Myovant, Pfizer: FDA Extends Review of Myfembree in Endometriosis
06 Mai 2022 - 1:41PM
Dow Jones News
By Colin Kellaher
Myovant Sciences Ltd. and Pfizer Inc. on Friday said the U.S.
Food and Drug Administration has extended its review of their
application for expanded approval of Myfembree after the companies
submitted more information regarding bone mineral density at the
agency's request.
Myovant and Pfizer, which are seeking approval for the
management of moderate to severe pain associated with
endometriosis, said the FDA needs more time to review the new
information and extended its target action date to Aug. 6 from May
6.
The agency last year approved Myfembree for the management of
heavy menstrual bleeding associated with uterine fibroids in
premenopausal women. Myovant and Pfizer are jointly developing and
commercializing Myfembree.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
May 06, 2022 07:26 ET (11:26 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
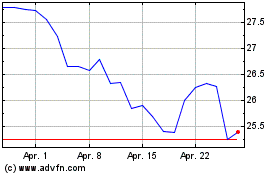
Pfizer (NYSE:PFE)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
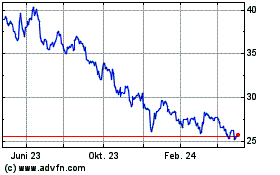
Pfizer (NYSE:PFE)
Historical Stock Chart
Von Apr 2023 bis Apr 2024