Appili Therapeutics Appoints Dr. Carl Gelhaus and Arthur Baran to support the ATI-1701 Biodefense Vaccine Program
17 Januar 2023 - 1:12PM
Business Wire
Appili Therapeutics has named Dr. Carl Gelhaus as Director of
Non-Clinical Research and Arthur Baran as Director of New Product
Development for the ATI-1701 program.
Appili Therapeutics Inc. (TSX: APLI; OTCQB: APLIF) (the
“Company” or “Appili”), a biopharmaceutical company focused on drug
development for infectious diseases and biodefense, has named Carl
Gelhaus, Ph.D., as Director of Non-Clinical Research, and Arthur
Baran, B.Sc., MBA, PMP, as Director of New Product Development
including ATI-1701, a potential first-in-class vaccine candidate
for the prevention of infection with aerosolized Francisella
tularensis, which is classified by the U.S. National Institutes of
Health as a Category A pathogen and top-priority biothreat.
Dr. Gelhaus brings over 20 years of infectious disease and
animal model research experience to the role, with an emphasis on
tularemia research. Carl has a long history as a research partner
with Appili on ATI-1701, including having designed and conducted
key animal model efficacy studies for ATI-1701. Carl also presented
the positive interim data in January 2020 at the 2020 ASM
Biothreats Conference . At Appili, Carl will recruit and lead the
research and development team of Ph.D. and non-Ph.D. scientists,
design and manage pre-clinical studies, and manage contract
research organizations to support pre-clinical development
programs.
Arthur Baran brings to Appili more than 22 years of experience
in global product manufacturing operations, drug product
development team leadership, program management, business
development, manufacturing site leadership and quality assurance.
Art will focus his work at Appili on building systems, processes,
and tools to facilitate business planning and decision-making, as
well as streamlining product leadership and management capabilities
within Appili.
“We are fortunate to have both Carl and Arthur join our team at
a pivotal time in Appili’s development and growth. With our recent
funding from DTRA for the ATI-1701 program, both Carl’s expertise
with Francisella tularensis and Art’s skills in program management
and internal organization will be valuable to us as we move
ATI-1701 forward,” said Don Cilla, Pharm.D., M.B.A., President and
Chief Executive Officer of Appili Therapeutics.
About Appili Therapeutics
Appili Therapeutics is an infectious disease and biodefense
biopharmaceutical company that is purposefully built,
portfolio-driven, and people-focused to fulfill its mission of
solving life-threatening infections. By systematically identifying
urgent infections with unmet needs, Appili’s goal is to
strategically develop a pipeline of novel therapies to prevent
deaths and improve lives. The Company is currently advancing a
diverse range of anti-infectives, including a vaccine candidate to
eliminate a serious biological weapon threat, a topical
antiparasitic for the treatment of a disfiguring disease, and a
novel easy to use, liquid oral formulation targeting parasitic and
anaerobic infections. Led by a proven management team, Appili is at
the epicenter of the global fight against infection. For more
information, visit www.AppiliTherapeutics.com.
Forward-Looking Statements
This news release contains “forward-looking statements”,
including with respect to the Company’s ongoing development plans
and timeline with respect to ATI-1701. Wherever possible, words
such as “may,” “would,” “could,” “should,” “will,” “anticipate,”
“believe,” “plan,” “expect,” “intend,” “estimate,” “potential for,”
and similar expressions have been used to identify these
forward-looking statements.
Forward looking statements involve significant known and unknown
risks, uncertainties and assumptions, including, without
limitation, the risk factors listed in the annual information form
of the Company dated June 23, 2022 and the other filings made by
the Company with the Canadian securities regulatory authorities
(which may be viewed at www.sedar.com). Should one or more of these
risks or uncertainties materialize or should assumptions underlying
the forward-looking statements prove incorrect, actual results,
performance or achievements may vary materially from those
expressed or implied by the forward-looking statements contained in
this news release. These factors should be considered carefully,
and prospective investors should not place undue reliance on the
forward-looking statements. The Company disclaims any intention or
obligation to revise forward-looking statements whether as a result
of new information, future developments or otherwise, except as
required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230117005675/en/
Media Contact: Celia Dewyer/APCO Worldwide T: 1-646-717-9915 E:
CDewyer@apcoworldwide.com
Investor Relations Contact: Jenna McNeil; Corporate Affairs and
Communications Manager Appili Therapeutics E:
Info@AppiliTherapeutics.com
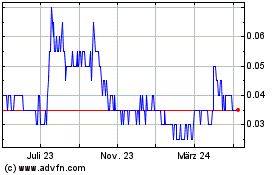
Appili Therapeutics (TSX:APLI)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
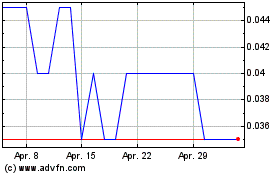
Appili Therapeutics (TSX:APLI)
Historical Stock Chart
Von Apr 2023 bis Apr 2024