LifeLabs, Bayer Inc. to Offer Complimentary Test to Detect TRK Fusion Cancer
02 September 2020 - 7:37PM
Dow Jones News
By Stephen Nakrosis
Canada's LifeLabs on Wednesday said it would work with Bayer
Inc. to launch FastTRK, a "complimentary clinical testing program
for the diagnosis of neurotrophic tyrosine receptor kinase gene
fusions in TRK fusion cancer patients in Canada."
Bayer, which founded the program with LifeLabs and the Kingston
Health Sciences Centre, will cover the full cost of the test until
at least the end of 2021, LifeLabs said.
Charles Brown, president and chief executive of LifeLabs, said
"Because this test is for such a rare form of cancer, it represents
an exciting innovation that may lead to other treatment options for
patients, where previous options have failed."
Patients with solid tumors that are metastatic, or when surgical
resection is likely to result in severe morbidity, and for whom no
satisfactory treatment options are available, meet the criteria to
be tested, LifeLabs said, adding a clinician must request the
test.
LifeLabs said it provides laboratory diagnostic information and
digital health connectivity systems.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
September 02, 2020 13:22 ET (17:22 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
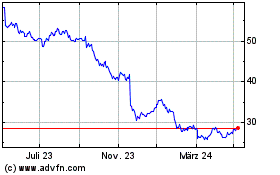
Bayer (TG:BAYN)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
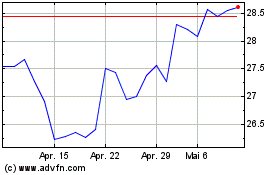
Bayer (TG:BAYN)
Historical Stock Chart
Von Apr 2023 bis Apr 2024