European Medicines Agency Approves Additional Manufacturing Sites for Pfizer-BioNTech Covid Vaccine
09 September 2021 - 6:56PM
Dow Jones News
By Giulia Petroni
The European Medicines Agency said Thursday that it has approved
two additional manufacturing sites for the production of the
Covid-19 vaccine developed by Pfizer Inc. and BioNTech SE.
The sites, which will manufacture the finished product, are set
to provide up to 50 million additional doses of Comirnaty by 2021,
according to the EU agency.
One site is operated by Sanofi-Aventis Deutschland GmbH, while
the other by Siegfried Hameln GmbH. They are both located in
Germany.
The European Medicines Agency said the sites can become
operational immediately, as the recommendation doesn't require a
decision from the European Commission.
Write to Giulia Petroni at giulia.petroni@wsj.com
(END) Dow Jones Newswires
September 09, 2021 12:41 ET (16:41 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
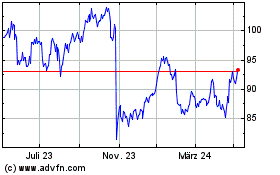
Sanofi (EU:SAN)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
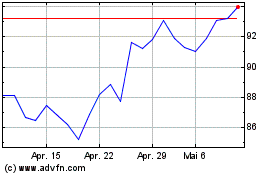
Sanofi (EU:SAN)
Historical Stock Chart
Von Apr 2023 bis Apr 2024