-
CAHtalyst™ Adult Phase 3 Study Met Primary and Important Key
Secondary Endpoints, with Crinecerfont Treatment Decreasing
Androstenedione Levels and Enabling Glucocorticoid Dose Reduction
While Maintaining Androstenedione Control
- 62.7% of
Crinecerfont Participants Achieved a Physiologic Glucocorticoid
Dose while Maintaining Androstenedione Control versus 17.5% of
Placebo Participants
-
Favorable Trends in Endpoints Reflecting Consequences of
Supraphysiologic Glucocorticoid Dosing
-
Crinecerfont was Generally Well Tolerated
SAN
DIEGO, June 3, 2024 /PRNewswire/ -- Neurocrine
Biosciences, Inc. (Nasdaq: NBIX) today announced that the primary
study results from its CAHtalyst™ Phase 3 study investigating
crinecerfont for the treatment of adults ages 18 and older with
congenital adrenal hyperplasia (CAH) due to 21-hydroxylase
deficiency have been published in The New England Journal of
Medicine online edition and will appear in a future print issue
of the journal. The study met the primary and important key
secondary endpoints related to androgen reduction (during an
initial glucocorticoid-stable period) and glucocorticoid (GC) dose
reduction while maintaining androgen control. Favorable trends were
observed with endpoints that reflect the consequences of long-term
supraphysiologic glucocorticoid therapy. In addition to being
published in The New England Journal of Medicine, CAHtalyst
Adult Phase 3 data were presented at ENDO 2024 in an oral
presentation by Dr. Richard
Auchus.
"Since the 1950s, glucocorticoids have been required not
only for cortisol replacement, but also to control the excessive
amount of adrenal androgens in patients with congenital adrenal
hyperplasia. As a result, CAH patients suffer from a higher
prevalence of disorders attributable to supraphysiologic GC levels,
including osteoporosis, obesity, insulin resistance, diabetes
mellitus, hyperlipidemia and hypertension," said Dr. Richard Auchus, M.D., Ph.D., Principal
Investigator, Professor of Pharmacology and Internal Medicine,
Division of Metabolism, Endocrinology, and Diabetes at the
University of Michigan. "The CAHtalyst
Adult Phase 3 study demonstrated that crinecerfont achieved a
significantly greater GC reduction from Week 4 through Week 24 than
placebo while androstenedione was maintained and did so safely.
Crinecerfont could be the treatment alternative that we've needed
to effectively treat our CAH patients while mitigating the
comorbidities found with the existing treatment paradigm."
Crinecerfont is an investigational, oral, selective
corticotropin-releasing factor type 1 receptor (CRF1)
antagonist being developed to reduce and control excess
adrenocorticotropic hormone (ACTH) and adrenal androgens through a
GC-independent mechanism for the treatment of CAH.
"The CAHtalyst Adult Phase 3 study demonstrated the ability of
crinecerfont to substantially reduce glucocorticoid doses to more
physiologic levels while maintaining or improving androgen
control," said Eiry W. Roberts, M.D., Chief Medical Officer at
Neurocrine Biosciences. "This represents a potential paradigm shift
in treatment for adult patients living with this disorder over many
years, who consistently struggle with the clinical consequences of
poor androgen control and/or excess glucocorticoid caused by
current inadequate treatment approaches."
The CAHtalyst Adult Phase 3 global registrational study was
conducted in 182 participants and designed to evaluate the safety,
efficacy, and tolerability of crinecerfont in adults ages 18 years
and older with CAH due to 21-hydroxylase deficiency. Over 95% of
participants completed the 24-week double-blind, placebo-controlled
treatment period of the study with minimal missing data.
The Phase 3 Adult study met the primary endpoint of percent
change from baseline in GC dose (while maintaining androstenedione
control) and the key secondary endpoint of achievement of reduction
to a physiologic GC dose (while maintaining androstenedione
control) at Week 24.
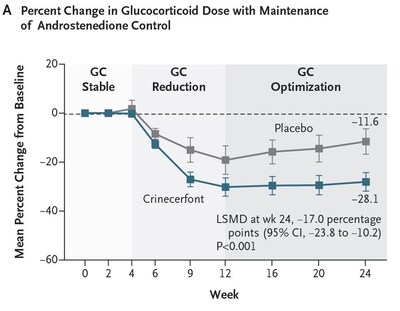
- Crinecerfont treatment led to a significantly greater GC dose
reduction at Week 24 while maintaining androstenedione control
compared to placebo (least squares mean [LSM] percent change from
baseline of -27.3% versus -10.3%, with LS mean difference [LSMD] of
-17.0%, P < 0.001). Actual value is P < 0.0001. (Figure A)
- This corresponded to changes of -4.8 and -2.1
mg/m2/day hydrocortisone equivalents for crinecerfont
and placebo, respectively
- After the initial 4-week GC stable period, mean percent GC
reduction (while androstenedione was controlled) was greater with
crinecerfont than placebo at all timepoints, and this effect was
maintained from Weeks 12 to 24, while GC levels increased from
Weeks 12 to 24 for participants on placebo.
Importantly, a significantly greater proportion of participants
treated with crinecerfont achieved reduction to a physiologic GC
dose (≤ 11 mg/m2/day) while maintaining androstenedione
control compared to placebo (62.7% vs. 17.5%; P < 0.001). Actual
value is P < 0.0001. (Figure B)
The Phase 3 Adult study also met the key secondary endpoint of
change from baseline in androstenedione (key adrenal androgen)
following the initial 4-week GC stable period:
- Crinecerfont treatment led to a significantly greater reduction
in androstenedione compared to an increase with placebo at Week 4
(LS mean change from baseline of -299 ng/dL versus +45.5
ng/dL, with LSMD of -345 ng/dL; P < 0.001). Actual value is
P < 0.0001.
- The significant reduction in androstenedione levels while
GC was stable provides clear evidence of the ability of
crinecerfont to reduce elevated adrenal androgen levels through a
non-GC mechanism.
At Week 24, mean androstenedione levels remained below baseline
with crinecerfont in the context of substantially reduced GC dose.
However, androstenedione levels increased to above baseline
with placebo, highlighting the inability to maintain androgen
control when GC was reduced in the absence of any other
intervention. (Figure C)
Despite the short-term duration of this Phase 3 study, favorable
trends were observed for endpoints associated with long-term
supraphysiologic GC therapy such as body weight, insulin
resistance, and glucose tolerance that were encouraging considering
the relatively short time frame of reduced GC dose. In addition,
bone resorption and formation markers increased more with
crinecerfont, reflecting relief from GC-induced suppression of bone
turnover.
Crinecerfont was generally well tolerated, with the most common
adverse events being fatigue and headache. Few serious adverse
events occurred, with none assessed as related to crinecerfont, and
there were few discontinuations due to adverse events. There
was no safety concern related to adrenal crisis, with few events
reported during the double-blind period and similar incidence of
adverse events leading to glucocorticoid stress dosing in the
crinecerfont and placebo groups.
Note: P-values in the text and figures reflect the Journal's
convention of calculating to two significant figures. Actual
P-values to three significant figures are also provided. Figures
reflect observed means rather than the LSM and LSMD described in
the text.
Figures are from The New England Journal of Medicine,
Phase 3 Trial of Crinecerfont in Adult Congenital Adrenal
Hyperplasia, Auchus R, Hamidi O, Pivonello R, et.al. This article
was published online on June 1, 2024.
Copyright © 2024 Massachusetts Medical Society. Reprinted with
permission from Massachusetts Medical Society.
About Congenital Adrenal Hyperplasia
Congenital
adrenal hyperplasia (CAH) is a rare genetic condition that
results in an enzyme deficiency that alters the production of
adrenal hormones which are essential for life. Approximately 95% of
CAH cases are caused by a mutation that leads to deficiency of the
enzyme 21-hydroxylase (21-OHD). Severe deficiency of this enzyme
leads to an inability of the adrenal glands to produce cortisol
and, in approximately 75% of cases, aldosterone. If left untreated,
CAH can result in salt wasting, dehydration, and even death.
Glucocorticoids (GCs) are currently used not only to correct the
endogenous cortisol deficiency, but doses used are higher than
cortisol replacement needed (supraphysiologic) to lower the levels
of adrenocorticotropic hormone (ACTH) and adrenal androgens.
However, glucocorticoid treatment at supraphysiologic doses has
been associated with serious and significant complications of
steroid excess, including metabolic issues such as weight gain and
diabetes, cardiovascular disease, and osteoporosis. Additionally,
long-term treatment with supraphysiologic GC doses may have
psychological and cognitive impact, such as changes in mood and
memory. Adrenal androgen excess has been associated with abnormal
bone growth and development in pediatric patients, female health
problems such as acne, excess hair growth and menstrual
irregularities, testicular rest tumors in males, and fertility
issues in both sexes.
To learn more about CAH, click here.
About Crinecerfont and the CAHtalyst™
Studies
Crinecerfont is an investigational, oral,
selective corticotropin-releasing factor type 1 receptor
(CRF1) antagonist being developed to reduce and control
excess adrenocorticotropic hormone (ACTH) and adrenal androgens
through a glucocorticoid-independent mechanism for the treatment of
congenital adrenal hyperplasia (CAH) due to 21-hydroxylase
deficiency. Antagonism of CRF1 receptors in the
pituitary has been shown to decrease ACTH levels, which in turn
decreases the production of adrenal androgens and potentially the
symptoms associated with CAH. Our data demonstrate that lowering
adrenal androgen levels enables lower, more physiologic dosing of
glucocorticoids and thus could potentially reduce the complications
associated with exposure to greater than normal glucocorticoid
doses in patients with CAH.
The CAHtalyst™ Pediatric and Adult Phase 3 global registrational
studies are designed to evaluate the safety, efficacy, and
tolerability of crinecerfont in children and adolescents, and
adults respectively, with congenital adrenal hyperplasia due to
21-hydroxylase deficiency. The primary portions of the CAHtalyst
Phase 3 studies have completed and enrollment is closed, while
the open-label treatment portions of both studies are ongoing.
Data from the CAHtalyst Pediatric and CAHtalyst Adult Phase 3
studies supported two New Drug Application submissions to the U.S.
Food and Drug Administration in April
2024.
To learn more about crinecerfont and the CAHtalyst studies,
click here.
About Neurocrine Biosciences, Inc.
Neurocrine Biosciences is a leading neuroscience-focused,
biopharmaceutical company with a simple purpose: to relieve
suffering for people with great needs, but few options. We are
dedicated to discovering and developing life-changing treatments
for patients with under-addressed neurological, neuroendocrine and
neuropsychiatric disorders. The company's diverse portfolio
includes FDA-approved treatments for tardive dyskinesia, chorea
associated with Huntington's disease, endometriosis* and uterine
fibroids*, as well as a robust pipeline including multiple
compounds in mid- to late-phase clinical development across our
core therapeutic areas. For three decades, we have applied our
unique insight into neuroscience and the interconnections between
brain and body systems to treat complex conditions. We relentlessly
pursue medicines to ease the burden of debilitating diseases and
disorders, because you deserve brave science. For more information,
visit neurocrine.com, and follow the company on LinkedIn, X
(formerly Twitter), and Facebook.
(*in collaboration with AbbVie)
The NEUROCRINE BIOSCIENCES Logo Lockup and YOU DESERVE BRAVE
SCIENCE are registered trademarks of Neurocrine Biosciences, Inc.
CAHtalyst is a trademark of Neurocrine Biosciences, Inc.
Forward-Looking Statements
In addition to historical
facts, this press release contains forward-looking statements that
involve a number of risks and uncertainties. These statements
include, but are not limited to, statements regarding the potential
benefits to be derived from crinecerfont. Among the factors that
could cause actual results to differ materially from those
indicated in the forward-looking statements include: the
crinecerfont NDAs may not be accepted for filing by the FDA or may
not obtain regulatory approval or such approval may be delayed;
additional regulatory submissions may not occur or be submitted in
a timely manner; the FDA may make adverse decisions regarding
crinecerfont; crinecerfont may not be found to be safe and/or
effective or may not prove to be beneficial to patients;
development activities for crinecerfont may not be completed on
time or at all; clinical development activities may be delayed for
regulatory or other reasons, may not be successful or replicate
previous and/or interim clinical trial results, or may not be
predictive of real-world results or of results in subsequent
clinical trials; competitive products and technological changes
that may limit demand for our products; uncertainties relating to
patent protection and intellectual property rights of third
parties; our dependence on third parties for development and
manufacturing activities related to crinecerfont, and our ability
to manage these third parties; our future financial and operating
performance; risks and uncertainties associated with the
commercialization of our products; and other risks described in the
Company's periodic reports filed with the Securities and Exchange
Commission, including without limitation the Company's quarterly
report on Form 10-Q for the quarter ended March 31, 2024. Neurocrine Biosciences disclaims
any obligation to update the statements contained in this press
release after the date hereof.

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/neurocrine-biosciences-announces-publication-of-primary-cahtalyst-adult-phase-3-study-results-of-crinecerfont-for-the-treatment-of-cah-in-the-new-england-journal-of-medicine-302161424.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/neurocrine-biosciences-announces-publication-of-primary-cahtalyst-adult-phase-3-study-results-of-crinecerfont-for-the-treatment-of-cah-in-the-new-england-journal-of-medicine-302161424.html
SOURCE Neurocrine Biosciences, Inc.