HP Delivers Innovations to Public Health with New CDC Pilot Program
27 August 2018 - 5:01PM

News highlights
Today HP Inc. announced its participation in a pilot program run by
the Centers for Disease Control and Prevention (CDC) that aims to
accelerate the testing of new antibiotics designed to fight
antimicrobial resistant bacteria. The pilot uses HP technology to
“print” pharmaceutical samples. As part of the pilot, CDC will
deploy new HP D300e Digital Dispenser BioPrinters to four regional
labs in the Antibiotic Resistance (AR) Lab Network, to develop
antimicrobial susceptibility test methods for new drugs. For the
first time, regional labs will be able to conduct rapid
susceptibility testing for health departments and hospitals
nationwide.
“Bacteria continuously develop new ways to resist
antibiotics—once a drug is approved for use, the countdown begins
until resistance emerges. In fact, resistance has even been
detected before FDA approval,” said Jean Patel, PH.D. D (ABMM),
Science Team Lead, Antibiotic Resistance Coordination and Strategy
Unit at CDC. “To save lives and protect people, it is vital to make
technology accessible to hospital labs nationwide. We hope this
pilot will help ensure our newest drugs last longer and put
gold-standard lab results in healthcare providers’ hands
faster.”
Antimicrobial resistance is an international public health
issue. New antibiotics for resistant bacteria can help save lives,
but hospitals often do not have access to antimicrobial
susceptibility testing to know if the new drug might be
effective. The HP printer helps to speed up testing
availability at the local level by “printing” the gold standard
test plates in a few minutes. When testing is not available, new
drugs can be either overutilized, contributing to antimicrobial
resistance, underutilized, not helping patients in need, or
misutilized, which can lead to side effects or prolonged
treatment.
How it WorksThe BioPrinter accurately dispenses
or “prints” volumes from picoliters to microliters for faster, more
reliable dispensing of small molecules and biomolecules to enable
drug discovery, genomics, and proteomics research. The
following labs within the AR Lab Network will use this technology
to develop and perform antimicrobial susceptibility testing for new
drugs: New York, Minnesota, Tennessee and Wisconsin. This new
capacity can be used by healthcare providers for patient care while
commercial methods commonly used in hospital laboratories are
developed and implemented.
Testing through CDC will be implemented in the regional
laboratories in the first quarter of FY19. CDC will prioritize
testing for highly resistant bacteria, with pan-resistant bacteria
being the top priority. HP will coordinate with CDC to evaluate the
pilot in the regional laboratories of the AR Regional Lab Network,
refine the system, and explore and support a larger rollout of
inkjet printing technology in labs nationwide.
To learn more, visit: http://www.hp.com/go/bioprint.
About HPHP Inc. creates technology that makes
life better for everyone, everywhere. Through our portfolio of
printers, PCs, mobile devices, solutions and services, we engineer
experiences that amaze. More information about HP Inc. is available
at http://www.hp.com.
© 2018 HP Development Company LP. The information contained
herein is subject to change without notice. The only warranties for
HP Inc. products and services are set forth in the express warranty
statements accompanying such products and services. Nothing herein
should be construed as constituting an additional warranty. HP Inc.
shall not be liable for technical or editorial errors or omissions
contained herein.
|
Lacey Haines,
HPlacey.haines@hp.com https://press.ext.hp.com/us/en/press-kits.html |
|
|
Photos accompanying this announcement are available
athttp://www.globenewswire.com/NewsRoom/AttachmentNg/01f2e198-f486-409c-b771-5d2d6957dd5d
http://www.globenewswire.com/NewsRoom/AttachmentNg/901abb39-5c62-4dda-8244-65f60ce94bfc
http://www.globenewswire.com/NewsRoom/AttachmentNg/a948b9cc-34fd-4cf1-b3df-977e6dacd583A
video accompanying this release is available
athttp://www.globenewswire.com/NewsRoom/AttachmentNg/d8e8e55d-29a6-461e-8388-aefbe4bcb53f
HP (NYSE:HPQ)
Historical Stock Chart
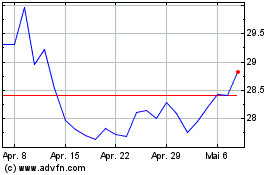
Von Jun 2024 bis Jul 2024
HP (NYSE:HPQ)
Historical Stock Chart
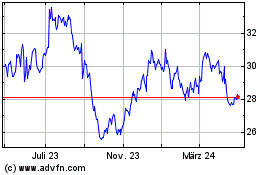
Von Jul 2023 bis Jul 2024