AbbVie Glioblastoma Drug Misses Primary Endpoint
17 Mai 2019 - 3:31PM
Dow Jones News
By Chris Wack
AbbVie (ABBV) said Friday that its phase 3 study of its
glioblastoma drug depatuxizumab mafodotin didn't meet its primary
endpoint.
The biopharmaceutical company said its Intellance-1 study of
depatuxizumab mafodotin in patients with newly diagnosed
glioblastoma failed to achieve its overall survival objective, and
demonstrated no survival benefit for patients receiving Depatux-M
at an interim analysis.
Abbvie said an independent data monitoring committee recommended
the study be stopped due to lack of survival benefit for patients
receiving Depatux-M compared with the placebo when added to the
standard regimen of radiation and temozolomide.
AbbVie said no new safety findings were observed, and enrollment
in all ongoing Depatux-M studies has been halted.
The Intellance-1 trial was conducted in collaboration with the
RTOG Foundation, an independent, non-profit cancer research
organization.
Glioblastoma, also known as glioblastoma multiforme, is an
aggressive type of cancer that can occur in the brain or spinal
cord.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
May 17, 2019 09:16 ET (13:16 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
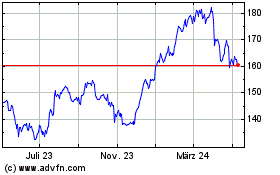
AbbVie (NYSE:ABBV)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
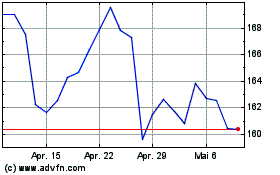
AbbVie (NYSE:ABBV)
Historical Stock Chart
Von Apr 2023 bis Apr 2024