MedinCell’s Partner Teva Provides Guidance for UZEDY in 2024 and an Update on the Treatment-Candidate of Olanzapine Long-Acting Injectable (LAI)
31 Januar 2024 - 9:59PM
Business Wire
- Guidance for 2024 UZEDY Teva’s revenue: ~ $80
million
- Olanzapine LAI ongoing Phase 3: 675 patients (recruitment
completed), 62% of the targeted 3,600 injections performed, no PDSS
(Post injection Delirium/Sedation Syndrome) observed
MedinCell (Paris:MEDCL):
Access here the complete press release
About UZEDY
During the Q4 2023 earnings call held today by Teva
Pharmaceutical Industries Ltd., President and CEO Richard Francis
stated that he expects a strong uptake and significant growth for
UZEDY in 2024. He notably provided the annual revenue guidance for
UZEDY, projecting approximately $80 million for 2024.
This revenue projection is aligned with MedinCell's forecasted
earnings from UZEDY, as the company receives royalties on sales and
may earn up to $105 million in commercial milestones.
UZEDY is the first product based on MedinCell's long-acting
injection technology, BEPO, to reach commercial stage
- US marketing authorization obtained from the U.S. FDA on April
28, 2023
- Commercial launch by Teva in May 2023
- MedinCell has already received first royalties of €0.6 million,
calculated on Teva's net sales from mid-May to end of September
2023
About Olanzapine LAI (mdc-TJK)
Eric Hughes, Executive Vice President, Global R&D &
Chief Medical Officer, announced during the call that 62% of the
targeted 3,600 injections have already been performed as part of
the ongoing Phase 3 clinical trial and that no PDSS has been
observed. Full clinical package on efficiency and safety is
expected in the second half of 2024.
mdc-TJK is an investigational once-monthly subcutaneous
long-acting injection of the atypical antipsychotic olanzapine for
the treatment of schizophrenia. It has the potential to be the
first long-acting Olanzapine with a favorable safety profile as
other LAIs of Olanzapine have a FDA black box warning for PDSS that
limits their use.
Teva is fully responsible to lead the development and
commercialization of olanzapine LAI globally.
MedinCell may receive up to $117 million in development and
commercial milestones over the coming years for mdc-TJK, and is
eligible for royalties on all net sales.
Christophe Douat, CEO of MedinCell, says: “The guidance
on UZEDY is very positive. Teva’s ambition reaffirms its confidence
in its potential. Olanzapine LAI Phase 3, potential First-in-Class
product, is progressing impressively and is ahead of schedule with
a major milestone this year. Both illustrate the ability of
MedinCell technology to do breakthrough products. Based on these
news, we confirm our objective to achieve operational profitability
as soon as possible and to generate additional revenue with new
partnerships to extend our cash visibility until this horizon.”
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240131431252/en/
David Heuzé - Head of Communications
david.heuze@medincell.com / +33 (0)6 83 25 21 86
Media Relations Nicolas Mérigeau
medincell@newcap.eu / +33 (0)1 44 71 94 94
Investor Relations France Louis-Victor
Delouvrier/Alban Dufumier medincell@newcap.eu / +33 (0)1 44 71
94 94
Head of US Financial Strategy & IR Grace Kim
grace.kim@medincell.com / +1 (646) 991-4023
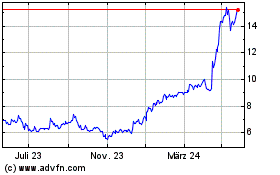
Medincell (EU:MEDCL)
Historical Stock Chart
Von Apr 2024 bis Mai 2024
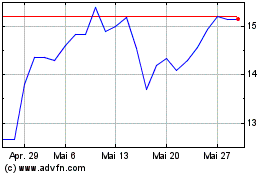
Medincell (EU:MEDCL)
Historical Stock Chart
Von Mai 2023 bis Mai 2024