Merck Gets Positive EU Committee Opinion for Cancer Drug
17 Dezember 2021 - 1:43PM
Dow Jones News
By Matt Grossman
Merck & Co. said its Keytruda cancer drug has received a
positive opinion from Europe's Committee for Medicinal Products for
Human Use for its deployment for some forms of kidney cancer.
The opinion concerns the drug's use as a monotherapy for
treatment of adults with renal cell carcinoma at increased risk of
recurrence following nephrectomy. In a Phase 3 trial, the drug
improved disease-free survival in patients, reducing the risk of
recurrence or death by 32% compared with a placebo in patients with
a higher risk of recurrence, the company said.
The European Commission will review the drug's potential
marketing authorization, and is expected to make a final decision
in the first quarter of 2022, Merck said.
Write to Matt Grossman at matt.grossman@wsj.com
(END) Dow Jones Newswires
December 17, 2021 07:28 ET (12:28 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
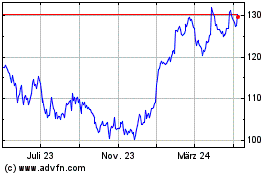
Merck (NYSE:MRK)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
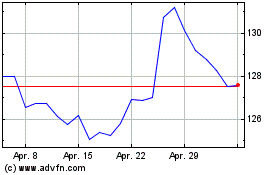
Merck (NYSE:MRK)
Historical Stock Chart
Von Apr 2023 bis Apr 2024