with a principal amount of $1,000,000 to Rgene which bears interest at 5%
per annum for the use of working capital that, if fully converted, would result in ABVC owning an additional 6.4% of Rgene. The Company
may convert the Note at any time into shares of Rgene’s common stock at either (i) a fixed conversion price equal to $1.00
per share or (ii) 20% discount of the stock price of the then most recent offering, whichever is lower; the conversion price is subject
to adjustment as set forth in the Note. The Note includes standard events of default, as well as a cross-default provision pursuant
to which a breach of the Service Agreement will trigger an event of default under the convertible note if not cured after 5 business
days of written notice regarding the breach is provided.
As of December 31, 2023, the outstanding
loan balance was $700,000; and accrued interest was $45,573. |
| (2) |
In
2022, the Company entered into several loan agreements with BioFirst (Australia) for a total amount of $507,000 to increase the cost
for upcoming projects. All the loans period was twelve months with an interest rate of 6.5% per annum. As of December 31, 2023 and
2022, the outstanding loan balance and allocated research fee was $0 and $660,484, respectively; and accrued interest was $0 and
$92,171, respectively. The outstanding amount was settled in 2023. |
Due to related parties:
| (1) |
Since
2019, BioFirst has advanced funds to the Company for working capital purpose. The advances bear interest 1% per month (or equivalent
to 12% per annum). As of December 31, 2022, the aggregate amount of outstanding balance and accrued interest is $188,753, a combination
of $147,875 from loan, and $40,878 from expense-sharing. The outstanding amount was being settled in 2023. |
| (2) |
Since
2019, the Jiangs advanced funds to the Company for working capital purpose. As of December 31, 2023 and 2022, the outstanding balance
due to the Jiangs amounted to $20,750 and $19,789, respectively. These loans bear interest rate of 0% to 1% per month, and are due
on demand. |
| (3) |
Since
2018, the Company’s shareholders have advanced funds to the Company for working capital purpose. The advances bear interest
rate from 12% to 13.6224% per annum. As of December 31, 2023 and 2022, the outstanding principal and accrued interest was $152,382
and $151,450, respectively. Interest expenses in connection with these loans were $20,094 and $21,378 for the years ended December
31, 2023 and 2022, respectively. |
Promoters and Certain Control Persons
None of our management or other control persons were “promoters” (within the meaning of Rule 405 under the Securities Act), and none of such persons took the initiative in the formation of our business or received any of our debt or equity securities or any of the proceeds from the sale of such securities in exchange for the contribution of property or services, during the last five years.
DESCRIPTION OF SECURITIES
General
The Company’s authorized capital stock consists of:
| |
● |
100,000,000 shares of Common Stock, $0.001 par value per share; and |
| |
● |
20,000,000 shares of preferred stock, $0.001 par value per share. |
Our Common Stock may be issued for such consideration as may be fixed from time to time by our board of directors. Our board of directors may issue such shares of our Common Stock in one or more series, with such voting powers, shall be stated in the resolution or resolutions.
Common Stock
As of the date hereof, there are 10,851,823
shares of our Common Stock issued and outstanding. Holders of Common Stock are entitled to cast one vote for each share on all matters
submitted to a vote of shareholders, including the election of directors. The holders of Common Stock are entitled to receive ratably
such dividends, if any, as may be declared by the Board out of funds legally available therefore. Such holders do not have any preemptive
or other rights to subscribe for additional shares. All holders of Common Stock are entitled to share ratably in any assets for distribution
to shareholders upon the liquidation, dissolution or winding up of the Company, subject to prior distribution rights of preferred stock
then outstanding. There are no conversions, redemptions or sinking fund provisions applicable to the Common Stock. All outstanding shares
of Common Stock are fully paid and non-assessable.
Preferred Stock
As of the date hereof, there is no preferred stock outstanding, including the class of Series A Convertible Preferred Stock. Pursuant to the articles of incorporation of the Company, the Board of Directors is expressly granted the authority to issue preferred stock up to 20,000,000 shares and prescribe its designations. On June 28, 2019, the Company filed a certificate of designation (the “Series A COD”) of Series A Convertible Preferred Stock (the “Series A Stock”) with the Secretary of the State of Nevada, pursuant to which the Company designated 3,500,000 shares of preferred stock as Series A Stock, par value of $0.001 per share.
The following description of the Series A Stock is not complete. The Company’s Board of Directors has the authority, without further action by the shareholders, to issue shares of preferred stock in one or more other series and to fix the rights, preferences, privileges and restrictions granted to or imposed upon the preferred stock. Any or all of these rights may be greater than the rights of the Company’s Common Stock. These descriptions are qualified in their entirety by reference to the Company’s Articles of Incorporation, as amended, and the certificate of designation relating to each such series.
Conversion Rights
Each share of Series A Stock is initially convertible at any time at the option of the holders into one share of Common Stock and automatically converts into one share of Common Stock (the “Conversion Ratio”) on its four-year anniversary of issuance and without the payment of additional consideration by the holder thereof.
No fractional shares shall be issued upon conversion of Series A Stock into Common Stock and no payment. In lieu of delivering fractional shares, we will pay to the holder, to the extent permitted by law, an amount in cash equal to the current fair market value of such fractional share as determined in good faith by our Board.
No Maturity, Sinking Fund or Mandatory Redemption
The Series A Stock has no maturity date and we are not required to redeem the Series A Stock at any time. However, we may choose to convert all the outstanding shares of the Series A Stock into our Common Stock at the same Conversion Ratio at any time, provided that we have prepaid and distributed all the dividend accrued and to be accrued at the end of the four-year period since issuance thereof. Accordingly, the Series A Stock will remain outstanding until automatically converted to Common Stock on the four-year anniversary of issuance, unless the holders of the Series A Convertible Preferred Stock or we choose to convert the Series A Stock into the Common Stock. The Series A Stock is also not subject to any sinking fund.
Voting Rights
Holders of shares of the Series A Stock vote on an as converted basis with the holders of our Common Stock.
Holders of shares of Series A Stock were entitled to receive a cash dividend at the per annum rate of an amount equal to the product of 5% multiplied by the public offering price per share of the shares that were to be sold pursuant to the registration statement on Form S-1 (File No. 333-228387) initially filed on November 14, 2018, but has since been withdrawn. Therefore, the Series A Stock is not eligible for dividends at this time.
Warrants and Options
As of the date hereof, we have 1,307,102 and
7,038,442 options and warrants, respectively of the Company outstanding. We are not registering shares of common stock underlying any
warrants in this S1.
August 2021 Follow-on Offering
On August 2, 2021, we closed a firm commitment underwritten public offering pursuant to which we issued and sold 1,100,000 units (the “Units”), with each Unit consisting of one share of the Company’s common stock, par value $0.001 per share (the “Common Stock”), one Series A warrant (the “Series A Warrants”) to purchase one share of common stock at an exercise price equal to $6.30 per share, exercisable until the fifth anniversary of the issuance date, and one Series B warrant (the “Series B Warrants,” and together with the Series A Warrants, the “Public Warrants”) to purchase one share of common stock at an exercise price equal to $10.00 per share, exercisable until the fifth anniversary of the issuance date; the exercise price of the Public Warrants are subject to certain adjustment and cashless exercise provisions as described therein. The Company completed the Offering pursuant to its registration statement on Form S-1 (File No. 333-255112), originally filed with the Securities and Exchange Commission (the “SEC”) on April 8, 2021 (as amended, the “Original Registration Statement”), that the SEC declared effective on August 2, 2021 and the registration statement on Form S-1 (File No. 333-258404) that was filed and automatically effective on August 4, 2021 (the “S-1MEF,” together with the Original Registration Statement, the “Registration Statement”). The Units were priced at $6.25 per Unit, before underwriting discounts and offering expenses, resulting in gross proceeds of $6,875,00
The following summary of certain terms and provisions of the warrants issued pursuant to the August 2021 public offering is not complete and is subject to, and qualified in its entirety by, the provisions of the warrant agent agreement between us and Vstock, as warrant agent, and the form of warrants, all of which are filed as exhibits to the registration statement of which this prospectus is a part. Prospective investors should carefully review the terms and provisions set forth in the warrant agent agreement, including the annexes thereto, and form of Purchase Warrant.
Series A Warrants
The Series A Warrants entitle the registered holder to purchase one share of our common stock at a price equal to $6.30 per share, subject to adjustment as discussed below, terminating at 5:00 p.m., New York City time, on the fifth (5th) anniversary of the date of issuance.
The exercise price and number of shares of common stock issuable upon exercise of the Series A Warrants may be adjusted in certain circumstances, including in the event of a stock dividend, extra common dividend on or recapitalization, reorganization, merger or consolidation.
The Series A Warrants may be exercised upon surrender of the warrant certificate on or prior to the expiration date at the offices of the Warrant Agent, with the exercise form attached to the warrant certificate completed and executed as indicated, accompanied by full payment of the exercise price, by certified or official bank check payable to us, for the number of warrants being exercised. The Series A Warrant holders do not have the rights or privileges of holders of common stock and any voting rights until they exercise their Series A Warrants and receive shares of common stock. After the issuance of shares of common stock upon exercise of the Series A warrants, each holder will be entitled to one vote for each share held of record on all matters to be voted on by stockholders.
No Series A Warrants will be exercisable for cash unless at the time of the exercise a prospectus or prospectus relating to common stock issuable upon exercise of the Series A Warrants is current and the common stock has been registered or qualified or deemed to be exempt under the securities laws of the state of residence of the holder of the warrants. Under the terms of the Series A Warrant Agent Agreement, we have agreed to use our best efforts to maintain a current prospectus or prospectus relating to common stock issuable upon exercise of the Series A Warrants until the expiration of the Series A Warrants. Additionally, the market for the Series A Warrants may be limited if the prospectus or prospectus relating to the common stock issuable upon exercise of the Series A Warrants is not current or if the common stock is not qualified or exempt from qualification in the jurisdictions in which the holders of such Series A Warrants reside. In no event will the registered holders of a Series A Warrant be entitled to receive a net-cash settlement in lieu of physical settlement in shares of our common stock.
No fractional shares of common stock will be issued upon exercise of the Series A Warrants. If, upon exercise of the Series A Warrants, a holder would be entitled to receive a fractional interest in a share, we will, upon exercise, round up to the nearest whole number the number of shares of common stock to be issued to the Warrant holder. If multiple Series A Warrants are exercised by the holder at the same time, we will aggregate the number of whole shares issuable upon exercise of all the Series A Warrants.
The price of the Series A Warrants has been arbitrarily established by us and the Underwriter after giving consideration to numerous factors, including but not limited to, the pricing of the Units in this offering. No particular weighting was given to any one aspect of those factors considered. We have not performed any method of valuation of the warrants.
The Series A Warrant form contains a governing law and forum selection provision which provides that, subject to applicable law, any action, proceeding or claim against us or the warrant agent arising out of or relating in any way to the warrant agreement shall be brought and enforced in the state and federal courts sitting in the City of New York, Borough of Manhattan, and that each party irrevocably submits to such jurisdiction, which jurisdiction shall be the exclusive forum for any such action, proceeding or claim. Each party to the Series A Warrant irrevocably waives personal service of process and consents to process being served in any such suit, action or proceeding by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to such party at the address in effect for notices to it under the warrant and agrees that such service shall constitute good and sufficient service of process and notice thereof. Notwithstanding the foregoing, the exclusive forum provision described above shall not apply to suits brought to enforce a duty or liability created by the Exchange Act, any other claim for which the federal courts have exclusive jurisdiction or any complaint asserting a cause of action arising under the Securities Act against us or any of our directors, officers, other employees or agents. Section 27 of the Exchange Act creates exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder.
Series B Warrants
The Series B Warrants entitle each holder to purchase one share of our common stock at an exercise price of $10.00 per share, subject to adjustment as discussed below, terminating at 5:00 p.m., New York City time, on the fifth (5th) anniversary of the date of issuance.
The exercise price and number of shares of common stock issuable upon exercise of the Series B Warrants may be adjusted in certain circumstances, including in the event of a stock dividend, extra common dividend on or recapitalization, reorganization, merger or consolidation.
The Series B Warrants may be exercised upon surrender of the warrant certificate on or prior to the expiration date at the offices of the Warrant Agent, with the exercise form attached to the warrant certificate completed and executed as indicated, accompanied by full payment of the exercise price, by certified or official bank check payable to us, for the number of warrants being exercised. The Series B Warrant holders do not have the rights or privileges of holders of common stock and any voting rights until they exercise their Series B Warrants and receive shares of common stock. After the issuance of shares of common stock upon exercise of the Series B warrants, each holder will be entitled to one vote for each share held of record on all matters to be voted on by stockholders.
No Series B Warrants will be exercisable for cash unless at the time of the exercise a prospectus or prospectus relating to common stock issuable upon exercise of the Series B Warrants is current and the common stock has been registered or qualified or deemed to be exempt under the securities laws of the state of residence of the holder of the warrants. Under the terms of the Series B Warrant Agent Agreement, we have agreed to use our best efforts to maintain a current prospectus or prospectus relating to common stock issuable upon exercise of the Series B Warrants until the expiration of the Series B Warrants. Additionally, the market for the Series B Warrants may be limited if the prospectus or prospectus relating to the common stock issuable upon exercise of the Series B Warrants is not current or if the common stock is not qualified or exempt from qualification in the jurisdictions in which the holders of such Series B Warrants reside. In no event will the registered holders of a Series B Warrant be entitled to receive a net-cash settlement in lieu of physical settlement in shares of our common stock.
No fractional shares of common stock will be issued upon exercise of the Series B Warrants. If, upon exercise of the Series B Warrants, a holder would be entitled to receive a fractional interest in a share, we will, upon exercise, round up to the nearest whole number the number of shares of common stock to be issued to the Warrant holder. If multiple Series B Warrants are exercised by the holder at the same time, we will aggregate the number of whole shares issuable upon exercise of all the Series B Warrants.
The price of the Series B Warrants has been arbitrarily established by us and the Underwriter after giving consideration to numerous factors, including but not limited to, the pricing of the Units in this offering. No particular weighting was given to any one aspect of those factors considered. We have not performed any method of valuation of the warrants.
If we fail to maintain a current prospectus or prospectus relating to the common stock issuable upon the exercise of the Series B Warrants, such holders may exercise their Series B warrants on a “cashless” basis pursuant to a formula set forth in the terms of the Series B Warrants. Additionally, holders of Series B Warrants may exercise such warrants on a “cashless” basis upon the earlier of (i) 15 trading days from the issuance date of such warrant or (ii) the time when $10.0 million of volume is traded in our common stock, if the volume weighted average price (“VWAP”) of our common stock on any trading day on or after the date of issuance fails to exceed the then current exercise price of the Series B Warrant (subject to adjustment for any stock splits, stock dividends, stock combinations, recapitalizations and similar events). In such event, the aggregate number of shares of common stock issuable in such cashless exercise shall equal the product of (x) the aggregate number of shares of common stock that would be issuable upon exercise of the Series B Warrant in accordance with its terms if such exercise were by means of a cash exercise rather than a cashless exercise and (y) 1.00.
Warrant Agent
The Series A Warrants and Series B Warrants will be issued in registered form under a warrant agent agreement (the “Warrant Agent Agreement”) between us and our warrant agent, VStock Transfer, LLC (the “Warrant Agent”). The Purchase Warrants will be issued in book-entry form and shall initially be represented only by one or more global warrants deposited with the warrant agent, as custodian on behalf of The Depository Trust Company, or DTC, and registered in the name of Cede & Co., a nominee of DTC, or as otherwise directed by DTC. The material provisions of the warrants are set forth herein and a copy of the Warrant Agent Agreement has been filed as an exhibit to the Registration Statement on Form S-1, of which this prospectus forms a part. The Company and the Warrant Agent may amend or supplement the Warrant Agent Agreement without the consent of any holder for the purpose of curing any ambiguity, or curing, correcting or supplementing any defective provision contained therein or adding or changing any other provisions with respect to matters or questions arising under the Warrant Agent Agreement as the parties thereto may deem necessary or desirable and that the parties determine, in good faith, shall not adversely affect the interest of the Series A Warrant or Series B Warrant holders. All other amendments and supplements to the Warrant Agent Agreement shall require the vote or written consent of holders of at least 50.1% of each of the Series A Warrants and Series B Warrants.
Representative’s Warrants
We issued the Representative (or its designed affiliates) share purchase warrants (the “Representative’s Warrants”) to purchase up to 55,000 shares of Common Stock at an exercise price equal to $6.25 per share. The Representative’s Warrants are not exercisable for six (6) months after the effective date of the registration statement of which this prospectus forms a part and will expire five (5) years after such effective date. The Representative’s Warrants are not be redeemable. The Representative’s Warrants have “piggy back” registration rights for a period of five (5) years from the effective date of the registration of which this prospectus forms a part in accordance with FINRA Rule 5110(g)(8)(D). The Representative’s Warrants may not be sold, transferred, assigned, pledged, or hypothecated, or be the subject of any hedging, short sale, derivative, put, or call transaction that would result in the effective economic disposition of the securities by any person for a period of 180 days following the effective date of the registration statement of which this prospectus forms a part, except that they may be assigned, in whole or in part, to any officer or partner of the Representative and to members of the underwriting syndicate or selling group and officers and partners thereof.
Transfer Agent
The transfer agent and registrar for our Common Stock is: Vstock Transfer, LLC; Address: 18 Lafayette Place, Woodmere, NY 11598; Phone: (212) 828 – 8436; website: https://www.vstocktransfer.com/.
Anti-Takeover Provisions
Nevada Revised Statutes
Acquisition of Controlling Interest Statutes. Nevada’s “acquisition of controlling interest” statutes contain provisions governing the acquisition of a controlling interest in certain Nevada corporations. These “control share” laws provide generally that any person that acquires a “controlling interest” in certain Nevada corporations may be denied certain voting rights, unless a majority of the disinterested shareholders of the corporation elects to restore such voting rights. These statutes provide that a person acquires a “controlling interest” whenever a person acquires shares of a subject corporation that, but for the application of these provisions of the Nevada Revised Statutes, would enable that person to exercise (1) one-fifth or more, but less than one-third, (2) one-third or more, but less than a majority or (3) a majority or more, of all of the voting power of the corporation in the election of directors. Once an acquirer crosses one of these thresholds, shares which it acquired in the transaction taking it over the threshold and within the 90 days immediately preceding the date when the acquiring person acquired or offered to acquire a controlling interest become “control shares” to which the voting restrictions described above apply. Our articles of incorporation and bylaws currently contain no provisions relating to these statutes, and unless our articles of incorporation or bylaws in effect on the tenth day after the acquisition of a controlling interest were to provide otherwise, these laws would apply to us if we were to (i) have 200 or more shareholders of record (at least 100 of which have addresses in the State of Nevada appearing on our stock ledger) and (ii) do business in the State of Nevada directly or through an affiliated corporation. If these laws were to apply to us, they might discourage companies or persons interested in acquiring a significant interest in or control of the Company, regardless of whether such acquisition may be in the interest of our shareholders.
Combinations with Interested Shareholders Statutes. Nevada’s “combinations with interested shareholders” statutes prohibit certain business “combinations” between certain Nevada corporations and any person deemed to be an “interested shareholder” for two years after such person first becomes an “interested shareholder” unless (i) the corporation’s board of directors approves the combination (or the transaction by which such person becomes an “interested shareholder”) in advance, or (ii) the combination is approved by the board of directors and sixty percent of the corporation’s voting power not beneficially owned by the interested shareholder, its affiliates and associates. Furthermore, in the absence of prior approval certain restrictions may apply even after such two-year period. For purposes of these statutes, an “interested shareholder” is any person who is (x) the beneficial owner, directly or indirectly, of ten percent or more of the voting power of the outstanding voting shares of the corporation, or (y) an affiliate or associate of the corporation and at any time within the two previous years was the beneficial owner, directly or indirectly, of ten percent or more of the voting power of the then outstanding shares of the corporation. The definition of the term “combination” is sufficiently broad to cover most significant transactions between the corporation and an “interested shareholder”. Subject to certain timing requirements set forth in the statutes, a corporation may elect not to be governed by these statutes. We have not included any such provision in our articles of incorporation.
The effect of these statutes may be to potentially discourage parties interested in taking control of the Company from doing so if it cannot obtain the approval of our board of directors.
PLAN OF DISTRIBUTION
Issuance of Shares Upon Exercise of Warrants
The prices at which the shares of common stock covered by this prospectus may actually be disposed of may be at fixed prices, at prevailing market prices at the time of sale, at prices related to the prevailing market price, at varying prices determined at the time of sale or at negotiated prices.
Pursuant to the terms of the warrants, the shares of common stock will be distributed to those holders who surrender the warrants and provide payment of the exercise price to us.
Upon receipt of proper notice by any of the holders of the warrants issued that such holder desires to exercise a warrant, we will, within the time allotted by the agreement governing the warrant, issue instructions to our transfer agent to issue to the holder shares of common stock, free of a restrictive legend. Warrant Shares that are held by affiliates will be issued free of legend but will be deemed control securities.
CERTAIN U.S. FEDERAL INCOME TAX CONSIDERATIONS
This section summarizes certain U.S. federal income tax considerations relating to the purchase, ownership and disposition of our Common Stock. This summary does not provide a complete analysis of all potential tax considerations. The information provided below is based upon provisions of the Internal Revenue Code of 1986, as amended (the “Code”), Treasury regulations promulgated thereunder and administrative rulings and judicial decisions, all as currently in effect. These authorities may change at any time, possibly on a retroactive basis, or the U.S. Internal Revenue Service (the “IRS”), might interpret the existing authorities differently. In either case, the tax considerations of purchasing, owning or disposing of Common Stock could differ from those described below.
This discussion is addressed only to U.S. holders (defined below) which hold our shares of Common Stock as a “capital asset” within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all of the U.S. federal income tax considerations that might be relevant to a beneficial owner in light of such beneficial owner’s particular circumstances or to beneficial owners subject to special treatment under the U.S. federal income tax laws, including:
| |
● |
a broker, dealer or trader in securities, currencies, commodities, or notional principal contracts; |
| |
● |
a bank, financial institution or insurance company; |
| |
● |
a regulated investment company, a real estate investment trust or grantor trust; |
| |
● |
a tax-exempt entity or organization, including an individual retirement account or Roth IRA as defined in Section 408 or 408A of the Code, respectively; |
| |
● |
a person holding the Common Stock as part of a hedging, integrated, or conversion transaction or a straddle, or a person deemed to sell Common Stock under the constructive sale provisions of the Code; |
| |
● |
a trader in securities that has elected the mark-to-market method of tax accounting for securities; |
| |
● |
an entity that is treated as a partnership or other pass-through entity for U.S. federal income tax purposes; |
| |
● |
a person who is a partner or investor in a partnership or other pass-through entity that holds the Common Stock; |
| |
● |
a U.S. person whose “functional currency” is not the U.S. dollar; |
| |
● |
a controlled foreign corporation or passive foreign investment company; |
| |
● |
a qualified foreign pension fund or an entity that is wholly owned by one or more qualified foreign pension funds; or |
For purposes of this discussion, a “U.S. holder” is a beneficial owner of a share of Common Stock that is, for U.S. federal income tax purposes:
| |
● |
an individual who is a citizen or resident of the United States; |
| |
● |
a corporation (or any other entity treated as a corporation for U.S. federal income tax purposes) created or organized in or under the laws of the United States, any state thereof or the District of Columbia; |
| |
● |
an estate the income of which is subject to U.S. federal income taxation regardless of its source; or |
| |
● |
a trust if (1) it is subject to the primary supervision of a court within the United States and one or more U.S. persons have the authority to control all substantial decisions of the trust or (2) it has a valid election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person. |
For purposes of this discussion, a “non-U.S. holder” is a beneficial owner of a share of Common Stock that is (i) a foreign corporation, (ii) a nonresident alien individual, or (iii) a foreign estate or trust that in each case is not subject to U.S. federal income tax on a net-income basis on income or gain from a share of Common Stock.
If a partnership holds shares of Common Stock, the tax treatment of a partner will generally depend upon the status of the partner and the activities of the partnership. A partnership holding shares of Common Stock or a partner therein should consult its own tax advisors as to the tax consequences of holding and disposing of shares of Common Stock.
You are urged to consult your tax advisor with respect to the application of the U.S. federal income tax laws to your particular situation, as well as any tax consequences of the purchase, ownership and disposition of our Common Stock arising under the U.S. federal estate or gift tax rules or under the laws of any U.S. state or local or any non-U.S. or other taxing jurisdiction or under any applicable tax treaty.
Certain U.S. Federal Income Tax Considerations for U.S. Holders of Common Stock
Dividends on our Common Stock
We do not expect to declare or pay any distributions on our Common Stock in the foreseeable future. If we do make any distributions on shares of our Common Stock, however, such distributions will be includible in the gross income of a U.S. holder as ordinary dividend income to the extent paid out of current or accumulated earnings and profits, as determined for U.S. federal income tax purposes. Any portion of a distribution in excess of current or accumulated earnings and profits would be treated as a return of the holder’s tax basis in its Common Stock and then as gain from the sale or exchange of the Common Stock. Under current law, if certain requirements are met, a preferential U.S. federal income tax rate will apply to any dividends paid to a holder of Common Stock who is a U.S. individual.
Distributions to U.S. holders that are corporate stockholders, constituting dividends for U.S. federal income tax purposes, may qualify for the dividends received deduction, or DRD, which is generally available to corporate stockholders. No assurance can be given that we will have sufficient earnings and profits (as determined for U.S. federal income tax purposes) to cause any distributions to be eligible for a DRD. In addition, a DRD is available only if certain holding periods and other taxable income requirements are satisfied.
Sale of Common Stock
A U.S. holder of Common Stock will generally recognize gain or loss on the taxable sale, exchange, or other taxable disposition of such stock in an amount equal to the difference between such U.S. holder’s amount realized on the sale and its adjusted tax basis in the Common Stock sold. A U.S. holder’s amount realized should equal the amount of cash and the fair market value of any property received in consideration of its stock. The gain or loss should be capital gain or loss and should be long-term capital gain or loss if the Common Stock is held for more than one year at the time of disposition. The deductibility of capital losses for U.S. federal income tax purposes is subject to limitations under the Code. Under current law, long-term capital gain recognized by an individual U.S. holder is generally eligible for a preferential U.S. federal income tax rate.
Information Reporting and Backup Withholding
Information reporting requirements generally will apply to payments of dividends on shares of Common Stock and to the proceeds of a sale of Common Stock unless a U.S. holder is an exempt recipient, such as a corporation. Backup withholding will apply to those payments if a U.S. holder fails to provide its correct taxpayer identification number and certification of exempt status, or fails to report in full dividend income. Any amounts withheld under the backup withholding rules will be allowed as a refund or a credit against U.S. federal income tax liability, provided the required information is timely furnished to the IRS.
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers or persons controlling the registrant pursuant to the foregoing provisions, the registrant has been informed that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
LEGAL MATTERS
The validity of the securities being offered by this prospectus been passed upon for us by Hunter Taubman Fischer & Li LLC. Carmel, Milazzo & Feil LLP, New York, New York is acting as counsel for the representative of the underwriters with respect to the offering.
EXPERTS
The consolidated financial statements of ABVC
BioPharma, Inc. as of December 31, 2023 and 2022 included elsewhere in this prospectus have been audited by WWC P.C. CPA, independent
registered public accounting firm, as set forth in their report appearing elsewhere herein, and are included in reliance upon such report
given on the authority of such firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We are a reporting company and file annual, quarterly and special reports, and other information with the SEC. Copies of the reports and other information may be read and copied at the SEC’s Public Reference Room at 100 F Street N.E., Washington, D.C. 20549. You can request copies of such documents by writing to the SEC and paying a fee for the copying cost. You may obtain information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC maintains a web site at http://www.sec.gov that contains reports, proxy and information statements and other information regarding registrants that file electronically with the SEC.
This prospectus is part of a registration statement on Form S-1 that we filed with the SEC. Certain information in the registration statement has been omitted from this prospectus in accordance with the rules and regulations of the SEC. We have also filed exhibits and schedules with the registration statement that are excluded from this prospectus. For further information you may:
| |
● |
read a copy of the registration statement, including the exhibits and schedules, without charge at the SEC’s Public Reference Room; or |
| |
● |
obtain a copy from the SEC upon payment of the fees prescribed by the SEC. |
We file periodic reports, proxy statements, and other information with the SEC. These periodic reports, proxy statements, and other information will be available for inspection and copying at the SEC’s public reference facilities and the website of the SEC referred to above. After the closing of this offering, you may access our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act with the SEC free of charge as soon as reasonably practicable after such material is electronically filed with, or furnished to, the SEC. The information contained in, or that can be accessed through, our website is not incorporated by reference into this prospectus.
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to our directors, officers and controlling persons pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by us of expenses incurred or paid by one of our directors, officers or controlling persons in the successful defense of any action, suit or proceeding) is asserted by that director, officer or controlling person in connection with the securities being registered, we will, unless in the opinion of our counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether that indemnification by us is against public policy as expressed in the Securities Act and will be governed by the final adjudication of that issue.
INDEX TO FINANCIAL INFORMATION
Our Consolidated Financial Statements and
Notes thereto and the report of WWC P.C. CPA, our independent registered public accounting firm, are set forth on pages F-1 through F-38
of this Report.

REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
| To: | The Board of Directors and Stockholders of |
| ABVC BioPharma, Inc. |
Opinion on the Financial Statements
We have audited the accompanying
consolidated balance sheets of ABVC BioPharma, Inc., and subsidiaries (collectively the “Company”) as of December 31, 2023,
and 2022, and the related consolidated statements of operation and comprehensive loss, cash flows, stockholders’ equity (deficit),
and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present
fairly, in all material respects, the financial position of the Company as of December 31, 2023, and 2022, and the results of its operations
and its cash flows in each of the years for the two-year period ended December 31, 2023, in conformity with accounting principles
generally accepted in the United States of America.
Substantial Doubt about the Company’s
Ability to Continue as a Going Concern
The accompanying financial
statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 2 to the financial statements,
the Company incurred substantial losses during the year ended December 31, 2023. As of December 31, 2023, the Company had a working capital
deficit and net cash outflows from operating activities. These conditions raise substantial doubt about the Company’s ability to
continue as a going concern. Management’s plans in regard to these matters are also described in Note 2. The financial statements
do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements
are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial
statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States)
(PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable
rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in
accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance
about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to
have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required
to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness
of our internal control over financial reporting. Accordingly, we express no such opinion .
Our audits included performing
procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures
that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the
financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management,
as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for
our opinion.

Critical Audit Matters
The critical audit matters
communicated below are matters arising from the current period audit of the financial statements that were communicated or required to
be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and
(2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter
in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit
matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Purchase of property via common stock
As described in Note 5
of the financial statements, the Company acquired the property by entered and governed by a complex cooperation agreement. The terms
and conditions of the agreement dictate the proper categorization and recognition of these property in the Company’s financial
statements. Accordingly, we have identified the purchase of property and equipment via common stock as a critical audit matter due to the
complexity of such cooperation agreements.
The primary audit procedures
we performed in order to address this critical audit matter were the following: (i) examined the cooperation agreement and other related
documents, evaluated the terms and conditions, (ii) gained an understanding of the structure set forth by the agreement by enquiring with
management, (iii) confirmed with the counterparty in the cooperation agreement with their understanding of the terms and conditions of
the cooperation agreements and compared their responses with the Company’s books and records, (iv) tested the reasonableness, completeness,
mathematical accuracy and relevance of key underlying data used in the valuation of the property. Common
stock, additional paid in capital and property and equipment, net is affected by this critical audit matter.
Stock-based compensation to third parties
As described in Note 12 of
the financial statements, the Company granted common stock to third parties as consideration to consultants for services rendered; these
grants were recorded as stock-based compensation expense in the Company’s results of operations. We identified the recognition of
stock-based compensation to non-employees as a critical audit matter due to the significant judgments and assumptions made by management
to apply proper valuation and allocation to such grants.
The primary procedures we
performed in order to address this critical audit matter were the following: (i) obtained and examined the board meeting minutes, board
resolutions, and service contracts, (ii) evaluated the reasonableness of the fair value of services received from the non-employees receiving
the grants, either measured at the fair value at the outset of the contract, or around the completion date of the service contract and
compared those amounts against the fair value of the grants based on the prevailing market value. Common
stock, additional paid in capital and stock-based compensation is affected by this critical audit matter.

WWC, P.C.
Certified Public Accountants
PCAOB ID No. 1171
We have served as the Company’s auditor since 2022.
San Mateo, California
March 13, 2024
ABVC BIOPHARMA, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
| | |
December 31,
2023 | | |
December 31,
2022 | |
| ASSETS | |
| | |
| |
| Current Assets | |
| | |
| |
| Cash and cash equivalents | |
$ | 60,155 | | |
$ | 85,265 | |
| Restricted cash | |
| 656,625 | | |
| 1,306,463 | |
| Accounts receivable, net | |
| 1,530 | | |
| 98,325 | |
| Accounts receivable – related parties, net | |
| 10,463 | | |
| 757,343 | |
| Due from related parties – current | |
| 747,573 | | |
| 513,819 | |
| Short-term investments | |
| 79,312 | | |
| 75,797 | |
| Prepaid expense and other current assets | |
| 101,051 | | |
| 150,235 | |
| Total Current Assets | |
| 1,656,709 | | |
| 2,987,247 | |
| | |
| | | |
| | |
| Property and equipment, net | |
| 7,969,278 | | |
| 573,978 | |
| Operating lease right-of-use assets | |
| 809,283 | | |
| 1,161,141 | |
| Long-term investments | |
| 2,527,740 | | |
| 842,070 | |
| Deferred tax assets, net | |
| - | | |
| 117,110 | |
| Prepaid expenses – non-current | |
| 78,789 | | |
| 135,135 | |
| Security deposits | |
| 62,442 | | |
| 58,838 | |
| Prepayment for long-term investments | |
| 1,274,842 | | |
| 2,838,578 | |
| Due from related parties – non-current, net | |
| 113,516 | | |
| 865,477 | |
| Total Assets | |
$ | 14,492,599 | | |
$ | 9,579,574 | |
| | |
| | | |
| | |
| LIABILITIES AND EQUITY | |
| | | |
| | |
| Current Liabilities | |
| | | |
| | |
| Short-term bank loans | |
$ | 899,250 | | |
$ | 1,893,750 | |
| Accrued expenses and other current liabilities | |
| 3,696,380 | | |
| 2,909,587 | |
| Contract liabilities | |
| 79,500 | | |
| 10,985 | |
| Taxes payables | |
| 112,946 | | |
| - | |
| Operating lease liabilities – current portion | |
| 401,826 | | |
| 369,314 | |
| Due to related parties | |
| 173,132 | | |
| 359,992 | |
| Convertible notes payable – third parties, net | |
| 569,456 | | |
| - | |
| Total Current Liabilities | |
| 5,932,490 | | |
| 5,543,628 | |
| | |
| | | |
| | |
| Tenant security deposit | |
| 21,680 | | |
| 7,980 | |
| Operating lease liability – non-current portion | |
| 407,457 | | |
| 791,827 | |
| Total Liabilities | |
| 6,361,627 | | |
| 6,343,435 | |
| COMMITMENTS AND CONTINGENCIES | |
| | | |
| | |
| Equity | |
| | | |
| | |
| Preferred stock, $0.001 par value, 20,000,000 authorized, nil shares issued and outstanding | |
| - | | |
| - | |
| Common stock, $0.001 par value, 100,000,000 authorized, 7,940,298 and 3,286,190 shares issued and outstanding as of December 31, 2023 and 2022, respectively(1) | |
| 7,940 | | |
| 3,286 | |
| Additional paid-in capital | |
| 82,636,966 | | |
| 67,937,050 | |
| Stock subscription receivable | |
| (451,480 | ) | |
| (1,354,440 | ) |
| Accumulated deficit | |
| (65,420,095 | ) | |
| (54,904,439 | ) |
| Accumulated other comprehensive income | |
| 516,387 | | |
| 517,128 | |
| Treasury stock | |
| (8,901,668 | ) | |
| (9,100,000 | ) |
| Total Stockholders’ equity | |
| 8,388,050 | | |
| 3,098,585 | |
| Noncontrolling interest | |
| (257,078 | ) | |
| 137,554 | |
| Total Equity | |
| 8,130,972 | | |
| 3,236,139 | |
| | |
| | | |
| | |
| Total Liabilities and Equity | |
$ | 14,492,599 | | |
$ | 9,579,574 | |
| (1) |
Prior period results have been adjusted to reflect the 1-for-10 reverse stock split effected on July 25, 2023. |
The accompanying notes are an integral part
of these consolidated financial statements.
ABVC BIOPHARMA, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE
LOSS
| |
|
Year Ended
December 31, |
|
| |
|
2023 |
|
|
2022 |
|
| Revenues |
|
$ |
152,430 |
|
|
$ |
969,783 |
|
| |
|
|
|
|
|
|
|
|
| Cost of revenues |
|
|
302,037 |
|
|
|
286,415 |
|
| |
|
|
|
|
|
|
|
|
| Gross (loss) profit |
|
|
(149,607 |
) |
|
|
683,368 |
|
| |
|
|
|
|
|
|
|
|
| Operating expenses |
|
|
|
|
|
|
|
|
| Selling, general and administrative expenses |
|
|
5,368,278 |
|
|
|
6,067,545 |
|
| Research and development expenses |
|
|
1,062,916 |
|
|
|
2,693,457 |
|
| Stock-based compensation |
|
|
1,635,708 |
|
|
|
7,036,778 |
|
| Total operating expenses |
|
|
8,066,902 |
|
|
|
15,797,780 |
|
| |
|
|
|
|
|
|
|
|
| Loss from operations |
|
|
(8,216,509 |
) |
|
|
(15,114,412 |
) |
| |
|
|
|
|
|
|
|
|
| Other income (expense) |
|
|
|
|
|
|
|
|
| Interest income |
|
|
185,481 |
|
|
|
187,817 |
|
| Interest expense |
|
|
(2,493,340 |
) |
|
|
(293,968 |
) |
| Operating sublease income |
|
|
65,900 |
|
|
|
107,150 |
|
| Impairment loss |
|
|
- |
|
|
|
(110,125 |
) |
| Investment loss |
|
|
- |
|
|
|
(7,446 |
) |
| Gain (loss) on foreign exchange changes |
|
|
22,690 |
|
|
|
(259,463 |
) |
| Loss on investment in equity securities |
|
|
(221,888 |
) |
|
|
- |
|
| Other income (expenses) |
|
|
3,384 |
|
|
|
(24,149 |
) |
| Total other income (expenses) |
|
|
(2,437,773 |
) |
|
|
(400,184 |
) |
| |
|
|
|
|
|
|
|
|
| Loss before provision income tax |
|
|
(10,654,282 |
) |
|
|
(15,514,596 |
) |
| |
|
|
|
|
|
|
|
|
| Provision for income tax expense |
|
|
256,006 |
|
|
|
797,778 |
|
| |
|
|
|
|
|
|
|
|
| Net loss |
|
|
(10,910,288 |
) |
|
|
(16,312,374 |
) |
| |
|
|
|
|
|
|
|
|
| Net loss attributable to noncontrolling interests |
|
|
(394,632 |
) |
|
|
110,865 |
|
| |
|
|
|
|
|
|
|
|
| Net loss attributed to ABVC and subsidiaries |
|
|
(10,515,656 |
) |
|
|
(16,423,239 |
) |
| Foreign currency translation adjustment |
|
|
(741 |
) |
|
|
(22,532 |
) |
| Comprehensive loss |
|
$ |
(10,516,397 |
) |
|
$ |
(16,445,771 |
) |
| |
|
|
|
|
|
|
|
|
| Net loss per share: |
|
|
|
|
|
|
|
|
| Basic and diluted |
|
$ |
(2.43 |
) |
|
$ |
(5.19 |
) |
| |
|
|
|
|
|
|
|
|
| Weighted average number of common shares outstanding(1): |
|
|
|
|
|
|
|
|
| Basic and diluted |
|
|
4,335,650 |
|
|
|
3,166,460 |
|
| (1) |
Prior period results have been adjusted to reflect the 1-for-10 reverse stock split effected on July 25, 2023. |
The accompanying notes are an integral part
of these consolidated financial statements.
ABVC BIOPHARMA, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE YEAR ENDED DECEMBER 31, 2023 AND 2022
| | |
2023 | | |
2022 | |
| Cash flows from operating activities | |
| | |
| |
| Net loss | |
$ | (10,910,288 | ) | |
$ | (16,312,374 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Depreciation | |
| 28,531 | | |
| 23,799 | |
| Stock-based compensation | |
| 1,635,708 | | |
| 7,036,778 | |
| Inventory allowance for valuation losses | |
| - | | |
| 25,975 | |
| Provision for doubtful accounts | |
| 1,455,101 | | |
| 184,589 | |
| Other non-cash expenses | |
| 2,413,746 | | |
| 32,350 | |
| Impairment of prepaid expenses | |
| - | | |
| 110,125 | |
| Loss on investment in equity securities | |
| 221,888 | | |
| - | |
| Deferred tax expense | |
| 115,668 | | |
| 864,802 | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Decrease (increase) in accounts receivable | |
| 228,557 | | |
| (614,166 | ) |
| Decrease (increase) in prepaid expenses and other current assets | |
| 101,926 | | |
| 238,092 | |
| Decrease (increase) in due from related parties | |
| (321,776 | ) | |
| (837,014 | ) |
| Increase (decrease) in accrued expenses and other current liabilities | |
| 786,793 | | |
| 1,608,784 | |
| Increase (decrease) in contract liabilities | |
| 68,515 | | |
| - | |
| Increase (decrease) in tenant security deposit | |
| 13,700 | | |
| (2,600 | ) |
| Increase (decrease) in Taxes payables | |
| 112,946 | | |
| - | |
| Increase (decrease) in due to related parties | |
| (186,860 | ) | |
| 242,469 | |
| Net cash used in operating activities | |
| (4,235,845 | ) | |
| (7,398,391 | ) |
| | |
| | | |
| | |
| Cash flows from investing activities | |
| | | |
| | |
| Purchase of equipment | |
| (21,201 | ) | |
| (119,692 | ) |
| Prepayment for equity investment | |
| (338,985 | ) | |
| (1,601,992 | ) |
| Net cash used in investing activities | |
| (360,186 | ) | |
| (1,721,684 | ) |
| | |
| | | |
| | |
| Cash flows from financing activities | |
| | | |
| | |
| Issuance of common stock | |
| 1,050,000 | | |
| 3,663,925 | |
| Repayment of short-term bank loans | |
| (1,000,000 | ) | |
| - | |
| Proceeds from issuance of warrants | |
| 2,406,338 | | |
| - | |
| Proceeds from short-term bank loans | |
| - | | |
| 350,000 | |
| Proceeds from convertible notes payable | |
| 1,462,622 | | |
| - | |
| Net cash provided by financing activities | |
| 3,918,960 | | |
| 4,013,925 | |
| | |
| | | |
| | |
| Effect of exchange rate changes on cash and cash equivalents and restricted cash | |
| 2,123 | | |
| (67,337 | ) |
| | |
| | | |
| | |
| Net increase (decrease) in cash and cash equivalents and restricted cash | |
| (674,948 | ) | |
| (5,173,487 | ) |
| | |
| | | |
| | |
| Cash and cash equivalents and restricted cash | |
| | | |
| | |
| Beginning | |
| 1,391,728 | | |
| 6,565,215 | |
| Ending | |
$ | 716,780 | | |
$ | 1,391,728 | |
| | |
| | | |
| | |
| Supplemental disclosure of cash flows | |
| | | |
| | |
| Cash paid during the year for: | |
| | | |
| | |
| Interest expense paid | |
$ | 33,180 | | |
$ | 285,465 | |
| Income taxes paid | |
$ | 27,392 | | |
$ | 1,600 | |
| Non-cash financing and investing activities | |
| | | |
| | |
| Purchase of Property and equipment by issuing common stock to a third party | |
$ | 7,400,000 | | |
$ | - | |
| Issuance of common stock for conversion of debt | |
$ | 3,306,112 | | |
$ | - | |
The accompanying notes are an integral part
of these consolidated financial statements.
ABVC BIOPHARMA, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’
EQUITY (DEFICIT)
FOR THE YEAR ENDED DECEMBER 31, 2023 AND 2022
| | |
Common
Stock | | |
Stock | | |
Additional | | |
| | |
Accumulated
Other | | |
Treasury
Stock | | |
Non | | |
Stockholders’ | |
| | |
Number
of
shares(1) | | |
Amounts(1) | | |
Subscription
Receivable | | |
Paid-in
Capital(1) | | |
Accumulated
Deficit | | |
Comprehensive
Income | | |
Number of
Shares(1) | | |
Amount | | |
controlling
Interest | | |
Total
Equity | |
| Balance
at December 31, 2021 | |
| 2,893,089 | | |
$ | 2,893 | | |
$ | (2,257,400 | ) | |
$ | 58,139,700 | | |
$ | (38,481,200 | ) | |
$ | 539,660 | | |
| (27,535 | ) | |
$ | (9,100,000 | ) | |
$ | 26,689 | | |
$ | 8,870,342 | |
| Issuance
of common shares for cash | |
| 200,000 | | |
| 200 | | |
| - | | |
| 3,663,725 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 3,663,925 | |
| Issuance
of common shares for consulting service | |
| 193,101 | | |
| 193 | | |
| - | | |
| 4,891,695 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 4,891,888 | |
| Stock-based
compensation for services | |
| - | | |
| - | | |
| 902,960 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 902,960 | |
| Stock-based
compensation for options granted | |
| - | | |
| - | | |
| - | | |
| 1,241,930 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,241,930 | |
| Net
loss for the year | |
| - | | |
| - | | |
| - | | |
| - | | |
| (16,423,239 | ) | |
| - | | |
| - | | |
| - | | |
| 110,865 | | |
| (16,312,374 | ) |
| Cumulative
transaction adjustments | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (22,532 | ) | |
| - | | |
| - | | |
| - | | |
| (22,532 | ) |
| Balance
at December 31, 2022 | |
| 3,286,190 | | |
$ | 3,286 | | |
$ | (1,354,440 | ) | |
$ | 67,937,050 | | |
$ | (54,904,439 | ) | |
$ | 517,128 | | |
| (27,535 | ) | |
$ | (9,100,000 | ) | |
$ | 137,554 | | |
$ | 3,236,139 | |
| Issuance
of common shares for cash | |
| 300,000 | | |
| 300 | | |
| - | | |
| 1,049,700 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,050,000 | |
| Issuance
of common shares for consulting service | |
| 51,941 | | |
| 52 | | |
| - | | |
| 732,696 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 732,748 | |
| Issuance
of common shares for property | |
| 370,000 | | |
| 370 | | |
| - | | |
| 7,399,630 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 7,400,000 | |
| Issuance
of common shares upon exercise of convertible notes | |
| 3,732,167 | | |
| 3,732 | | |
| - | | |
| 3,302,380 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 3,306,112 | |
| Warrant issued with convertible notes payable | |
| - | | |
| - | | |
| - | | |
| 1,706,338 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 1,706,338 | |
| Issuance
of pre-funded warrant | |
| - | | |
| - | | |
| - | | |
| 700,000 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 700,000 | |
| Exercise
of pre-funded warrant | |
| 200,000 | | |
| 200 | | |
| - | | |
| (200 | ) | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | |
| Stock-based
compensation for services | |
| - | | |
| - | | |
| 902,960 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 902,960 | |
| Net
loss for the year | |
| - | | |
| - | | |
| - | | |
| - | | |
| (10,515,656 | ) | |
| - | | |
| - | | |
| - | | |
| (394,632 | ) | |
| (10,910,288 | ) |
| Cumulative
transaction adjustments | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (741 | ) | |
| - | | |
| - | | |
| - | | |
| (741 | ) |
| Distribute
as Employee Compensation | |
| - | | |
| - | | |
| - | | |
| (190,628 | ) | |
| - | | |
| - | | |
| 591 | | |
| 198,332 | | |
| - | | |
| 7,704 | |
| Balance
at December 31, 2023 | |
| 7,940,298 | | |
$ | 7,940 | | |
$ | (451,480 | ) | |
$ | 82,636,966 | | |
$ | (65,420,095 | ) | |
$ | 516,387 | | |
| (26,553 | ) | |
$ | (8,901,668 | ) | |
$ | (257,078 | ) | |
$ | 8,130,972 | |
| (1) |
Prior period results have been adjusted to reflect the 1-for-10 reverse stock split effected on July 25, 2023. |
The accompanying notes are an integral part
of these consolidated financial statements.
ABVC BIOPHARMA, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. ORGANIZATION AND DESCRIPTION OF BUSINESS
ABVC BioPharma, Inc. (the “Company”),
formerly known as American BriVision (Holding) Corporation, a Nevada corporation, through the Company’s operating entity, American
BriVision Corporation (“BriVision”), which was incorporated in July 2015 in the State of Delaware, engages in biotechnology
to fulfill unmet medical needs and focuses on the development of new drugs and medical devices derived from plants. BriVision develops
its pipeline by carefully tracking new medical discoveries or medical device technologies in research institutions in the Asia-Pacific
region. Pre-clinical, disease animal model and Phase I safety studies are examined closely by the Company to identify drugs that BriVision
believes demonstrate efficacy and safety. Once a drug appears to be a good candidate for development and ultimately commercialization,
BriVision licenses the drug or medical device from the original researchers and begins to introduce the drugs clinical plan to highly
respected principal investigators in the United States, Australia and Taiwan to conduct a Phase II clinical trial. At present, clinical
trials for the Company’s drugs and medical devices are being conducted at such world-famous institutions as Memorial Sloan Kettering
Cancer Center (“MSKCC”) and MD Anderson Cancer Center. BriVision had no predecessor operations prior to its formation on July
21, 2015.
Acquisition of AiBtl BioPharma Inc.
On November 12, 2023, the Company and one of its
subsidiaries, BioLite, Inc. (“BioLite Taiwan”) each entered into a multi-year, global licensing agreement with AiBtl BioPharma
Inc. (“AIBL”, or the acquired company) for the Company and BioLite Taiwan’s CNS drugs with the indications of MDD (Major
Depressive Disorder) and ADHD (Attention Deficit Hyperactivity Disorder) (collectively, the “Licensed Products”). The potential
license will cover the Licensed Products’ clinical trial, registration, manufacturing, supply, and distribution rights. The parties
are determined to collaborate on the global development of the Licensed Products. The parties are also working to strengthen new drug
development and business collaboration, including technology, interoperability, and standards development. As per each of the respective
agreements, each of ABVC and BioLite Taiwan received 23 million shares of AIBL stock and as a result, the Company has a controlling interest
over AIBL. If certain milestones are met, the Company and BioLite Taiwan are each eligible to receive $3,500,000 and royalties equaling
5% of net sales, up to $100 million.
The Company concluded the assets acquired and
liabilities assumed did not meet the definition of a business as a limited number of inputs were acquired but no substantive business
processes or signs of output were acquired. As such, the acquisition was accounted for as an asset purchase. The purchase consideration
was nonmonetary assets (patent) and transfer on November12, 2023. The equity interest transferred to ABVC and BioLite Taiwan on December
15, 2023.
2. LIQUIDITY AND GOING CONCERN
The accompanying financial statements have been prepared in conformity
with U.S. GAAP which contemplates continuation of the Company on a going concern basis. The going concern basis assumes that assets are
realized, and liabilities are settled in the ordinary course of business at amounts disclosed in the financial statements. The Company’s
ability to continue as a going concern depends upon its ability to market and sell its products to generate positive operating cash flows.
For the year ended December 31, 2023, the Company reported net loss of $10,910,288. As of December 31, 2023, the Company’s working
capital deficit was $4,275,781. In addition, the Company had net cash outflows of $4,235,845 from operating activities for
the year ended December 31, 2023. These conditions give rise to substantial doubt as to whether the Company will be able to continue as
a going concern.
To sustain its ability to support the Company’s
operating activities, the Company may have to consider supplementing its available sources of funds through the following sources:
| ● | cash
generated from operations; |
| ● | other
available sources of financing from Taiwan banks and other financial institutions; and |
| ● | financial
support from the Company’s related party and shareholders. |
Management’s plan is to continue improve operations to generate
positive cash flows and raise additional capital through private or public offerings, or financial support from related parties or shareholders.
If the Company is not able to generate positive operating cash flows, and raise additional capital, there is the risk that the Company
may not be able to meet its short-term obligations. All of these factors raise substantial doubt about the ability of the Company to continue as a going concern.
The audited financial statements for the years ended December 31, 2023 and 2022 have been prepared on a going concern basis and do not
include any adjustments to reflect the possible future effects on the recoverability and classifications of assets or the amounts and
classifications of liabilities that may result from the inability of the Company to continue as a going concern.
3. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accompanying consolidated financial statements have been prepared
in accordance with the generally accepted accounting principles in the United States of America (the “U.S. GAAP”) and
pursuant to the regulations of the Securities and Exchange Commission (the “SEC”). All significant intercompany transactions
and account balances have been eliminated.
Reclassifications of Prior Year Presentation
Certain prior year unaudited consolidated balance
sheet and unaudited consolidated cash flow statement amounts have been reclassified for consistency with the current year presentation.
These reclassifications had no effect on the reported results of operations.
Fiscal Year
The Company changed its fiscal year from the period
beginning on October 1st and ending on September 30th to the period beginning on January 1st and ending on December 31st, beginning January
1, 2018.
Use of Estimates
The preparation of financial statements in conformity
with generally accepted accounting principles in the United States of America requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated
financial statements and the amount of revenues and expenses during the reporting periods. Actual results could differ materially from
those results.
Stock Reverse Split
On July 25, 2023, the Company filed a Certificate
of Amendment to its Articles of Incorporation authorizing a 1-for-10 reverse stock split of the issued and outstanding shares of its common
stock. The Company’s stockholders previously approved the Reverse Stock Split at the Company’s Special Shareholder Meeting
held on July 7, 2023. The Reverse Stock Split was effected to reduce the number of issued and outstanding shares and to increase the per
share trading value of the Company’s common stock, although that outcome is not guaranteed. In turn, the Company believes that the
Reverse Stock Split will enable the Company to restore compliance with certain continued listing standards of NASDAQ Capital Market. All
shares and related financial information in this Form 10-K reflect this 1-for-10 reverse stock split.
Fair Value Measurements
FASB ASC 820, “Fair Value Measurements”
defines fair value for certain financial and nonfinancial assets and liabilities that are recorded at fair value, establishes a framework
for measuring fair value and expands disclosures about fair value measurements. It requires that an entity measure its financial instruments
to base fair value on exit price, maximize the use of observable units and minimize the use of unobservable inputs to determine the exit
price. It establishes a hierarchy which prioritizes the inputs to valuation techniques used to measure fair value. This hierarchy increases
the consistency and comparability of fair value measurements and related disclosures by maximizing the use of observable inputs and minimizing
the use of unobservable inputs by requiring that observable inputs be used when available. Observable inputs are inputs that reflect the
assumptions market participants would use in pricing the assets or liabilities based on market data obtained from sources independent
of the Company. Unobservable inputs are inputs that reflect the Company’s own assumptions about the assumptions market participants
would use in pricing the asset or liability developed based on the best information available in the circumstances. The hierarchy prioritizes
the inputs into three broad levels based on the reliability of the inputs as follows:
| |
● |
Level 1– Inputs are quoted prices in active markets for identical
assets or liabilities that the Company has the ability to access at the measurement date. Valuation of these instruments does not require
a high degree of judgment as the valuations are based on quoted prices in active markets that are readily and regularly available. |
| |
● |
Level 2– Inputs other than quoted prices in active markets that
are either directly or indirectly observable as of the measurement date, such as quoted prices for similar assets or liabilities; quoted
prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially
the full term of the assets or liabilities. |
| |
● |
Level 3– Valuations based on inputs that are unobservable and
not corroborated by market data. The fair value for such assets and liabilities is generally determined using pricing models, discounted
cash flow methodologies, or similar techniques that incorporate the assumptions a market participant would use in pricing the asset or
liability. |
The carrying values of certain assets and liabilities
of the Company, such as cash and cash equivalents, restricted cash, accounts receivable, due from related parties, inventory, prepaid
expenses and other current assets, accrued expenses and other current liabilities, and due to related parties approximate fair value due
to their relatively short maturities. The carrying value of the Company’s short-term bank loans, convertible notes payable, and accrued
interest approximates their fair value as the terms of the borrowing are consistent with current market rates and the duration to maturity
is short. The carrying value of the Company’s long-term bank loan approximates fair value because the interest rates approximate
market rates that the Company could obtain for debt with similar terms and maturities.
Concentration of Credit Risk
The Company’s financial instruments that
are exposed to concentrations of credit risk consist primarily of cash and cash equivalents. The Company places its cash and temporary
cash investments in high quality credit institutions, but these investments may be in excess of Taiwan Central Deposit Insurance Corporation
and the U.S. Federal Deposit Insurance Corporation’s insurance limits. The Company does not enter into financial instruments for
hedging, trading or speculative purposes.
We perform ongoing credit evaluation of our customers
and requires no collateral. An allowance for doubtful accounts is provided based on a review of the collectability of accounts receivable.
We determine the amount of allowance for doubtful accounts by examining its historical collection experience and current trends in the
credit quality of its customers as well as its internal credit policies. Actual credit losses may differ from our estimates.
Concentration of Clients
As of December 31, 2023, the most major client,
specializes in developing and commercializing of dietary supplements and therapeutics in dietary supplement industry, accounted for 87.24%
of the Company’s total account receivable.
As of December 31, 2022, the most major clients,
specializes in developing and commercializing of dietary supplements and therapeutics in dietary supplement industry, accounted for 71.89%
of the Company’s total account receivable; the second major client with its Chairman being the Board of Director of BioKey, accounted
for 16.62% of the Company’s total account receivable.
For
the year ended December 31, 2023, the most major client, distributing nutritional supplement in Asia Pacific, accounted for 80.04% of
the Company’s total revenues. For the year ended December 31, 2022, one major client, who is a Shareholder of the Company that works
in development and commercialization of new drugs in Taiwan, accounted for 93.22% of the Company’s total revenues.
Cash and Cash Equivalents
The Company considers highly liquid investments with maturities of
three months or less to be cash equivalents when purchased. As of December 31, 2023 and 2022, the Company’s cash and cash equivalents
amounted to $60,155 and $85,265, respectively. Some of the Company’s cash deposits are held in financial institutions located
in Taiwan where there is currently regulation mandated on obligatory insurance of bank accounts. The Company believes this financial institution
is of high credit quality.
Restricted Cash
Restricted cash primarily consist of cash held
in a reserve bank account in Taiwan. As of December 31, 2023 and 2022, the Company’s restricted cash amounted $656,625 and $1,306,463,
respectively.
Inventory
Inventory consists of raw materials, work-in-process,
finished goods, and merchandise. Inventories are stated at the lower of cost or market and valued on a moving weighted average cost basis.
Market is determined based on net realizable value. The Company periodically reviews the age and turnover of its inventory to determine
whether any inventory has become obsolete or has declined in value, and incurs a charge to operations for known and anticipated inventory
obsolescence.
Accounts
receivable and allowance for expected credit losses accounts
Accounts receivable is recorded and carried at
the original invoiced amount less an allowance for any potential uncollectible amounts.
The Company make estimates of expected credit
and collectability trends for the allowance for credit losses and allowance for unbilled receivables based upon our assessment of various
factors, including historical experience, the age of the accounts receivable balances, credit quality of customers, current economic conditions
reasonable and supportable forecasts of future economic conditions, and other factors that may affect our ability to collect from customers.
The provision is recorded against accounts receivable balances, with a corresponding charge recorded in the consolidated statements of income.
Actual amounts received may differ from management’s estimate of credit worthiness and the economic environment. Delinquent account
balances are written-off against the allowance for doubtful accounts after management has determined that the likelihood of collection
is not probable.
Allowance
for expected credit losses accounts was $616,505 and $194,957 as of December 31, 2023 and 2022, respectively.
Revenue Recognition
During the fiscal year 2018, the Company adopted
Accounting Standards Codification (“ASC”), Topic 606 (ASC 606), Revenue from Contracts with Customers, using the modified
retrospective method to all contracts that were not completed as of January 1, 2018, and applying the new revenue standard as an adjustment
to the opening balance of accumulated deficit at the beginning of 2018 for the cumulative effect. The results for the Company’s
reporting periods beginning on and after January 1, 2018 are presented under ASC 606, while prior period amounts are not adjusted and
continue to be reported under the accounting standards in effect for the prior period. Based on the Company’s review of existing
collaborative agreements as of January 1, 2018, the Company concluded that the adoption of the new guidance did not have a significant
change on the Company’s revenue during all periods presented.
Pursuant to ASC 606, the Company recognizes revenue
when its customer obtains control of promised goods or services, in an amount that reflects the consideration that the Company expects
to receive in exchange for those goods or services. To determine revenue recognition for arrangements that the Company determines is within
the scope of ASC 606, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance
obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations
in the contract; and (v) recognize revenue when (or as) the Company satisfies a performance obligation. The Company only applies the five-step
model to contracts when it is probable that the Company will collect the consideration the Company is entitled to in exchange for the
goods or services the Company transfers to the customers. At inception of the contract, once the contract is determined to be within the
scope of ASC 606, the Company assesses the goods or services promised within each contract, determines those that are performance obligations,
and assesses whether each promised good or service is distinct. The Company then recognizes as revenue the amount of the transaction price
that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied.
The following are examples of when the Company
recognizes revenue based on the types of payments the Company receives.
Collaborative Revenues — The Company
recognizes collaborative revenues generated through collaborative research, development and/or commercialization agreements. The terms
of these agreements typically include payment to the Company related to one or more of the following: non-refundable upfront license fees,
development and commercial milestones, partial or complete reimbursement of research and development costs, and royalties on net sales
of licensed products. Each type of payments results in collaborative revenues except for revenues from royalties on net sales of licensed
products, which are classified as royalty revenues. To date, the Company has not received any royalty revenues. Revenue is recognized
upon satisfaction of a performance obligation by transferring control of a good or service to the collaboration partners.
As part of the accounting for these arrangements,
the Company applies judgment to determine whether the performance obligations are distinct, and develop assumptions in determining the
stand-alone selling price for each distinct performance obligation identified in the collaboration agreements. To determine the stand-alone
selling price, the Company relies on assumptions which may include forecasted revenues, development timelines, reimbursement rates for
R&D personnel costs, discount rates and probabilities of technical and regulatory success.
The Company had multiple deliverables under the
collaborative agreements, including deliverables relating to grants of technology licenses, regulatory and clinical development, and
marketing activities. Estimation of the performance periods of the Company’s deliverables requires the use of management’s
judgment. Significant factors considered in management’s evaluation of the estimated performance periods include, but are not limited
to, the Company’s experience in conducting clinical development, regulatory and manufacturing activities. The Company reviews the
estimated duration of its performance periods under its collaborative agreements on an annually basis, and makes any appropriate adjustments
on a prospective basis. Future changes in estimates of the performance period under its collaborative agreements could impact the timing
of future revenue recognition.
(i) Non-refundable upfront payments
If a license to the Company’s intellectual
property is determined to be distinct from the other performance obligations identified in an arrangement, the Company recognizes revenue
from the related non-refundable upfront payments based on the relative standalone selling price prescribed to the license compared to
the total selling price of the arrangement. The revenue is recognized when the license is transferred to the collaboration partners and
the collaboration partners are able to use and benefit from the license. To date, the receipt of non-refundable upfront fees was solely
for the compensation of past research efforts and contributions made by the Company before the collaborative agreements entered into and
it does not relate to any future obligations and commitments made between the Company and the collaboration partners in the collaborative
agreements.
(ii) Milestone payments
The Company is eligible to receive milestone payments
under the collaborative agreement with collaboration partners based on achievement of specified development, regulatory and commercial
events. Management evaluated the nature of the events triggering these contingent payments, and concluded that these events fall into
two categories: (a) events which involve the performance of the Company’s obligations under the collaborative agreement with collaboration
partners, and (b) events which do not involve the performance of the Company’s obligations under the collaborative agreement with
collaboration partners.
The former category of milestone payments consists
of those triggered by development and regulatory activities in the territories specified in the collaborative agreements. Management concluded
that each of these payments constitute substantive milestone payments. This conclusion was based primarily on the facts that (i) each
triggering event represents a specific outcome that can be achieved only through successful performance by the Company of one or more
of its deliverables, (ii) achievement of each triggering event was subject to inherent risk and uncertainty and would result in additional
payments becoming due to the Company, (iii) each of the milestone payments is non-refundable, (iv) substantial effort is required to complete
each milestone, (v) the amount of each milestone payment is reasonable in relation to the value created in achieving the milestone, (vi)
a substantial amount of time is expected to pass between the upfront payment and the potential milestone payments, and (vii) the milestone
payments relate solely to past performance. Based on the foregoing, the Company recognizes any revenue from these milestone payments in
the period in which the underlying triggering event occurs.
(iii) Multiple Element Arrangements
The Company evaluates multiple element arrangements
to determine (1) the deliverables included in the arrangement and (2) whether the individual deliverables represent separate units of
accounting or whether they must be accounted for as a combined unit of accounting. This evaluation involves subjective determinations
and requires management to make judgments about the individual deliverables and whether such deliverables are separate from other aspects
of the contractual relationship. Deliverables are considered separate units of accounting provided that: (i) the delivered item(s) has
value to the customer on a standalone basis and (ii) if the arrangement includes a general right of return relative to the delivered item(s),
delivery or performance of the undelivered item(s) is considered probable and substantially within its control. In assessing whether an
item under a collaboration has standalone value, the Company considers factors such as the research, manufacturing, and commercialization
capabilities of the collaboration partner and the availability of the associated expertise in the general marketplace. The Company also
considers whether its collaboration partners can use the other deliverable(s) for their intended purpose without the receipt of the remaining
element(s), whether the value of the deliverable is dependent on the undelivered item(s), and whether there are other vendors that can
provide the undelivered element(s).
The Company recognizes arrangement consideration
allocated to each unit of accounting when all of the revenue recognition criteria in ASC 606 are satisfied for that particular unit of
accounting. In the event that a deliverable does not represent a separate unit of accounting, the Company recognizes revenue from the
combined unit of accounting over the Company’s contractual or estimated performance period for the undelivered elements, which is
typically the term of the Company’s research and development obligations. If there is no discernible pattern of performance or objectively
measurable performance measures do not exist, then the Company recognizes revenue under the arrangement on a straight-line basis over
the period the Company is expected to complete its performance obligations. Conversely, if the pattern of performance in which the service
is provided to the customer can be determined and objectively measurable performance measures exist, then the Company recognizes revenue
under the arrangement using the proportional performance method. Revenue recognized is limited to the lesser of the cumulative amount
of payments received or the cumulative amount of revenue earned, as determined using the straight-line method or proportional performance
method, as applicable, as of the period ending date.
At the inception of an arrangement that includes
milestone payments, the Company evaluates whether each milestone is substantive and at risk to both parties on the basis of the contingent
nature of the milestone. This evaluation includes an assessment of whether: (1) the consideration is commensurate with either the Company’s
performance to achieve the milestone or the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting
from its performance to achieve the milestone, (2) the consideration relates solely to past performance and (3) the consideration is reasonable
relative to all of the deliverables and payment terms within the arrangement. The Company evaluates factors such as the scientific, clinical,
regulatory, commercial, and other risks that must be overcome to achieve the particular milestone and the level of effort and investment
required to achieve the particular milestone in making this assessment. There is considerable judgment involved in determining whether
a milestone satisfies all of the criteria required to conclude that a milestone is substantive. Milestones that are not considered substantive
are recognized as earned if there are no remaining performance obligations or over the remaining period of performance, assuming all other
revenue recognition criteria are met.
(iv) Royalties and Profit Sharing Payments
Under the collaborative agreement with the collaboration
partners, the Company is entitled to receive royalties on sales of products, which is at certain percentage of the net sales. The Company
recognizes revenue from these events based on the revenue recognition criteria set forth in ASC 606. Based on those criteria, the Company
considers these payments to be contingent revenues, and recognizes them as revenue in the period in which the applicable contingency is
resolved.
Revenues Derived from Research and Development
Activities Services — Revenues related to research and development and regulatory activities are recognized when the related services
or activities are performed, in accordance with the contract terms. The Company typically has only one performance obligation at the inception
of a contract, which is to perform research and development services. The Company may also provide its customers with an option to request
that the Company provides additional goods or services in the future, such as active pharmaceutical ingredient, API, or IND/NDA/ANDA/510K
submissions. The Company evaluates whether these options are material rights at the inception of the contract. If the Company determines
an option is a material right, the Company will consider the option a separate performance obligation.
If the Company is entitled to reimbursement from
its customers for specified research and development expenses, the Company accounts for the related services that it provides as separate
performance obligations if it determines that these services represent a material right. The Company also determines whether the reimbursement
of research and development expenses should be accounted for as revenues or an offset to research and development expenses in accordance
with provisions of gross or net revenue presentation. The Company recognizes the corresponding revenues or records the corresponding offset
to research and development expenses as it satisfies the related performance obligations.
The Company then determines the transaction price
by reviewing the amount of consideration the Company is eligible to earn under the contracts, including any variable consideration. Under
the outstanding contracts, consideration typically includes fixed consideration and variable consideration in the form of potential milestone
payments. At the start of an agreement, the Company’s transaction price usually consists of the payments made to or by the Company
based on the number of full-time equivalent researchers assigned to the project and the related research and development expenses incurred.
The Company does not typically include any payments that the Company may receive in the future in its initial transaction price because
the payments are not probable. The Company would reassess the total transaction price at each reporting period to determine if the Company
should include additional payments in the transaction price.
The Company receives payments from its customers
based on billing schedules established in each contract. Upfront payments and fees may be recorded as advance from customers upon receipt
or when due, and may require deferral of revenue recognition to a future period until the Company performs its obligations under these
arrangements. Amounts are recorded as accounts receivable when the right of the Company to consideration is unconditional. The Company
does not assess whether a contract has a significant financing component if the expectation at contract inception is such that the period
between payment by the customers and the transfer of the promised goods or services to the customers will be one year or less.
Property and Equipment, net
Property and equipment, net is carried at cost
net of accumulated depreciation. Repairs and maintenance are expensed as incurred. Expenditures that improve the functionality of the
related asset or extend the useful life are capitalized. When property and equipment is retired or otherwise disposed of, the related
gain or loss is included in operating income. Leasehold improvements are depreciated on the straight-line method over the shorter of the
remaining lease term or estimated useful life of the asset. Depreciation is calculated on the straight-line method, including property
and equipment under capital leases, generally based on the following useful lives:
| |
|
Estimated Life
in Years |
| Buildings and leasehold improvements |
|
5 ~ 50 |
| Machinery and equipment |
|
5 ~ 10 |
| Office equipment |
|
3 ~ 6 |
Construction-in-Progress
The Company acquires constructions that constructs
certain of its fixed assets. All direct and indirect costs that are related to the construction of fixed assets and incurred before the
assets are ready for their intended use are capitalized as construction-in-progress. No depreciation is provided in respect of construction-in-progress.
Construction in progress is transferred to specific fixed asset items and depreciation of these assets commences when they are ready for
their intended use.
Impairment of Long-Lived Assets
The Company has adopted Accounting Standards Codification
subtopic 360-10, Property, Plant and Equipment (“ASC 360-10”). ASC 360-10 requires that long-lived assets and certain identifiable
intangibles held and used by the Company be reviewed for impairment whenever events or changes in circumstances indicate that the carrying
amount of an asset may not be recoverable. The Company evaluates its long-lived assets for impairment annually or more often if events
and circumstances warrant. Events relating to recoverability may include significant unfavorable changes in business conditions, recurring
losses, or a forecasted inability to achieve break-even operating results over an extended period. Should impairment in value be indicated,
the carrying value of intangible assets will be adjusted, based on estimates of future discounted cash flows resulting from the use and
ultimate disposition of the asset. ASC 360-10 also requires assets to be disposed of be reported at the lower of the carrying amount or
the fair value less costs to sell.
Long-term Equity Investment
The Company acquires the equity investments to
promote business and strategic objectives. The Company accounts for non-marketable equity and other equity investments for which the Company
does not have control over the investees as:
| |
● |
Equity method investments when the Company has the ability to exercise significant influence, but not control, over the investee. Its proportionate share of the income or loss is recognized monthly and is recorded in gains (losses) on equity investments. |
| |
● |
Non-marketable cost method investments when the equity method does not apply. |
Significant judgment is required to identify whether an impairment
exists in the valuation of the Company’s non-marketable equity investments, and therefore the Company considers this a critical
accounting estimate. Its yearly analysis considers both qualitative and quantitative factors that may have a significant impact on the
investee’s fair value. Qualitative analysis of its investments involves understanding the financial performance and near-term prospects
of the investee, changes in general market conditions in the investee’s industry or geographic area, and the management and governance
structure of the investee. Quantitative assessments of the fair value of its investments are developed using the market and income approaches.
The market approach includes the use of comparable financial metrics of private and public companies and recent financing rounds. The
income approach includes the use of a discounted cash flow model, which requires significant estimates regarding the investees’
revenue, costs, and discount rates. The Company’s assessment of these factors in determining whether an impairment exists could
change in the future due to new developments or changes in applied assumptions.
Other-Than-Temporary Impairment
The Company’s long-term equity investments
are subject to a periodic impairment review. Impairments affect earnings as follows:
| |
● |
Marketable equity securities include the consideration of general market conditions, the duration and extent to which the fair value is below cost, and our ability and intent to hold the investment for a sufficient period of time to allow for recovery of value in the foreseeable future. The Company also considers specific adverse conditions related to the financial health of, and the business outlook for, the investee, which may include industry and sector performance, changes in technology, operational and financing cash flow factors, and changes in the investee’s credit rating. The Company records other-than-temporary impairments on marketable equity securities and marketable equity method investments in gains (losses) on equity investments. |
| |
● |
Non-marketable equity investments based on the
Company’s assessment of the severity and duration of the impairment, and qualitative and quantitative analysis of the operating
performance of the investee; adverse changes in market conditions and the regulatory or economic environment; changes in operating structure
or management of the investee; additional funding requirements; and the investee’s ability to remain in business. A series of operating
losses of an investee or other factors may indicate that a decrease in value of the investment has occurred that is other than temporary
and that shall be recognized even though the decrease in value is in excess of what would otherwise be recognized by application of the
equity method. A loss in value of an investment that is other than a temporary decline shall be recognized. Evidence of a loss in value
might include, but would not necessarily be limited to, absence of an ability to recover the carrying amount of the investment or inability
of the investee to sustain an earnings capacity that would justify the carrying amount of the investment. The Company records other-than-temporary
impairments for non-marketable cost method investments and equity method investments in gains (losses) on equity investments.
Other-than-temporary impairments of equity investments
were $0 and $0 for the year ended December 31, 2023 and 2022, respectively. |
Goodwill
The Company evaluates goodwill for impairment
annually or more frequently when an event occurs or circumstances change that indicate the carrying value may not be recoverable. In testing
goodwill for impairment, the Company may elect to utilize a qualitative assessment to evaluate whether it is more likely than not that
the fair value of a reporting unit is less than its carrying amount. If the qualitative assessment indicates that goodwill impairment
is more likely than not, the Company performs a two-step impairment test. The Company tests goodwill for impairment under the two-step
impairment test by first comparing the book value of net assets to the fair value of the reporting units. If the fair value is determined
to be less than the book value or qualitative factors indicate that it is more likely than not that goodwill is impaired, a second step
is performed to compute the amount of impairment as the difference between the estimated fair value of goodwill and the carrying value.
The Company estimates the fair value of the reporting units using discounted cash flows. Forecasts of future cash flows are based on our
best estimate of future net sales and operating expenses, based primarily on expected category expansion, pricing, market segment share,
and general economic conditions.
The Company completed the required testing of
goodwill for impairment as of December 31, 2023, and determined that goodwill was impaired because of the current financial condition
of the Company and the Company’s inability to generate future operating income without substantial sales volume increases, which
are highly uncertain. Furthermore, the Company anticipates future cash flows indicate that the recoverability of goodwill is not reasonably
assured.
Convertible Notes Payable
The Company accounts for the convertible notes
issued at a discount, by comparing the principal amount and book value, with the calculation of discounted method. The Company assess
the discount per month. The amortization period of the promissory note is 18 months.
Beneficial Conversion Feature
From
time to time, the Company may issue convertible notes that may contain an imbedded beneficial conversion feature. A beneficial conversion
feature exists on the date a convertible note is issued when the fair value of the underlying common stock to which the note is convertible
into is in excess of the remaining unallocated proceeds of the note after first considering the allocation of a portion of the note proceeds
to the fair value of the warrants, if related warrants have been granted. The intrinsic value of the beneficial conversion feature is
recorded as a debt discount with a corresponding amount to additional paid in capital. The debt discount is amortized to interest expense
over the life of the note using the effective interest method.
Warrants
The Company accounts for warrants as either equity-classified
or liability-classified instruments based on an assessment of the warrant’s specific terms and applicable authoritative guidance
in FASB ASC 480, Distinguishing Liabilities from Equity (“ASC 480”) and ASC 815, Derivatives and Hedging (“ASC 815”).
The assessment considers whether the warrants are freestanding financial instruments pursuant to ASC 480, meet the definition of a liability
pursuant to ASC 480, and whether the warrants meet all of the requirements for equity classification under ASC 815, including whether
the warrants are indexed to the Company’s own common shares and whether the warrant holders could potentially require “net
cash settlement” in a circumstance outside of the Company’s control, among other conditions for equity classification. This
assessment, which requires the use of professional judgment, is conducted at the time of warrant issuance and as of each subsequent quarterly
period end date while the warrants are outstanding. The Company determined that upon further review of the warrant agreement, the Public
Warrants issued pursuant to the warrant agreement qualify for equity accounting treatment.
For
issued or modified warrants that meet all of the criteria for equity classification, the warrants are required to be recorded as a component
of equity at the time of issuance. For issued or modified warrants that do not meet all the criteria for equity classification, the warrants
are required to be recorded as liabilities at their initial fair value on the date of issuance, and each balance sheet date thereafter.
Changes in the estimated fair value of the warrants are recognized as a non-cash gain or loss on the statements of operations.
Research and Development Expenses
The Company accounts for the cost of using licensing
rights in research and development cost according to ASC Topic 730-10-25-1. This guidance provides that absent alternative future uses
the acquisition of product rights to be used in research and development activities must be charged to research and development expenses
when incurred.
The Company accounts for R&D costs in accordance
with Accounting Standards Codification (“ASC”) 730, Research and Development (“ASC 730”). Research and development
expenses are charged to expense as incurred unless there is an alternative future use in other research and development projects or otherwise.
Research and development expenses are comprised of costs incurred in performing research and development activities, including personnel-related
costs, facilities-related overhead, and outside contracted services including clinical trial costs, manufacturing and process development
costs for both clinical and preclinical materials, research costs, and other consulting services. Non-refundable advance payment for goods
and services that will be used in future research and development activities are expensed when the activity has been performed or when
the goods have been received rather than when the payment is made. In instances where the Company enters into agreements with third parties
to provide research and development services, costs are expensed as services are performed.
Post-retirement and post-employment benefits
The Company’s subsidiaries in Taiwan adopted
the government mandated defined contribution plan pursuant to the Labor Pension Act (the “Act”) in Taiwan. Such labor regulations
require that the rate of contribution made by an employer to the Labor Pension Fund per month shall not be less than 6% of the worker’s
monthly salaries. Pursuant to the Act, the Company makes monthly contribution equal to 6% of employees’ salaries to the employees’
pension fund. The Company has no legal obligation for the benefits beyond the contributions made. The total amounts for such employee
benefits, which were expensed as incurred, were $10,314 and $13,031 for the years ended December 31, 2023 and 2022, respectively.
Other than the above, the Company does not provide any other post-retirement or post-employment benefits.
Stock-based Compensation
The Company measures expense associated with all
employee stock-based compensation awards using a fair value method and recognizes such expense in the consolidated financial statements
on a straight-line basis over the requisite service period in accordance with FASB ASC Topic 718 “Compensation-Stock Compensation”.
Total employee stock-based compensation expenses were $0 and $1,241,930 for the years ended December 31, 2023 and 2022, respectively.
The Company accounted for stock-based compensation
to non-employees in accordance with FASB ASC Topic 718 “Compensation-Stock Compensation” and FASB ASC Topic 505-50 “Equity-Based
Payments to Non-Employees” which requires that the cost of services received from non-employees is measured at fair value at the
earlier of the performance commitment date or the date service is completed and recognized over the period the service is provided. Total
non-employee stock-based compensation expenses were $1,635,708 and $5,794,848 for the years ended December 31, 2023 and 2022, respectively.
Income Taxes
The Company accounts for income taxes using the
asset and liability approach which allows the recognition and measurement of deferred tax assets to be based upon the likelihood of realization
of tax benefits in future years. Under the asset and liability approach, deferred taxes are provided for the net tax effects of temporary
differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax
purposes. A valuation allowance is provided for deferred tax assets if it is more likely than not these items will expire before the Company
is able to realize their benefits, or future deductibility is uncertain.
Under ASC 740, a tax position is recognized as
a benefit only if it is “more likely than not” that the tax position would be sustained in a tax examination, with a tax examination
being presumed to occur. The evaluation of a tax position is a two-step process. The first step is to determine whether it is more-likely-than-not
that a tax position will be sustained upon examination, including the resolution of any related appeals or litigations based on the technical
merits of that position. The second step is to measure a tax position that meets the more-likely-than-not threshold to determine the amount
of benefits recognized in the financial statements. A tax position is measured at the largest amount of benefit that is greater than 50
percent likely of being realized upon ultimate settlement. Tax positions that previously failed to meet the more-likely-than-not recognition
threshold should be recognized in the first subsequent period in which the threshold is met. Previously recognized tax positions that
no longer meet the more-likely-than-not criteria should be de-recognized in the first subsequent financial reporting period in which the
threshold is no longer satisfied. Penalties and interest incurred related to underpayment of income tax are classified as income tax expense
in the year incurred. No significant penalty or interest relating to income taxes has been incurred for the years ended December 31, 2023
and 2022. GAAP also provides guidance on de-recognition, classification, interest and penalties, accounting in interim periods, disclosures
and transition.
Valuation of Deferred Tax Assets
A valuation allowance is recorded to reduce the
Company’s deferred tax assets to the amount that is more likely than not to be realized. In assessing the need for the valuation
allowance, management considers, among other things, projections of future taxable income and ongoing prudent and feasible tax planning
strategies. If the Company determines that sufficient negative evidence exists, then it will consider recording a valuation allowance
against a portion or all of the deferred tax assets in that jurisdiction. If, after recording a valuation allowance, the Company’s
projections of future taxable income and other positive evidence considered in evaluating the need for a valuation allowance prove, with
the benefit of hindsight, to be inaccurate, it could prove to be more difficult to support the realization of its deferred tax assets.
As a result, an additional valuation allowance could be required, which would have an adverse impact on its effective income tax rate
and results. Conversely, if, after recording a valuation allowance, the Company determines that sufficient positive evidence exists in
the jurisdiction in which the valuation allowance was recorded, it may reverse a portion or all of the valuation allowance in that jurisdiction.
In such situations, the adjustment made to the deferred tax asset would have a favorable impact on its effective income tax rate and results
in the period such determination was made.
Loss Per Share of Common Stock
The Company calculates net loss per share in accordance
with ASC Topic 260, “Earnings per Share”. Basic loss per share is computed by dividing the net loss by the weighted average
number of common shares outstanding during the period. Diluted loss per share is computed similar to basic loss per share except that
the denominator is increased to include the number of additional common shares that would have been outstanding if the potential common
stock equivalents had been issued and if the additional common shares were dilutive. Diluted earnings per share excludes all dilutive
potential shares if their effect is anti-dilutive.
Commitments and Contingencies
The Company has adopted ASC Topic 450 “Contingencies”
subtopic 20, in determining its accruals and disclosures with respect to loss contingencies. Accordingly, estimated losses from loss contingencies
are accrued by a charge to income when information available before financial statements are issued or are available to be issued indicates
that it is probable that an asset had been impaired or a liability had been incurred at the date of the financial statements and the amount
of the loss can be reasonably estimated. Legal expenses associated with the contingency are expensed as incurred. If a loss contingency
is not probable or reasonably estimable, disclosure of the loss contingency is made in the financial statements when it is at least reasonably
possible that a material loss could be incurred.
Foreign-currency Transactions
For the Company’s subsidiaries in Taiwan,
the foreign-currency transactions are recorded in New Taiwan dollars (“NTD”) at the rates of exchange in effect when the transactions
occur. Gains or losses resulting from the application of different foreign exchange rates when cash in foreign currency is converted into
New Taiwan dollars, or when foreign-currency receivables or payables are settled, are credited or charged to income in the year of conversion
or settlement. On the balance sheet dates, the balances of foreign-currency assets and liabilities are restated at the prevailing exchange
rates and the resulting differences are charged to current income except for those foreign currencies denominated investments in shares
of stock where such differences are accounted for as translation adjustments under the Statements of Stockholders’ Equity (Deficit).
Translation Adjustment
The accounts of the Company’s subsidiaries
in Taiwan were maintained, and their financial statements were expressed, in New Taiwan Dollar (“NT$”). Such financial statements
were translated into U.S. Dollars (“$” or “USD”) in accordance ASC 830, “Foreign Currency Matters”,
with the NT$ as the functional currency. According to the Statement, all assets and liabilities are translated at the current exchange
rate, stockholder’s deficit are translated at the historical rates and income statement items are translated at an average exchange
rate for the period. The resulting translation adjustments are reported under other comprehensive income (loss) as a component of stockholders’
equity (deficit).
Recent Accounting Pronouncements
In August 2020, the FASB issued ASU 2020-06, Debt
— Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity
(Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity (“ASU 2020-06”). ASU
2020-06 simplifies the accounting for convertible debt by eliminating the beneficial conversion and cash conversion accounting models.
Upon adoption of ASU 2020-06, convertible debt, unless issued with a substantial premium or an embedded conversion feature that is not
clearly and closely related to the host contract, will no longer be allocated between debt and equity components. This modification will
reduce the issue discount and result in less non-cash interest expense in financial statements. ASU 2020-06 also updates the earnings
per share calculation and requires entities to assume share settlement when the convertible debt can be settled in cash or shares. For
contracts in an entity’s own equity, the type of contracts primarily affected by ASU 2020-06 are freestanding and embedded features
that are accounted for as derivatives under the current guidance due to a failure to meet the settlement assessment by removing the requirements
to (i) consider whether the contract would be settled in registered shares, (ii) consider whether collateral is required to be posted,
and (iii) assess shareholder rights. ASU 2020-06 is effective for fiscal years beginning after December 15, 2023. Early adoption is permitted,
but no earlier than fiscal years beginning after December 15, 2020, and only if adopted as of the beginning of such fiscal year. The Company
is currently evaluating the impact that the standard will have on its consolidated financial statements.
The Company is currently evaluating the impact
that the standards mentioned above will have on its consolidated financial statements.
4. COLLABORATIVE AGREEMENTS
Collaborative agreements with BHK, a related
party
| (i) | On February 24, 2015, BioLite Taiwan and BioHopeKing Corporation
(the “BHK”) entered into a co-development agreement, (the “BHK Co-Development Agreement”), pursuant to which
it is collaborative with BHK to develop and commercialize BLI-1401-2 (Botanical Drug) Triple Negative Breast Cancer (TNBC) Combination
Therapy (BLI-1401-2 Products) in Asian countries excluding Japan for all related intellectual property rights, and has developed it for
medicinal use in collaboration with outside researchers. The development costs shall be shared 50/50 between BHK and the Company. The
BHK Co-Development Agreement will remain in effect for fifteen years from the date of first commercial sale of the Product in in Asia
excluding Japan. |
On July 27, 2016, BioLite Taiwan and BHK agreed
to amend the payment terms of the milestone payment in an aggregate amount of $10 million based on the following schedule:
| |
● |
Upon the signing of the BHK Co-Development Agreement: $1 million, or 10% of total payment |
| |
● |
Upon the first Investigational New Drug (IND) submission and BioLite Taiwan will deliver all data to BHK according to FDA Reviewing requirement: $1 million, or 10% of total payment |
| |
● |
At the completion of first phase II clinical trial: $1 million, or 10% of total payment |
| |
● |
At the initiation of phase III of clinical trial research: $3 million, or 30% of total payment |
| |
● |
Upon the New Drug Application (NDA) submission: $4 million, or 40% of total payment |
In December 2015, BHK has paid a non-refundable
upfront cash payment of $1 million, or 10% of $10,000,000, upon the signing of BHK Co-Development Agreement. The Company concluded that
the deliverables are considered separate units of accounting as the delivered items have value to the customer on a standalone basis and
recognized this cash receipt as collaboration revenue when all research, technical, and development data was delivered to BHK in 2015.
The receipt is for the compensation of past research efforts and contributions made by BioLite Taiwan before this collaborative agreement
was signed and it does not relate to any future commitments made by BioLite Taiwan and BHK in this collaborative agreement. In August
2016, the Company has received the second milestone payment of NT$31,649,000, approximately equivalent to $1 million, and recognized collaboration
revenue for the year ended December 31, 2016. As of the date of this report, the Company has not completed the first phase II clinical
trial.
In
addition to the milestone payments, BioLite Taiwan is entitled to receive royalty on 12% of BHK’s net sales related to BLI-1401-2
Products. As of December 31, 2023 and 2022, the Company has not earned the royalty under the BHK Co-Development Agreement.
(ii) On December 9, 2015, BioLite Taiwan entered
into another two collaborative agreements (the “BHK Collaborative Agreements”), pursuant to which it is collaborative with
BHK to co-develop and commercialize BLI-1005 for “Targeting Major Depressive Disorder” (BLI-1005 Products) and BLI-1006 for
“Targeting Inflammatory Bowel Disease” (BLI-1006 Products) in Asia excluding Japan for all related intellectual property rights,
and has developed it for medicinal use in collaboration with outside researchers. The development costs shall be shared 50/50 between
BHK and the Company. The BHK Co-Development Agreement will remain in effect for fifteen years from the date of first commercial sale of
the Product in in Asia excluding Japan.
In 2015, the Company recognized the cash receipt
in a total of NT$50 million, approximately equivalent to $1.6 million, as collaboration revenue when all research, technical, and development
data was delivered to BHK. The Company concluded that the deliverables are considered separate units of accounting as the delivered items
have value to the customer on a standalone basis and recognized this payment as collaboration revenue when all research, technical, data
and development data was delivered to BHK. The cash receipt is for the compensation of past research efforts and contributions made by
BioLite Taiwan before this BHK Collaborative Agreements was signed and it does not relate to any future commitments made by BioLite Taiwan
and BHK in this BHK Collaborative Agreements.
In addition to the total of NT$50 million, approximately
equivalent to $1.60 million, BioLite Taiwan is entitled to receive 50% of the future net licensing income or net sales profit. As of December
31, 2023 and 2022, the Company has not earned the royalty under the BHK Collaborative Agreements.
Collaborative agreement with BioLite, Inc.,
a related party
The Company entered into a collaborative agreement
with BioLite, Inc. on December 29, 2015, and then entered into two addendums to such agreement, as amended and revised, (the “BioLite
Agreement”). The majority shareholder of BioLite is one of the Company’s subsidiaries, Mr. Jiang, the Company’s Chairman
is a director of BioLite and Dr. Jiang, the Company’s Chief Strategy Officer and a director, is the Chairman of BioLite.
Pursuant to the BioLite Agreement, the Company
acquired the sole licensing rights to develop and commercialize for therapeutic purposes six compounds from BioLite. In accordance with
the terms of the Agreement, the Company shall pay BioLite (i) milestone payments of up to $100 million in cash and equity of the Company
or equity securities owned by it at various stages on a schedule dictated by BioLite’s achievements of certain milestones, as set
forth in the Agreement (the “Milestone Payments”) and (ii) a royalty payment equal to 5% of net sales of the drug products
when ABV-1501 is approved for sale in the licensed territories. If BioLite fails to reach any of the milestones in a timely manner, it
may not receive the rest of the payments from the Company.
According to the BioLite Agreement, after Phase
II clinical trials are completed, 15% of the Milestone Payment becomes due and shall be paid in two stages: (i) 5% no later than December
31, 2021 (the “December 2021 Payment”) and (ii) 10% no later than December 31, 2022.
On February 12, 2022, the Company’s Board
of Directors determined that the December 2021 Payment, which is equal to $5,000,000, shall be paid via the cancellation of certain outstanding
debt, in the amount of $5,000,000, that BioLite owes the Company as of December 31, 20212023.
On February 22, 2022, the parties entered into
an amendment to the BioLite Agreement allowing the Company to make all payments due under the Agreement via the forgiveness of debt, in
equal value, owed by BioLite to the Company.
This was a related party transaction.
Co-Development agreement with Rgene Corporation,
a related party
On May 26, 2017, BriVision entered into a co-development
agreement (the “Co-Dev Agreement”) with Rgene Corporation (the “Rgene”), a related party under common control
by controlling beneficiary shareholder of YuanGene Corporation and the Company (See Note 12). Pursuant to Co-Dev Agreement, BriVision
and Rgene agreed to co-develop and commercialize ABV-1507 HER2/neu Positive Breast Cancer Combination Therapy, ABV-1511 Pancreatic Cancer
Combination Therapy and ABV-1527 Ovary Cancer Combination Therapy. Under the terms of the Co-Dev Agreement, Rgene is required to pay
the Company $3,000,000 in cash or stock of Rgene with equivalent value by August 15, 2017. The payment is for the compensation of BriVision’s
past research efforts and contributions made by BriVision before the Co-Dev Agreement was signed and it does not relate to any future
commitments made by BriVision and Rgene in this Co-Dev Agreement. In addition to $3,000,000, the Company is entitled to receive 50% of
the future net licensing income or net sales profit earned by Rgene, if any, and any development costs shall be equally shared by both
BriVision and Rgene.
On June 1, 2017, the Company has delivered all
research, technical, data and development data to Rgene. Since both Rgene and the Company are related parties and under common control
by a controlling beneficiary shareholder of YuanGene Corporation and the Company, the Company has recorded the full amount of $3,000,000
in connection with the Co-Dev Agreement as additional paid-in capital during the year ended December 31, 2017. During the year ended December
31, 2017, the Company has received $450,000 in cash. On December 24, 2018, the Company received the remaining balance of $2,550,000 in
the form of newly issued shares of Rgene’s Common Stock, at the price of NT$50 (approximately equivalent to $1.60 per share), for
an aggregate number of 1,530,000 shares, which accounted for equity method long-term investment as of December 31, 2018. During the year
ended December 31, 2018, the Company has recognized investment loss of $549. On December 31, 2018, the Company determined to fully write
off this investment based on the Company’s assessment of the severity and duration of the impairment, and qualitative and quantitative
analysis of the operating performance of the investee, adverse changes in market conditions and the regulatory or economic environment,
changes in operating structure of Rgene, additional funding requirements, and Rgene’s ability to remain in business. All projects
that have been initiated will be managed and supported by the Company and Rgene.
The Company and Rgene signed an amendment to the
Co-Dev Agreement on November 10, 2020, pursuant to which both parties agreed to delete AB-1507 HER2/neu Positive Breast Cancer Combination
Therapy and AB 1527 Ovary Cancer Combination Therapy and add ABV-1519 EGFR Positive Non-Small Cell Lung Cancer Combination Therapy and
ABV-1526 Large Intestine / Colon / Rectal Cancer Combination Therapy to the products to be co-developed and commercialized. Other provisions
of the Co-Dev Agreement remain in full force and effect.
On June 10, 2022, the Company expanded its co-development
partnership with Rgene. On that date, BioKey, ABVC has entered into a Clinical Development Service Agreement with Rgene to guide three
Rgene drug products, RGC-1501 for the treatment of Non-Small Cell Lung Cancer (NSCLC), RGC-1502 for the treatment of pancreatic cancer
and RGC 1503 for the treatment of colorectal cancer patients, through completion of Phase II clinical studies under the U.S. FDA IND regulatory
requirements. Under the terms of the new Services Agreement, BioKey is eligible to receive payments totaling $3.0 million over a 3-year
period with each payment amount to be determined by certain regulatory milestones obtained during the agreement period. The Service Agreement
shall remain in effect until the expiration date of the last patent and automatically renew for 5 more years unless terminated earlier
by either party with six months written notice. Either party may terminate the Service Agreement for cause by providing 30 days written
notice.
Through a series of transactions over the past
5 years, the Company and Rgene have co-developed the three drug products covered by the Service Agreement, which has resulted in the Company
owning 31.62% of Rgene.
As part of the Rgene Studies, the Company agreed
to loan $1.0 million to Rgene, for which Rgene has provided the Company with a 5% working capital convertible loan (the “Note”).
If the Note is fully converted, the Company will own an additional 6.4% of Rgene. The Company is expected to receive the outstanding loan
from the related party by the first half of 2024, either by cash or conversion of shares of Rgene. The Company may convert the Note at
any time into shares of Rgene’s common stock at either (i) a fixed conversion price equal to $1.00 per share or (ii) 20% discount
of the stock price of the then most recent offering, whichever is lower; the conversion price is subject to adjustment as set forth in
the Note. The Note includes standard events of default, as well as a cross default provision pursuant to which a breach of the Service
Agreement will trigger an event of default under the Note if not cured after 5 business days of written notice regarding the breach is
provided. Upon an event of default, the outstanding principal and any accrued and unpaid interest shall be immediately due and payable.
The Service Agreement shall remain in effect until
the expiration date of the last patent and automatically renew for 5 more years unless terminated earlier by either party with six months
written notice. Either party may terminate the Service Agreement for cause by providing 30 days written notice.
Rgene has further agreed, effective July 1, 2022,
to provide the Company with a seat on Rgene’s Board of Directors until the loan is repaid in full. The Company has nominated Dr.
Jiang, its Chief Strategy Officer and Director to occupy that seat; Dr. Jiang is also one of the Company’s largest shareholders,
owning 12.8% of the Company.
The Rgene Studies is a related party transaction.
Collaborative agreement with BioFirst Corporation,
a related party
On July 24, 2017, BriVision entered into a collaborative
agreement (the “BioFirst Collaborative Agreement”) with BioFirst Corporation (“BioFirst”), pursuant to which BioFirst
granted the Company the global licensing right for medical use of the product (the “Product”): BFC-1401 Vitreous Substitute
for Vitrectomy. BioFirst is a related party to the Company because a controlling beneficiary shareholder of YuanGene Corporation and the
Company is one of the directors and Common Stock shareholders of BioFirst (See Note 8).
Pursuant to the BioFirst Collaborative Agreement,
the Company will co-develop and commercialize the Product with BioFirst and pay BioFirst in a total amount of $3,000,000 in cash or stock
of the Company before September 30, 2018. The amount of $3,000,000 is in connection with the compensation for BioFirst’s past research
efforts and contributions made by BioFirst before the BioFirst Collaborative Agreement was signed and it does not relate to any future
commitments made by BioFirst and BriVision in this BioFirst Collaborative Agreement. In addition, the Company is entitled to receive 50%
of the future net licensing income or net sales profit, if any, and any development cost shall be equally shared by both BriVision and
BioFirst.
On September 25, 2017, BioFirst has delivered
all research, technical, data and development data to BriVision. The Company determined to fully expense the entire amount of $3,000,000
since currently the related licensing rights do not have alternative future uses. According to ASC 730-10-25-1, absent alternative future
uses the acquisition of product rights to be used in research and development activities must be charged to research and development expenses
immediately. Hence, the entire amount of $3,000,000 is fully expensed as research and development expense during the year ended December
31, 2017.
On June 30, 2019, BriVision entered into a Stock
Purchase Agreement (the “Purchase Agreement”) with BioFirst. Pursuant to the Purchase Agreement, the Company issued 428,571
shares of the Company’s common stock to BioFirst in consideration for $3,000,000 owed by the Company to BioFirst (the “Total
Payment”) in connection with a certain collaborative agreement between the Company and BioFirst dated July 24, 2017 (the “Collaborative
Agreement”). Pursuant to the Collaborative Agreement, BioFirst granted the Company the global licensing right to co-develop BFC-1401
or ABV-1701 Vitreous Substitute for Vitrectomy for medical purposes in consideration for the Total Payment.
On August 5, 2019, BriVision entered into a second
Stock Purchase Agreement (“Purchase Agreement 2”) with BioFirst. Pursuant to Purchase Agreement 2, the Company issued 414,702
shares of the Company’s common stock to BioFirst in consideration for $2,902,911 owed by the Company to BioFirst in connection with
a loan provided to BriVision from BioFirst.
On November 4, 2020, the Company executed an amendment
to the BioFirst Agreement with BioFirst to add ABV-2001 Intraocular Irrigation Solution and ABV-2002 Corneal Storage Solution to the agreement.
ABV-2002 is utilized during a corneal transplant procedure to replace a damaged or diseased cornea while ABV-2001 has broader utilization
during a variety of ocular procedures.
Initially the Company will focus on ABV-2002,
a solution utilized to store a donor cornea prior to either penetrating keratoplasty (full thickness cornea transplant) or endothelial
keratoplasty (back layer cornea transplant). ABV-2002 is a solution comprised of a specific poly amino acid that protects ocular tissue
from damage caused by external osmolarity exposure during pre-surgery storage. The specific polymer in ABV-2002 can adjust osmolarity
to maintain a range of 330 to 390 mOsM thereby permitting hydration within the corneal stroma during the storage period. Stromal hydration
results in (a) maintaining acceptable corneal transparency and (b) prevents donor cornea swelling. ABV-2002 also contains an abundant
phenolic phytochemical found in plant cell walls that provides antioxidant antibacterial properties and neuroprotection.
Early testing by BioFirst indicates that ABV-2002
may be more effective for protecting the cornea and retina during long-term storage than other storage media available today and can be
manufactured at lower cost. Further ABV-2002 product development was put on hold due the lack of funding.
In addition, BioFirst was incorporated on November
7, 2006, focusing on the R&D, manufacturing, and sales of innovative patented pharmaceutical products. The technology of BioFirst
comes from the global exclusive licensing from domestic R & D institutions. Currently, the main research and development product is
the vitreous substitute (Vitargus®) Licensed by the National Health Research Institutes. Vitargus is the world’s first bio-degradable
vitreous substitute and offers a number of advantages over current vitreous substitutes by minimizing medical complications and reducing
the need for additional surgeries.
Vitargus has started the construction of a GMP
factory in Hsinchu Biomedical Science Park, Taiwan, with the aim at building a production base to supply the global market, and promote
the construction of bio-degradable vitreous substitute manufacturing centers in Taiwan. Completion of this factory would allow ABVC to
manufacture Vitargus with world-class technology in a GMP certified pharmaceutical factory. BioFirst is targeting to complete the construction
in 2024.
The above-mentioned equity is before the reverse
stock split in 2023.
5. PROPERTY AND EQUIPMENT
Property and
equipment as of December 31, 2023 and 2022 are summarized as follows:
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Land | |
$ | 363,416 | | |
$ | 361,193 | |
| Construction-in-Progress | |
| 7,400,000 | | |
| - | |
| Buildings and leasehold improvements | |
| 2,227,431 | | |
| 2,226,687 | |
| Machinery and equipment | |
| 1,138,675 | | |
| 1,116,789 | |
| Office equipment | |
| 174,797 | | |
| 173,766 | |
| | |
| 11,304,319 | | |
| 3,878,435 | |
| Less: accumulated depreciation | |
| (3,335,041 | ) | |
| (3,304,457 | ) |
| Property and equipment, net | |
$ | 7,969,278 | | |
$ | 573,978 | |
Construction-in-progress consists of the property
recently acquired in Chengdu, China. The Company entered into a cooperation agreement on August 14, 2023, with Zhong Hui Lian He Ji Tuan,
Ltd. (the “Zhonghui”). Pursuant thereto, the Company acquired 20% of the ownership of certain property and a parcel of the
land, with a view to jointly develop the property into a healthcare center for senior living, long-term care, and medical care in the
areas of ABVC’s special interests, such as Ophthalmology, Oncology, and Central Nervous Systems. The plan is to establish a base
for the China market and global development of these interests.
The valuation of such property is US$37,000,000;
based on the Company’s 20% ownership, the Company acquired the value of US$7,400,000. In exchange, the Company issued to Zhonghui
an aggregate of 370,000 shares (the “Shares”) of common stock, at a per share price of $20.0. The Shares are subject to a
lock-up period of one year following the closing date of the transaction. In addition, the parties agreed that, after one year following
the closing of the transaction, if the market value of the Shares or the value of the Property increases or decreases, the parties will
negotiate in good faith to make reasonable adjustments.
The asset ownership certification is in the application
process. However, the Company’s ownership rights to the property and the associated land parcel, or a suitable replacement property,
are safeguarded under the terms of the cooperation agreement, which is legally binding and enforceable.
The Construction-in-progress is planned to finish
before the end of 2024.
Depreciation
expenses were $28,531 and $23,799 for the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023 and 2022,
Land with book value amounted to approximately $363,416 and $361,193, respectively, were pledged for obtaining bank loan (see Notes 9
Bank loans).
6. LONG-TERM INVESTMENTS
| (1) |
The ownership percentages of each investee are listed as follows: |
| | |
Ownership percentage | | |
|
| | |
December 31, | | |
December 31, | | |
Accounting |
| Name of related party | |
2023 | | |
2022 | | |
treatments |
| Braingenesis Biotechnology Co., Ltd. | |
| 0.17 | % | |
| 0.17 | % | |
Cost Method |
| Genepharm Biotech Corporation | |
| 0.67 | % | |
| 0.67 | % | |
Cost Method |
| BioHopeKing Corporation | |
| 5.90 | % | |
| 5.90 | % | |
Cost Method |
| BioFirst Corporation | |
| 18.68 | % | |
| 15.51 | % | |
Equity Method |
| Rgene Corporation | |
| 26.65 | % | |
| 26.65 | % | |
Equity Method |
| (2) |
The extent the investee relies on the company for its business are summarized as follows: |
| Name of related party |
|
The extent the investee relies on the Company for its business |
| Braingenesis Biotechnology Co., Ltd. |
|
No specific business relationship |
| Genepharm Biotech Corporation |
|
No specific business relationship |
| BioHopeKing Corporation |
|
Collaborating with the Company to develop and commercialize drugs |
| BioFirst Corporation |
|
Loaned from the investee and provides research and development support service |
| Rgene Corporation |
|
Collaborating with the Company to develop and commercialize drugs |
| (3) |
Long-term investment mainly consists of the following: |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Non-marketable Cost Method Investments, net | |
| | |
| |
| Braingenesis Biotechnology Co., Ltd. | |
$ | 7,213 | | |
$ | 7,169 | |
| Genepharm Biotech Corporation | |
| 22,021 | | |
| 21,887 | |
| BioHopeKing Corporation | |
| 818,018 | | |
| 813,014 | |
| Subtotal | |
| 847,252 | | |
| 842,070 | |
| Equity Method Investments, net | |
| | | |
| | |
| BioFirst Corporation(a) | |
| 1,680,488 | | |
| - | |
| Rgene Corporation(b) | |
| - | | |
| - | |
| Total | |
$ | 2,527,740 | | |
$ | 842,070 | |
| (a) |
BioFirst Corporation (the “BioFirst”): |
The
Company holds an equity interest in BioFirst Corporation, accounting for its equity interest using the equity method to accounts for
its equity investment as prescribed in ASC 323, Investments—Equity Method and Joint Ventures (“ASC 323”). Equity method
adjustments include the Company’s proportionate share of investee’s income or loss and other adjustments required by the
equity method. As of December 31, 2023 and 2022, the Company owns 18.68% and 15.51% common stock shares of BioFirst, respectively.
The Company made a prepayment for equity investment in BioFirst
to purchase additional shares to be issued by BioFirst in the aggregate amount of $2,688,578, recorded as prepayment for long-term investments
as of December 31, 2022. On July 19, 2023, the Company successfully completed the registration process for this investment. The initial
prepayment was $1,895,556, which is a portion of the prepayment as of December 31, 2022, and was converted into 994,450 shares of BioFirst
stock. As of December 31, 2023, the amount of prepayment for long-term investments in Biofirst is $1,124,842.
Summarized financial information for the Company’s
equity method investee, BioFirst, is as follows:
Balance Sheet
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Current Assets | |
$ | 1,451,877 | | |
$ | 1,543,151 | |
| Non-current Assets | |
| 686,206 | | |
| 739,472 | |
| Current Liabilities | |
| 2,286,058 | | |
| 2,663,051 | |
| Non-current Liabilities | |
| 347,193 | | |
| 103,447 | |
| Stockholders’ Equity | |
| (495,168 | ) | |
| (483,874 | ) |
Statement of operation
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Net sales | |
$ | 734 | | |
$ | 30,162 | |
| Gross profit | |
| 289 | | |
| 8,239 | |
| Net loss | |
| (1,194,797 | ) | |
| (1,274,539 | ) |
| Share of losses from investments accounted for using the equity method | |
| (221,888 | ) | |
| - | |
| (b) |
Rgene Corporation (the “Rgene”) |
Both Rgene and the Company are under
common control by Dr. Tsung-Shann Jiang, the CEO and chairman of the BioLite Inc. Since Dr. Tsung-Shann Jiang is able to exercise significant
influence, but not control, over the Rgene, the Company determined to use the equity method to accounts for its equity investment as prescribed
in ASC 323, Investments—Equity Method and Joint Ventures (“ASC 323”). Equity method adjustments include the Company’s
proportionate share of investee’s income or loss and other adjustments required by the equity method. As of December 31, 2023 and
2022, the Company owns 26.65% and 26.65% common stock shares of Rgene, respectively.
Summarized financial information for the Company’s
equity method investee, Rgene, is as follows:
Balance Sheets
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Current Assets | |
$ | 50,538 | | |
$ | 68,302 | |
| Non-current Assets | |
| 250,716 | | |
| 303,893 | |
| Current Liabilities | |
| 2,591,960 | | |
| 2,478,868 | |
| Non-current Liabilities | |
| 811 | | |
| 2,441 | |
| Shareholders’ Deficit | |
| (2,291,517 | ) | |
| (2,481,309 | ) |
Statement of operations
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Net sales | |
$ | - | | |
$ | - | |
| Gross Profit | |
| - | | |
| - | |
| Net loss | |
| (291,522 | ) | |
| (1,550,123 | ) |
| Share of loss from investments accounted for using the equity method | |
| - | | |
| - | |
| (4) |
Disposition of long-term investment |
During the years ended December 31,
2023 and 2022, there is no disposition of long-term investment.
| (5) | Loss on investment in equity securities |
The components of loss on investment in equity
securities for each period were as follows:
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Share of equity method investee losses | |
$ | (221,888 | ) | |
$ | - | |
7. CONVERTIBLE NOTES PAYABLE
On February 23, 2023, the Company entered into a securities purchase
agreement (the “Lind Securities Purchase Agreement”) with Lind Global Fund II, LP (“Lind”), pursuant to which
the Company issued Lind a secured, convertible note in the principal amount of $3,704,167 (the “Lind Offering”), for a purchase
price of $3,175,000 (the “Lind Note”), that is convertible into shares of the Company’s common stock at an initial conversion
price of $1.05 per share, subject to adjustment (the “Note Shares”). The Company also issued Lind a common stock purchase
warrant (the “Lind Warrant”) to purchase up to 5,291,667 shares of the Company’s common stock at an initial exercise
price of $1.05 per share for a period of 5 years, subject to adjustment that immediately upon such issuance or sale, the Exercise Price
in effect immediately prior to such issuance or sale shall be reduced (and in no event increased) to an Exercise Price equal to the consideration
per share paid for such Additional Shares of Common Stock. The warrants were valued using the Black-Scholes model. The fair value of the
warrants was determined to be $1,225,543, which was recorded to debt discount.
Beginning with the date that is six months from
the issuance date of the Lind Note and on each one (1) month anniversary thereafter, the Company shall pay Lind an amount equal to $308,650.58,
until the outstanding principal amount of the Lind Note has been paid in full prior to or on the Maturity Date or, if earlier, upon acceleration,
conversion or redemption of the Lind Note in accordance with the terms thereof (the “Monthly Payments”). At the Company’s
discretion, the Monthly Payments shall be made in (i) cash, (ii) shares of the Company’s common stock, or (iii) a combination of
cash and Shares; if made in shares, the number of shares shall be determined by dividing (x) the principal amount being paid in shares
by (y) 90% of the average of the 5 lowest daily VWAPs during the 20 trading days prior to the applicable payment date. The Lind Notes
sets forth certain conditions that must be satisfied before the Company may make any Monthly Payments in shares of common stock. If the
Company makes a Monthly Payment in cash, the Company must also pay Lind a cash premium of 5% of such Monthly Payment.
Upon the occurrence of any Event of Default (as
defined in the Lind Note), the Company must pay Lind an amount equal to 120% of the then outstanding principal amount of the Lind
Note (the “Mandatory Default Amount”), in addition to any other remedies under the Note or the other Transaction Documents.
The Company and Lind entered into a letter agreement on September 12, 2023, pursuant to which the Mandatory Default Amount was reduced
to 115% of the then outstanding principal amount of the Lind Note; pursuant to the letter agreement, Lind also agreed to waive any default
associated with the Company’s market capitalization being below $12.5 million for 10 consecutive days through February 23, 2024,
but retained its right to convert its Note. In addition, if the Company is unable to increase its market capitalization and is unable
to obtain a further waiver or amendment to the Lind Note, then the Company could experience an event of default under the Lind Note, which
could have a material adverse effect on the Company’s liquidity, financial condition, and results of operations. The Company cannot
make any assurances regarding the likelihood, certainty, or exact timing of the Company’s ability to increase its market capitalization,
as such metric is not within the immediate control of the Company and depends on a variety of factors outside the Company’s control.
The Lind Warrant may be exercised via cashless
exercise.
The warrant exercise price was
reset to $3.5 in accordance to the issuance of common stock in relation to securities purchase agreement on July 2023.
On November 17, 2023, the Company entered another
securities purchase agreement with Lind, pursuant to which the Company issued Lind a secured, convertible note in the principal amount
of $1,200,000, for a purchase price of $1,000,000, that is convertible into shares of the Company’s common stock at a conversion
price, which shall be the lesser of (i) $3.50 and (ii) 90% of the average of the three lowest VWAPs during the 20 trading days prior to
conversion. Lind will also receive a 5-year, common stock purchase warrant to purchase up to 1,000,000 shares of the Company’s common
stock at an initial exercise price of $2 per share for a period of 5 years. The warrants were valued using the Black-Scholes model. The
fair value of the warrants was determined to be $480,795, which was recorded to debt discount. An amendment was filed on February 29,
2024 to disclose that due to Nasdaq requirements, the parties entered into an amendment to the Note, pursuant to which the conversion
price shall have a floor price of $1.00 (the “Amendment”). Additionally, the Amendment requires the Company to make a cash
payment to Lind if in connection with a conversion, the conversion price is deemed to be the floor price.
As
of December 31, 2023 and 2022, the aggregate carrying values of the convertible debentures were $569,456 and $0, respectively; and accrued
convertible interest were both $0.
Total interest expenses in connection with the
above convertible note payable were $2,412,951 and $0 for the years ended December 31, 2023 and 2022, respectively.
8.
ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
Accrued expenses and other current liabilities
consisted of the following as of the periods indicated:
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Accrued research and development expense | |
$ | 1,799,583 | | |
$ | 1,600,221 | |
| Accrued compensation and employee benefits | |
| 1,184,505 | | |
| 568,865 | |
| Accrued royalties | |
| 274,028 | | |
| 272,352 | |
| Others | |
| 438,264 | | |
| 468,150 | |
| Total | |
$ | 3,696,380 | | |
$ | 2,909,587 | |
9. BANK LOANS
| (1) |
Short-term bank loans consists of the following: |
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Cathay United Bank | |
$ | 245,250 | | |
$ | 243,750 | |
| CTBC Bank | |
| 654,000 | | |
| 650,000 | |
| Cathay Bank | |
| - | | |
| 1,000,000 | |
| Total | |
$ | 899,250 | | |
$ | 1,893,750 | |
Cathay United Bank
On
June 28, 2016, BioLite Taiwan and Cathay United Bank entered into a one-year bank loan agreement (the “Cathay United Loan Agreement”)
in a credit limit amount of NT$7,500,000, equivalent to $245,250. The term started June 28, 2016 with maturity date at June 28, 2017.
The loan balance bears interest at a floating rate of prime rate plus 1.31%. The prime rate is based on term deposit saving interest
rate of Cathay United Bank. The Company renews the agreement with the bank every year. On September 6, 2022, BioLite Taiwan extended
the Cathay United Loan Agreement with the same principal amount of NT$7,500,000, equivalent to $245,250 for one year, which is due on
September 6, 2023. On September 6, 2023, BioLite Taiwan extended the Cathay United Loan Agreement with the same principal amount of NT$7,500,000,
equivalent to $245,250 for one year, which is due on September 6, 2024. As of December 31, 2023 and
December 31, 2022, the effective interest rates per annum was 2.87% and 2.67%, respectively. The loan is collateralized by the building
and improvement of BioLite Taiwan, and is also personal guaranteed by the Company’s chairman.
Interest expenses were $6,856 and $5,960 for the
years ended December 31, 2023 and 2022, respectively.
CTBC Bank
On June 12, 2017 and July 19, 2017, BioLite Taiwan
and CTBC Bank entered into two short-term saving secured bank loan agreements (the “CTBC Loan Agreements”) in a credit limit
amount of NT$10,000,000, equivalent to $327,000, and NT$10,000,000, equivalent to $327,000, respectively. Both two loans with the same
maturity date at January 19, 2018. In February 2018, BioLite Taiwan combined two loans and extended the loan contract with CTBC for one
year. The Company renews the agreement with the bank every year. The loan balances bear interest at a fixed rate of 2.5% per annum.
The loan is secured by the money deposited in a savings account with the CTBC Bank. This loan was also personal guaranteed by the Company’s
chairman and BioFirst. During the year ended December 31, 2020, BioLite Taiwan has opened a TCD account with CTBC bank to guarantee the
loan going forward.
Interest expenses were $15,610 and $12,220 for
the years ended December 31, 2023 and 2022, respectively.
Cathay Bank
On January 21, 2019, the Company received a loan
in the amount of $500,000 from Cathay Bank (the “Bank”) pursuant to a business loan agreement (the “Loan Agreement”)
entered by and between the Company and Bank on January 8, 2019 and a promissory note (the “Note”) executed by the Company
on the same day. The Loan Agreement provides for a revolving line of credit in the principal amount of $1,000,000 with a maturity date
(the “Maturity Date”) of January 1, 2020. The Note executed in connection with the Loan Agreement bears an interest rate (the
“Regular Interest Rate”) equal to the sum of one percent (1%) and the prime rate as published in the Wall Street Journal (the
“Index”) and the accrued interest shall become payable each month from February 1, 2019. Pursuant to the Note, the Company
shall pay the entire outstanding principal plus accrued unpaid interest on the Maturity Date and may prepay portion or all of the Note
before the Maturity Date without penalty. If the Company defaults on the Note, the default interest rate shall become five percent (5%)
plus the Regular Interest Rate.
In connection with the Note and Loan Agreement,
on January 8, 2019, each of Dr. Tsung Shann Jiang and Dr. George Lee, executed a commercial guaranty (the “Guaranty”) to guaranty
the loans for the Company pursuant to the Loan Agreement and Note, severally and individually, in the amount not exceeding $500,000 each
until the entire Note plus interest are fully paid and satisfied. Dr. Tsung Shann Jiang is the Chairman and Chief Executive Officer of
BioLite Holding, Inc. and Dr. George Lee serves as the Chairman of the board of directors of BioKey. On December 29, 2020, the Company
entered into a new loan extension agreement and assignment of deposit account with the Bank, which allowed Dr. Tsung Shann Jiang and Dr.
George Lee to be removed as guarantees from the list of Guaranty.
In addition, on January 8, 2019, each of the Company
and BioKey, a wholly-owned subsidiary of the Company, signed a commercial security agreement (the “Security Agreement”) to
secure the loans under the Loan Agreement and the Note. Pursuant to the Security Agreements, each of the Company and BioKey (each, a “Grantor”,
and collectively, the “Grantors”) granted security interest in the collaterals as defined therein, comprised of almost all
of the assets of each Grantor, to secure such loans for the benefit of the Bank. On June 30, 2020, the Company extended the Loan Agreement
with the same term for seven months, which is due on October 31, 2020. On April 8, 2020 and October 3, 2020, the Company repaid an aggregated
principal amount of $350,000. On December 3, 2020, the Company renewed the Loan Agreement with the principal amount of $650,000 for ten
months, which is due on October 31, 2021. On October 31, 2021, the Company renewed the Loan Agreement with the principal amount of $650,000
for twelve months, which is due on October 30, 2022. On September 24, 2021, the Cathay Bank has increased the line of credit to $1,000,000
from $650,000. The Loan Agreement was further extended and due on December 31, 2022. The outstanding loan balance was $1,000,000 as
of December 31, 2022. On February 23, 2023, the bank loan from Cathay Bank was fully repaid. As of December 31, 2023 and 2022, the effective interest rates per annum was 0% and 8%, respectively and the outstanding loan balance were $0 and $1,000,000.
Interest expenses were $10,209 and $46,957 for
the years ended December 31, 2023 and 2022, respectively.
10. RELATED PARTIES TRANSACTIONS
The related parties of the company with whom transactions
are reported in these financial statements are as follows:
| Name of entity or Individual |
|
Relationship with the Company and its subsidiaries |
| BioFirst Corporation (the “BioFirst”) |
|
Entity controlled by controlling beneficiary shareholder of YuanGene |
| BioFirst (Australia) Pty Ltd. (the “BioFirst (Australia)”) |
|
100% owned by BioFirst; Entity controlled by controlling beneficiary shareholder of YuanGene |
| Rgene Corporation (the “Rgene”) |
|
Shareholder of the Company; Entity controlled by controlling beneficiary shareholder of YuanGene; the Chairman of Rgene is Mr. Tsung-Shann Jiang |
| Eugene Jiang |
|
Former President and Chairman |
| GenePharm Inc. (the “GenePharm”) |
|
Dr. George Lee, Board Director of Biokey, is the Chairman of GenePharm. |
| The Jiangs |
|
Mr. Tsung-Shann Jiang, the controlling beneficiary shareholder of the Company and Rgene, the Chairman and CEO of the BioLite Holding Inc. and BioLite Inc. and the President and a member of board of directors of BioFirst
Ms. Shu-Ling Jiang, Mr. Tsung-Shann Jiang’s wife, is the Chairman of Keypoint; and a member of board of directors of BioLite Inc.
Mr. Eugene Jiang is Mr. and Ms. Jiang’s son. Mr. Eugene Jiang is the chairman, and majority shareholder of the Company and a member of board of directors of BioLite Inc.
Mr. Chang-Jen Jiang is Mr. Tsung-Shann Jiang’s sibling and the director of the Company. Ms. Mei-Ling Jiang is Ms. Shu-Ling Jiang’s sibling. |
| Zhewei Xu |
|
Shareholder of the Company. |
| BioHopeKing Corporation |
|
Entity controlled by controlling beneficiary shareholder of ABVC |
| Jaimes Vargas Russman |
|
CEO of AiBtl BioPharma Inc. |
Accounts receivable - related parties
Accounts receivable due from related parties consisted
of the following as of the periods indicated:
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| GenePharm Inc. | |
$ | - | | |
$ | 142,225 | |
| Rgene | |
| 10,463 | | |
| 615,118 | |
| Total | |
$ | 10,463 | | |
$ | 757,343 | |
Revenue - related parties
Revenue due from related parties consisted of
the following as of the periods indicated:
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Rgene | |
$ | 2,055 | | |
$ | 904,043 | |
| Total | |
$ | 2,055 | | |
$ | 904,043 | |
Due from related parties
Amount due from related parties consisted of the
following as of the periods indicated:
Due from related party- Current
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Rgene | |
$ | 541,486 | | |
$ | 513,819 | |
| BioFirst | |
| 206,087 | | |
| - | |
| Total | |
$ | 747,573 | | |
$ | 513,819 | |
Due from related parties- Non-current, net
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| BioFirst (Australia) | |
$ | 839,983 | | |
| $ 752,655 | |
| BioHopeKing Corporation | |
| 113,516 | | |
| 112,822 | |
| Total | |
| 953,499 | | |
| 865,477 | |
| Less: allowance for expected credit losses accounts | |
| (839,983 | ) | |
| - | |
| Net | |
$ | 113,516 | | |
$ | 865,477 | |
| (1) |
On June 16, 2022, the Company entered into a one-year
convertible loan agreement with Rgene, with a principal amount of $1,000,000 to Rgene which bears interest at 5% per annum for the use
of working capital that, if fully converted, would result in ABVC owning an additional 6.4% of Rgene. The Company may convert the Note
at any time into shares of Rgene’s common stock at either (i) a fixed conversion price equal to $1.00 per share or (ii) 20% discount
of the stock price of the then most recent offering, whichever is lower; the conversion price is subject to adjustment as set forth in
the Note. The Note includes standard events of default, as well as a cross-default provision pursuant to which a breach of the Service
Agreement will trigger an event of default under the convertible note if not cured after 5 business days of written notice regarding the
breach is provided.
As of December 31, 2023 and December 31,
2022, the outstanding loan balance were both $500,000; and accrued interest was $38,819 and $13,819.
As of December 31, 2023, the Company has other receivables amounted
$2,667 from Rgene due to daily operations. |
| (2) |
On July 1, 2020, the Company entered into a loan agreement with BioFirst (Australia) for $361,487 to properly record R&D cost and tax refund allocation based on co-development contract executed on July 24, 2017. The loan was originally set to be mature on September 30, 2021 with an interest rate of 6.5% per annum, but on September 7, 2021, the Company entered into a loan agreement with BioFirst (Australia) for $67,873 to meet its new project needs. On July 27, 2021, the Company repaid a loan 249,975 to BioFirst (Australia). On December 1, 2021, the Company entered into a loan agreement with BioFirst (Australia) for $250,000 to increase the cost for upcoming projects. The loan will be matured on November 30, 2022 with an interest rate of 6.5% per annum. In 2022, the Company entered into several loan agreements with BioFirst (Australia) for a total amount of $507,000 to increase the cost for upcoming projects. During the first quarter of 2023, the Company entered into several loan agreements with BioFirst (Australia) for a total amount of $88,091 to increase the cost for upcoming projects. During the second quarter of 2023, the Company entered into several loan agreements with BioFirst (Australia) for a total amount of $25,500 to increase the cost for upcoming projects. All the loans period was twelve months with an interest rate of 6.5% per annum. For accounting purpose, the due from and due to related party balances was being net off. As of December 31, 2023 and December 31, 2022, the outstanding loan balance and allocated research fee was $681,185 and $660,484, respectively; and accrued interest was $158,798 and $92,171, respectively. The outstanding amount was settled in 2023.
The balances mainly represent advances to BioFirst (Australia) for research and development purposes. The business conditions of BioFirst (Australia) deteriorated and, as a result, the Company recognized expected credit losses of $839,983 for the year ended December 31, 2023. |
| (3) |
On February 24, 2015, BioLite Taiwan and BioHopeKing Corporation (the “BHK”) entered into a co-development agreement, (the “BHK Co-Development Agreement”, see Note 4). The development costs shall be shared 50/50 between BHK and the Company. Under the term of the agreement, BioLite issued relevant development cost to BHK. As of December 31, 2023 and 20212 due from BHK was $113,516 and $112,822, respectively. |
Due to related parties
Amount due to related parties consisted of the
following as of the periods indicated:
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| BioFirst | |
$ | - | | |
$ | 188,753 | |
| The Jiangs | |
| 19,789 | | |
| 19,789 | |
| Due to shareholders | |
| 152,382 | | |
| 151,450 | |
| Due to a Director | |
| 961 | | |
| - | |
| Total | |
$ | 173,132 | | |
$ | 359,992 | |
| (1) |
Since 2019, BioFirst has advanced funds to the Company for working
capital purpose. The advances bear interest 1% per month (or equivalent to 12% per annum). As of December 31, 2022, the aggregate amount
of outstanding balance and accrued interest is $188,753, a combination of $147,875 from loan, and $40,878 from expense-sharing. The outstanding
amount was being net off with amount due from BioFirst in 2023.
|
| (2) |
Since 2019, the Jiangs advanced funds to the Company for working capital purpose. As of December 31, 2023 and 2022, the outstanding balance due to the Jiangs amounted to $19,789 and $19,789, respectively. These loans bear interest rate of 0% to 1% per month, and are due on demand. |
| (3) |
Since 2018, the Company’s shareholders have advanced funds to the Company for working capital purpose. The advances bear interest rate from 12% to 13.6224% per annum. As of December 31, 2023 and 2022, the outstanding principal and accrued interest was $152,382 and $151,450, respectively. Interest expenses in connection with these loans were $20,094 and $21,378 for the years ended December 31, 2023 and 2022, respectively. |
| (4) |
As of December 31, 2023, due to a Director amounted $961 was related to the entity setup fee paid by the Director of AiBtl BioPharma Inc. on behalf of the entity. |
11. INCOME TAXES
Income tax expense for the years ended
December 31, 2023 and 2022 consisted of the following:
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Current: | |
| | |
| |
| Federal | |
$ | - | | |
$ | - | |
| State | |
| - | | |
| 2,400 | |
| Foreign | |
| 140,338 | | |
| - | |
| Total Current | |
$ | 140,338 | | |
$ | 2,400 | |
| Deferred: | |
| | | |
| | |
| Federal | |
$ | - | | |
$ | - | |
| State | |
| - | | |
| - | |
| Foreign | |
| 115,668 | | |
| 795,378 | |
| Total Deferred | |
$ | 115,668 | | |
$ | 795,378 | |
| Total provision for income taxes | |
$ | 256,006 | | |
$ | 797,778 | |
Deferred
tax assets (liability) as of December 31, 2023 and 2022 consist approximately of:
| | |
December 31, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Loss on impairment of Assets | |
| 713,223 | | |
| 709,961 | |
| Net operating loss carryforwards | |
| 5,568,391 | | |
| 5,866,623 | |
| Tax credit of investment | |
| - | | |
| - | |
| Operating lease liabilities | |
| 213,482 | | |
| 213,482 | |
| Operating lease assets | |
| (213,482 | ) | |
| (213,482 | ) |
| Deferred tax assets, Gross | |
| 6,281,614 | | |
| 6,576,584 | |
| Valuation allowance | |
| (6,281,614 | ) | |
| (6,459,474 | ) |
| Deferred tax assets, net | |
| - | | |
| 117,110 | |
12. EQUITY
In January 2022, the Company agreed to pay the
deferred service fees related to Public Offering amounted $4,296,763 by issuing 1,306,007 shares of unrestricted common stock, valued
at $3.29 per share on the grant date. These shares have been issued in January 2022.
In March 2022, the Company issued 75,000 common
stock to BarLew Holdings, LLC for consulting and advisory services amounted to $169,500, valued at $2.26 per share.
In May 2022, the Company and an institutional
investor entered into certain securities purchase agreement relating to the offer and sale of 2,000,000 shares of common stock at an offering
price of $2.11 per share in a registered direct offering. The shares of the Company’s common stock were issued for gross proceeds
of $4,220,000, before placement agent fees and legal fees of $556,075. Pursuant to the offering, the Company will also issue 5-year warrants
to purchase 2,000,000 shares of common stock, exercisable at a price of $2.45 per share. As of December 31, 2023, these warrants have
been issued but not exercised.
On July 10, 2022, the Board approved the issuance
of 75,000 shares of common stock to Barlew Holdings, LLC pursuant to the consulting agreement by and between Barlew Holdings, LLC and
the Company dated July 1, 2022, and 250,000 shares of common stock to Inverlew Advisors, LLC, in accordance with the consulting agreement
by and between Inverlew Advisors, LLC and the Company dated July 1, 2022.
On December 1, 2022, the Company issued 125,000 and 100,000 common
stock to Euro-Asia Investment & Finance Corp Ltd. and Thalia Media Ltd. for consulting and advisory services.
On January 3, 2023, the Company issued 223,411 common
stock to a consultant for providing consulting services on listing to NASDAQ in 2021.
On February 23, 2023, the Company entered into a securities purchase
agreement with Lind Global Fund II, LP (“Lind”), pursuant to which the Company issued Lind a secured, convertible note in
the principal amount of $3,704,167, for a purchase price of $3,175,000, that is convertible into shares of the Company’s common
stock at an initial conversion price of $1.05 per share, subject to adjustment. The Company also issued Lind a common stock purchase
warrant to purchase up to 5,291,667 shares of the Company’s common stock at an initial exercise price of $1.05 per
share for a period of 5 years, subject to adjustment that immediately upon such issuance or sale, the Exercise Price in effect immediately
prior to such issuance or sale shall be reduced (and in no event increased) to an Exercise Price equal to the consideration per share
paid for such Additional Shares of Common Stock. The warrants were valued using the Black-Scholes model. The fair value of the warrants
was determined to be $1,225,543, which was recorded to debt discount. During the year ended December 31, 2023, the Company has been repaying
Lind with securities for 3,732,167 shares, totaling $3,306,112.
The warrant exercise price was reset to $3.5 in
accordance to the issuance of common stock in relation to securities purchase agreement on July 2023. As of December 31, 2023, the warrant
has not yet been exercised.
On
July 27, 2023, the Company entered into that certain securities purchase agreement. relating to the offer and sale of 300,000 shares of
common stock, par value $0.001 per share and 200,000 pre-funded warrants, at an exercise price of $0.001 per share, in a registered direct
offering. Pursuant to the Purchase Agreement, the Company agreed to sell the Shares and/or Pre-funded Warrants at a per share purchase
price of $3.50, for gross proceeds of $1,750,000, before deducting any estimated offering expenses. On August 1, 2023, 200,000 pre-funded
warrants were exercised.
The above-mentioned equity is before the reverse stock
split in July 2023.
On
August 14, 2023, the Company entered into a cooperation agreement with Zhonghui. Pursuant thereto, the Company acquired 20% of the ownership
of a property and the parcel of the land owned by Zhonghui in Leshan, Sichuan, China. During the third quarter of 2023, the Company issued
to Zhonghui, an aggregate of 370,000 shares of the Company’s common stock, at a per share price of $20. The Company also issued 29,600 common stock to consultants for providing
consulting services on the above transaction.
On November 17, 2023, the Company entered another
securities purchase agreement with Lind, pursuant to which the Company issued Lind a secured, convertible note in the principal amount
of $1,200,000, for a purchase price of $1,000,000, that is convertible into shares of the Company’s common stock at a conversion
price, which shall be the lesser of (i) $3.50 and (ii) 90% of the average of the three lowest VWAPs during the 20 trading days prior to
conversion. Lind will also receive a 5-year, common stock purchase warrant to purchase up to 1,000,000 shares of the Company’s common
stock at an initial exercise price of $2 per share for a period of 5 years. The warrants were valued using the Black-Scholes model. The
fair value of the warrants was determined to be $480,795, which was recorded to debt discount. An amendment was filed on February
29, 2024 to disclose that due to Nasdaq requirements, the parties entered into an amendment to the Note, pursuant to which the conversion
price shall have a floor price of $1.00 (the “Amendment”). Additionally, the Amendment requires the Company to make a cash
payment to Lind if in connection with a conversion, the conversion price is deemed to be the floor price.
13. STOCK OPTIONS
On October 30, 2020, the Company issued an aggregate
of 545,182 shares of common stock in lieu of unpaid salaries of certain employees and unpaid consulting fees under the 2016 Equity Incentive
Plan, as amended, at a conversion price of $2 per share; the total amount of converted salaries and consulting fees was $1,090,361. On
November 21, 2020, the Company entered into acknowledgement agreements and stock option purchase agreements with these
employees and consultant; pursuant to which the Company granted stock options to purchase 545,182 shares of the Company’s common
stock in lieu of common stock. The options were vested at the grant date and become exercisable for 10 years from the grant date.
On October 15, 2021, the Company entered into
stock option agreements with 11 directors and 3 employees, pursuant to which the Company granted options to purchase an aggregate of 1,280,002
shares of common stock under the 2016 Equity Incentive Plan, as amended, at an exercise price of $3 per share. The options were vested
at the grant date and become exercisable for 10 years from the grant date.
On April 16, 2022, the Company entered into stock
option agreements with 5 directors, pursuant to which the Company agreed to grant options to purchase an aggregate of 761,920 shares of
common stock under the 2016 Equity Incentive Plan, at an exercise price of $3 per share, exercisable for 10 years from the grant date.
Options issued and outstanding as of December
31, 2023, and their activities during the year then ended are as follows:
| | |
| | |
| | |
Weighted- | | |
| |
| | |
| | |
Weighted- | | |
Average | | |
| |
| | |
| | |
Average | | |
Contractual | | |
| |
| | |
Number of | | |
Exercise | | |
Life | | |
Aggregate | |
| | |
Underlying
Shares | | |
Price
Per Share | | |
Remaining
in Years | | |
Intrinsic
Value | |
| Outstanding as of January 1, 2023 | |
| 2,587,104 | | |
$ | 2.79 | | |
| 8.74 | | |
| - | |
| Granted | |
| - | | |
| - | | |
| - | | |
| - | |
| Forfeited | |
| - | | |
| - | | |
| - | | |
| - | |
| Outstanding as of December 31, 2023 | |
| 2,587,104 | | |
| 2.79 | | |
| 7.74 | | |
$ | - | |
| Exercisable as of December 31, 2023 | |
| 2,587,104 | | |
| 2.79 | | |
| 7.74 | | |
$ | - | |
| Vested and expected to vest | |
| 2,587,104 | | |
$ | 2.79 | | |
| 7.74 | | |
$ | - | |
The fair value of stock options granted for the
years ended December 31, 2023 and 2022 was calculated using the Black-Scholes option-pricing model applying the following assumptions:
| | |
Year ended
December 31 | |
| | |
2022 | |
| | |
| |
| Risk free interest rate | |
| 2.79 | % |
| Expected term (in years) | |
| 5.00 | |
| Dividend yield | |
| 0 | % |
| Expected volatility | |
| 83.86 | % |
The
Company granted options to purchase 0 and 761,920 shares of common stock to employees and certain consultants during
the years ended December 31, 2023 and 2022, respectively. The weighted average grant date fair value of options
granted during the years ended December 31, 2023 and 2022 was $2.79 and
$2.79, respectively. There are 3,860,211 options available for grant under the 2016 Equity Incentive Plan as of December 31, 2023.
Compensation costs associated with the Company’s stock options are recognized, based on the grant-date fair values of these options
over vesting period. Accordingly, the Company recognized stock-based compensation expense of $0 and $1,241,930 for the years ended December
31, 2023 and 2022, respectively. As of December 31, 2023 and 2022, there were no unvested options. There were no options exercised during
the years ended December 31, 2023 and 2022.
The
above-mentioned equity is before the reverse stock split in July 2023.
14. LOSS PER SHARE
Basic loss per share is computed by dividing net
loss by the weighted-average number of common shares outstanding during the year. Diluted loss per share is computed by dividing net loss
by the weighted-average number of common shares and dilutive potential common shares outstanding during the years ended December 31, 2023
and 2022.
| | |
For the Year Ended | |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Numerator: | |
| | |
| |
| Net loss attributable to ABVC’s common stockholders | |
$ | (10,856,656 | ) | |
$ | (16,423,239 | ) |
| | |
| | | |
| | |
| Denominator: | |
| | | |
| | |
| Weighted-average shares outstanding: | |
| | | |
| | |
| Weighted-average shares outstanding - Basic | |
| 4,335,650 | | |
| 3,166,460 | |
| Stock options | |
| | | |
| | |
| Weighted-average shares outstanding - Diluted | |
| 4,335,650 | | |
| 3,166,460 | |
| | |
| | | |
| | |
| Loss per share | |
| | | |
| | |
| -Basic | |
$ | (2.43 | ) | |
$ | (5.19 | ) |
| -Diluted | |
$ | (2.43 | ) | |
$ | (5.19 | ) |
Diluted loss per share takes into account the
potential dilution that could occur if securities or other contracts to issue Common Stock were exercised and converted into Common Stock.
15. LEASE
The Company adopted FASB Accounting Standards
Codification, Topic 842, Leases (“ASC 842”) using the modified retrospective approach, electing the practical expedient that
allows the Company not to restate its comparative periods prior to the adoption of the standard on January 1, 2019.
The Company applied the following practical expedients
in the transition to the new standard and allowed under ASC 842:
| |
● |
Reassessment of expired or existing contracts: The Company elected not to reassess, at the application date, whether any expired or existing contracts contained leases, the lease classification for any expired or existing leases, and the accounting for initial direct costs for any existing leases. |
| |
● |
Use of hindsight: The Company elected to use hindsight in determining the lease term (that is, when considering options to extend or terminate the lease and to purchase the underlying asset) and in assessing impairment of right-to-use assets. |
| |
● |
Reassessment of existing or expired land easements: The Company elected not to evaluate existing or expired land easements that were not previously accounted for as leases under ASC 840, as allowed under the transition practical expedient. Going forward, new or modified land easements will be evaluated under ASU No. 2016-02. |
| |
● |
Separation of lease and non- lease components: Lease agreements that contain both lease and non-lease components are generally accounted for separately. |
| |
● |
Short-term lease recognition exemption: The Company also elected the short-term lease recognition exemption and will not recognize ROU assets or lease liabilities for leases with a term less than 12 months. |
The new leasing standard requires recognition
of leases on the consolidated balance sheets as right-of-use (“ROU”) assets and lease liabilities. ROU assets represent the
Company’s right to use underlying assets for the lease terms and lease liabilities represent the Company’s obligation to make
lease payments arising from the leases. Operating lease ROU assets and operating lease liabilities are recognized based on the present
value and future minimum lease payments over the lease term at commencement date. The Company’s future minimum based payments used
to determine the Company’s lease liabilities mainly include minimum based rent payments. As most of Company’s leases do not
provide an implicit rate, the Company uses its estimated incremental borrowing rate based on the information available at commencement
date in determining the present value of lease payments.
The Company recognized lease liabilities, with
corresponding ROU assets, based on the present value of unpaid lease payments for existing operating leases longer than twelve months.
The ROU assets were adjusted per ASC 842 transition guidance for existing lease-related balances of accrued and prepaid rent, unamortized
lease incentives provided by lessors, and restructuring liabilities. Operating lease cost is recognized as a single lease cost on a straight-line
basis over the lease term and is recorded in Selling, general and administrative expenses. Variable lease payments for common area maintenance,
property taxes and other operating expenses are recognized as expense in the period when the changes in facts and circumstances on which
the variable lease payments are based occur.
The Company has no finance leases. The Company’s
leases primarily include various office and laboratory spaces, copy machine, and vehicles under various operating lease arrangements.
The Company’s operating leases have remaining lease terms of up to approximately five years.
| | |
December 31,
2023 | | |
December 31,
2022 | |
| ASSETS | |
| | |
| |
| Operating lease right-of-use assets | |
$ | 809,283 | | |
$ | 1,161,141 | |
| LIABILITIES | |
| | | |
| | |
| Operating lease liabilities (current) | |
| 401,826 | | |
| 369,314 | |
| Operating lease liabilities (non-current) | |
| 407,457 | | |
| 791,827 | |
Supplemental Information
The following provides details of the Company’s
lease expenses:
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Operating lease expenses | |
$ | 358,576 | | |
$ | 358,576 | |
Other information related to leases is presented
below:
| | |
Year Ended
December 31, | |
| | |
2023 | | |
2022 | |
| Cash paid for amounts included in the measurement of operating lease liabilities | |
$ | 385,659 | | |
$ | 358,576 | |
| | |
December 31,
2023 | | |
December 31,
2022 | |
| Weighted Average Remaining Lease Term: | |
| | |
| |
| Operating leases | |
| 1.73 years | | |
| 2.48 years | |
| | |
| | | |
| | |
| Weighted Average Discount Rate: | |
| | | |
| | |
| Operating leases | |
| 1.5 | % | |
| 1.49 | % |
The minimum future annual payments under non-cancellable
leases during the next five years and thereafter, at rates now in force, are as follows:
| |
|
Operating leases |
|
| 2024 |
|
$ |
404,745 |
|
| 2025 |
|
|
351,352 |
|
| 2026 |
|
|
56,916 |
|
| 2027 |
|
|
- |
|
| Thereafter |
|
|
- |
|
| Total future minimum lease payments, undiscounted |
|
|
813,013 |
|
| Less: Imputed interest |
|
|
(3,730 |
) |
| Present value of future minimum lease payments |
|
$ |
809,283 |
|
16. COMMITMENTS AND CONTINGENCIES
Contingencies
In the ordinary course of business, the Company
may be subject to legal proceedings regarding contractual and employment relationships and a variety of other matters. The Company records
contingent liabilities resulting from such claims, when a loss is assessed to be probable, and the amount of the loss is reasonably estimable.
In the opinion of management, there were no pending or threatened claims and litigation as of December 31, 2023 and up through March 13,
2024, date of the consolidated financial statements were available to the issued.
17. ACQUISITION
On November 12, 2023, the Company and one of its
subsidiaries, BioLite, Inc. (“BioLite Taiwan”) each entered into a multi-year, global licensing agreement with AiBtl
BioPharma Inc. (“AIBL”, or the acquired company) for the Company and BioLite Taiwan’s CNS drugs with the indications
of MDD (Major Depressive Disorder) and ADHD (Attention Deficit Hyperactivity Disorder) (collectively, the “Licensed Products”).
The potential license will cover the Licensed Products’ clinical trial, registration, manufacturing, supply, and distribution rights.
The parties are determined to collaborate on the global development of the Licensed Products. The parties are also working to strengthen
new drug development and business collaboration, including technology, interoperability, and standards development. As per each of the
respective agreements, each of ABVC and BioLite Taiwan received 23 million shares of AIBL stock and as a result, the Company has a controlling
interest over AIBL. If certain milestones are met, the Company and BioLite Taiwan are each eligible to receive $3,500,000 and royalties
equaling 5% of net sales, up to $100 million.
The
Company concluded the assets acquired and liabilities assumed did not meet the definition of a business as a limited number of
inputs were acquired but no substantive business processes or signs of output were acquired. As such, the acquisition was accounted
for as an asset purchase. The purchase consideration was nonmonetary assets (patent) and
transfer on November12, 2023. The equity interest transferred to ABVC and BioLite Taiwan on December 15, 2023.
| Cash and cash equivalents | |
$ | - | |
| Total assets acquired | |
| - | |
| Accrued expense | |
| (243,888 | ) |
| Due
to Director | |
| (498 | ) |
| Total liabilities acquired | |
| (243,386 | ) |
| Total consideration (Intangible assets) | |
| - | |
18. SUBSEQUENT EVENTS
On January 12, 2024, BioLite Taiwan extended the
CTBC Loan Agreement with the same principal amount of NT$20,000,000, equivalent to $654,000 for one year.
On January 17, 2024, the Company entered another
securities purchase agreement with Lind, pursuant to which the Company issued Lind a secured, convertible note in the principal amount
of $1,000,000, for a purchase price of $833,333, that is convertible into shares of the Company’s common stock at a conversion
price, which shall be the lesser of (i) $3.50 and (ii) 90% of the average of the three lowest VWAPs during the 20 trading days prior
to conversion. Lind will also receive a 5-year, common stock purchase warrant to purchase up to 1,000,000 shares of the Company’s
common stock at an initial exercise price of $2 per share.
An amendment was filed on February 29, 2024 to disclose that due to
Nasdaq requirements, the parties entered into an amendment to the Note, pursuant to which the conversion price shall have a floor price
of $1.00 (the “Amendment”). Additionally, the Amendment requires the Company to make a cash payment to Lind if in connection
with a conversion, the conversion price is deemed to be the floor price.
On January 27, 2024, the company granted 1,241,615
restricted shares to its employees and directors under the 2016 Equity Incentive Plan, with an issuance date of February 2, 2024. These
shares are subject to a three-year restriction period.
On February 6, 2024, the Company entered into a definitive agreement
with Shuling Jiang (“Shuling”), pursuant to which Shuling shall transfer the ownership of certain land she owns located
at Taoyuan City, Taiwan (the “Land”) to the Company (the “Agreement”). In consideration for the
Land, the Company issued Shuling (i) 703,495 restricted shares of the Company’s common stock (the “Shares”) at
a price of $3.50 per share and (ii) five-year warrants to purchase up to 1,000,000 shares of the Company’s common stock, with an
exercise price of $2.00 per share.
The Company has assessed all events from December
31, 2023, up through March 13, 2024, which is the date that these consolidated financial statements are available to be issued, Other
than the events disclosed above, no other subsequent events have occurred that would require recognition or disclosure in the Company’s
consolidated financial statements.
ABVC BioPharma (NASDAQ:ABVC)
Historical Stock Chart
Von Mai 2024 bis Jun 2024
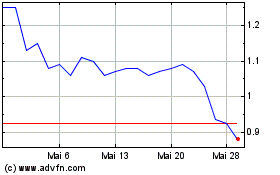
ABVC BioPharma (NASDAQ:ABVC)
Historical Stock Chart
Von Jun 2023 bis Jun 2024