Press release Biocartis Group NV: Biocartis Reports Results of First Quarter of 2023
20 April 2023 - 7:00AM

Press release Biocartis Group NV: Biocartis Reports Results of
First Quarter of 2023
PRESS RELEASE: REGULATED INFORMATION20 April 2023, 07:00
CEST
Biocartis Reports
Results of First Quarter of
2023:16%
Growth of Oncology Cartridge
RevenuesGross
Margin on
Products of
37%
and EBITDA of EUR
-8.4m
Mechelen, Belgium, 20
April 2023 – Biocartis Group NV (the
"Company" or "Biocartis"), an innovative molecular diagnostics
company (Euronext Brussels: BCART), today provides a business
update for the first quarter of 2023 and the outlook for the full
year 2023.
Commenting on the
Q1 2023
results, Herman Verrelst, Chief Executive
Officer of Biocartis, said: “The start of
2023 was broadly in line with our expectations, with 16% growth of
cartridge revenues in our core oncology business and a 35% gross
margin. While we still expect to grow such product related revenues
by 25-30% for the entire year, we saw Q1 2023 revenues were
affected by the impact of the price increases we implemented end of
2022 that led customers to place anticipative orders, particularly
so in the US. Furthermore, the macroeconomic environment continued
to slow down investments in the development and the
commercialization of certain partner products, affecting both
cartridge as well as instrument sales. Despite these effects, which
we believe to be temporary and short-term, we again took critical
steps in securing future growth that will be driven by continued
menu - and geographical expansion. Our Idylla™ MSI Test received
510(k) clearance1 by the US FDA, marking the start of the
commercialization of our in-vitro diagnostic solution for clinical
use and paving the way to unlock significant growth potential in
the US. Launching the Idylla™ IDH1-2 Mutation Assay Kit (RUO2)
among selected customers was a breakthrough for our new Idylla™
FLEX technology that we expect will allow us to bring new assays to
the market faster and to further tap into the vast market of liquid
biopsy-based monitoring. We also welcomed APIS Assay Technologies
as a new collaboration partner and look forward to adding their
novel Breast Cancer Subtyping test to our broad oncology menu on
Idylla™. Finally, we implemented management changes to enhance our
focus on the US market. Bryan Dechairo was appointed to the board
of directors and in my new role of executive chairman of the board
of directors, I look forward to partnering with Roger Moody,
Biocartis’ new CEO who brings a wealth of experience in the US
diagnostic industry to our executive leadership team. We are more
confident than ever about our ability to sustain future growth to
profitability and, save for further impact of the current economic
climate on ongoing collaborations, we reiterate our expectations
for the year."
Q1
2023
HIGHLIGHTS
- Product related
revenue of EUR 10.8m, up 2% year-on-year and including EUR 8.5m
cartridge revenue from 75k cartridges sold and EUR 2.3m from
instrument sales, rentals and servicing:
- Oncology
cartridge revenue of EUR 8m (+16% year-on-year)
- Revenue
contribution from Idylla™ SARS-CoV-2 product sales down by 53%,
from EUR 1.1m in Q1 2022 to EUR 0.5m in Q1 2023
- Cartridge
Average Sales Price (ASP) of EUR 120 in oncology and EUR 113
overall vs EUR 114 and EUR 101 in Q1 2022, respectively
- EUR 2.2m revenue
from instruments. 57 net new instruments placed year-to-date, total
installed base of 2,142 instruments end of Q1 2023
- Gross profit on
product sales3 of EUR 3.8m (Q1 2022: EUR 3.5m), reflecting a gross
margin of 37% (34% for the full year 2022). Q1 2023 was the last
quarter that included cartridge production on the old manufacturing
line ML1, which is now no longer in use for commercial cartridge
production. Continued scaling of the more automated high-throughput
manufacturing line ML2 is expected to further reduce cartridge
production cost and contribute to a gross margin on products of
40-45% for the full year 2023
- EBITDA4 of EUR
-8.4m, an improvement of EUR 1.1m or 12% year-on-year. The cash
position end Q1 2023 amounts to EUR 43.9m
- Idylla™ test
menu & partnerships:
- Announcement 9
February 2023: Launch among selected customers of the Idylla™
IDH1-2 Mutation Assay Kit (RUO), the first test developed with the
new Idylla™ FLEX technology that separates the generic components
of an Idylla™ test from the test-specific components
- Announcement 2
March 2023: 510(k) clearance by the U.S. Food and Drug
Administration (FDA) for the Idylla™ MSI Test
- Post the
reporting period, announcement 4 April 2023: New partnership
agreement with APIS Assay Technologies Ltd. for development of
APIS’ Breast Cancer Subtyping assay on the Idylla™ platform. This
assay, already available for in vitro diagnostic use5 in
centralized expert laboratories in the UK, will be commercialized6
by Biocartis ahead of the Idylla™ version of the assay
- Organizational
news:
- Recapitalization
– Announcement on 16 January 2023 on the completion of the final
steps of the comprehensive recapitalization transactions
- Strengthening US
market positioning - During Q1 2023 and shortly after, the Company
undertook several actions to strengthen its orientation towards the
US market:
- Announcement on
22 February 2023: Resignation of Mr. Roald Borré as Director and
appointment of Mr. Bryan Dechairo as a new independent board member
and member of the Audit Committee of the Company
- Announcement
post the reporting period, on 11 April 2023: Appointment of Roger
Moody to the position of Chief Executive Officer effective 24 April
20237. Herman Verrelst, who has been the Company’s Chief Executive
Officer since August 2017, will move into the new position of
Executive Chairman of the board of directors. Christian Reinaudo,
who has served as Chairman of the board since May 2018, will assume
the role of Lead Independent Director to act as principal liaison
between the non-executive members of the board and the executive
leadership team
2023 OUTLOOKBiocartis
reconfirms its 2023 guidance:
- Product related revenues8 of
between EUR 55m and EUR 60m, reflecting growth of 25%-35% when
excluding sales of SARS-CoV-2 tests that are expected to further
decrease
- A gross margin on product sales9 of
between 40% and 45%
- EBITDA of between EUR -25m and EUR
-28m, an improvement of between EUR 8.5m to EUR 11.5mThese
projections are based on foreign currency exchange rates applicable
on 23 February 2023, the date on which the 2022 results and 2023
outlook were published.
IDYLLA™ TEST MENU OUTLOOKAfter
having obtained US FDA 510(k) clearance for the Idylla™ MSI Test in
Q1 2023, Biocartis expects to achieve the following regulatory
milestones and to launch the assays listed below. The timing of the
planned launch of partner tests remains subject to changes imposed
by the relevant partners:
- SeptiCyte® RAPID on Idylla™ EDTA –
submission of 510(k) to the US FDA by Immunexpress
- Idylla™ IDH1-2 Mutation Assay Kit
(RUO) – Global availability to all customers
- Idylla™ PIK3CA-AKT1 Mutation Assay
– RUO product development in collaboration with LifeArc
- Idylla™ Merlin CP-GEP Assay – RUO
launch in collaboration with SkylineDx
- Idylla™ ThyroidPrint Assay – RUO
launch in collaboration with GeneproDx
FINANCIAL CALENDAR
- 12 May 2023
Annual Shareholders’ Meeting Biocartis Group
NV
- 31 August
2023 H1 2023
results
- 9 November 2023
Q3 2023 Business Update
--- END ---
More information: Renate Degrave |
Head of Corporate Communications & Investor Relations
Biocartise-mail rdegrave@biocartis.com tel
+32 15 631
729mobile +32 471 53 60 64
About Biocartis
With its revolutionary and proprietary Idylla™
platform, Biocartis (Euronext Brussels: BCART) aspires to enable
personalized medicine for patients around the world through
universal access to molecular testing, by making molecular testing
actionable, convenient, fast and suitable for any lab. The Idylla™
platform is a fully automated sample-to-result, real-time PCR
(Polymerase Chain Reaction) based system designed to offer in-house
access to accurate molecular information in a minimum amount of
time for faster, informed treatment decisions. Biocartis’
continuously expanding menu of molecular diagnostic tests addresses
key unmet clinical needs, with a focus in oncology. This is the
fastest growing segment of the molecular diagnostics market
worldwide. Today, Biocartis offers tests supporting melanoma,
colorectal, lung, liver and breast cancer, as well as for COVID-19,
Flu, RSV and sepsis. For more information,
visit www.biocartis.com or follow Biocartis on Twitter
@Biocartis_ , Facebook or LinkedIn.
Biocartis and Idylla™ are registered trademarks
in Europe, the United States and other countries. The Biocartis and
Idylla™ trademark and logo are used trademarks owned by Biocartis.
Please refer to the product labeling for applicable intended uses
for each individual Biocartis product. This press release is not
for distribution, directly or indirectly, in any jurisdiction where
to do so would be unlawful. Any persons reading this press release
should inform themselves of and observe any such restrictions.
Biocartis takes no responsibility for any violation of any such
restrictions by any person. This press release does not constitute
an offer or invitation for the sale or purchase of securities in
any jurisdiction. No securities of Biocartis may be offered or sold
in the United States of America absent registration with the United
States Securities and Exchange Commission or an exemption from
registration under the U.S. Securities Act of 1933, as amended.
Forward-looking
statementsCertain statements, beliefs and opinions in this
press release are forward-looking, which reflect the Company's or,
as appropriate, the Company directors' or managements' current
expectations and projections concerning future events such as the
Company's results of operations, financial condition, liquidity,
performance, prospects, growth, strategies and the industry in
which the Company operates. By their nature, forward-looking
statements involve a number of risks, uncertainties, assumptions
and other factors that could cause actual results or events to
differ materially from those expressed or implied by the
forward-looking statements. These risks, uncertainties, assumptions
and factors could adversely affect the outcome and financial
effects of the plans and events described herein. A multitude of
factors including, but not limited to, changes in demand,
competition and technology, can cause actual events, performance or
results to differ significantly from any anticipated development.
Forward-looking statements contained in this press release
regarding past trends or activities are not guarantees of future
performance and should not be taken as a representation that such
trends or activities will continue in the future. In addition, even
if actual results or developments are consistent with the
forward-looking statements contained in this press release, those
results or developments may not be indicative of results or
developments in future periods. No representations and warranties
are made as to the accuracy or fairness of such forward-looking
statements. As a result, the Company expressly disclaims any
obligation or undertaking to release any updates or revisions to
any forward-looking statements in this press release as a result of
any change in expectations or any change in events, conditions,
assumptions or circumstances on which these forward-looking
statements are based, except if specifically required to do so by
law or regulation. Neither the Company nor its advisers or
representatives nor any of its subsidiary undertakings or any such
person's officers or employees guarantees that the assumptions
underlying such forward-looking statements are free from errors nor
does either accept any responsibility for the future accuracy of
the forward-looking statements contained in this press release or
the actual occurrence of the forecasted developments. You should
not place undue reliance on forward-looking statements, which speak
only as of the date of this press release.
1 A 510(k) is a premarketing submission made to FDA to
demonstrate that the device to be marketed is as safe and
effective, that is, substantially equivalent (SE), to a legally
marketed device that is not subject to premarket approval (PMA).
510(k) (premarket notification) to FDA is required at least 90 days
before marketing unless the device is exempt from 510(k)
requirements. Source:
https://www.fda.gov/medical-devices/products-and-medical-procedures/device-approvals-denials-and-clearances,
last consulted on 17 April 20232 RUO = Research Use Only, not for
use in diagnostic procedures3 Excluding instrument servicing4
Earnings before interest, taxes, depreciation and amortization5
Registered as IVD in the UK, submission for IVDR CE marking
pending6 In the European Union and selected export markets 7 Mr
Moody will also become a member of the Board of Directors, subject
to the approval by the Company’s general shareholders’ meeting 8
Including revenue from instrument servicing9 Excluding revenue from
instrument servicing
Biocartis Group NV (EU:BCART)
Historical Stock Chart
Von Jun 2024 bis Jul 2024
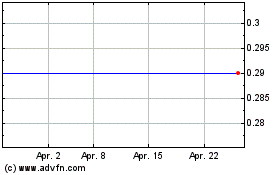
Biocartis Group NV (EU:BCART)
Historical Stock Chart
Von Jul 2023 bis Jul 2024