Penumbra Announces European Launch of RED® Reperfusion Catheters for Stroke Care
07 September 2022 - 3:00PM
Business Wire
- Physicians in Europe now have access to Penumbra’s latest
stroke technology for mechanical thrombectomy
- RED® reperfusion catheters are designed with optimized
trackability and aspiration efficiency to help navigate the complex
anatomy of the brain and deliver powerful aspiration for the
removal of blood clots
- Initial INSIGHT Registry data presented at WFITN demonstrated
RED catheters were highly effective in removing all clot types with
an impressive 68.9% mTICI > 2b-3 overall first pass effect
rate
Penumbra, Inc. (NYSE: PEN), a global healthcare company focused
on innovative therapies, announced that its RED® Reperfusion
Catheters have secured CE Mark (Conformité Européenne) and are now
commercially available in Europe. The catheters are part of the
company’s Penumbra System®, which is a fully integrated mechanical
aspiration thrombectomy system designed to restore blood flow in
acute ischemic stroke (AIS) patients.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220907005565/en/
Penumbra’s RED® Reperfusion Catheters are
now commercially available in Europe. The catheters are part of the
company’s Penumbra System®, which is a fully integrated mechanical
aspiration thrombectomy system designed to restore blood flow in
acute ischemic stroke (AIS) patients. (Graphic: Penumbra)
“When addressing stroke, our goal is to remove the blood clot as
quickly as possible for patients to have the best outcome,” said
Saleh Lamin, M.D., neurointerventionalist, Queen Elizabeth
Hospital, Birmingham, UK. “With RED catheters, we will be able to
get up to the clot quickly and efficiently remove it, most of the
time within a single pass. Restoring blood flow promptly is key as
it is associated with less damage to the brain and better
outcomes.”
The RED family of catheters is engineered with the latest
technology in trackability and aspiration to address a wide range
of large vessel occlusions via mechanical aspiration thrombectomy.
The RED catheters feature REDglide™ technology, which enhances the
trackability of the catheters as they navigate the challenging
vessel anatomy of the brain while also maximizing aspiration
efficiency to remove blood clots. Additionally, the RED catheters
have a full-length PTFE liner designed to maintain their true lumen
size under powerful vacuum.
“Penumbra’s commitment to advancing technology allows us to have
one of the most expansive aspiration stroke portfolios on the
market, enabling physicians to have the latest innovations with the
most options to provide the best care to their patients,” said Joan
Kristensen, vice president and head of the Europe, Middle East and
Africa region for Penumbra, Inc. “With enhanced trackability and
powerful aspiration, initial data has shown that Penumbra’s RED
catheters can restore blood flow quickly across all clot types,
which would potentially have a significant impact on patient
outcomes here in Europe.”
Initial INSIGHT Registry data presented in August at the World
Federation of Interventional and Therapeutic Neuroradiology (WFITN)
showed Penumbra’s RED® reperfusion catheters were successful in
removing all clot types with a significant first pass rate. The
data from 161 AIS patients showed 68.9% successful first-pass
revascularization (mTICI≥2b-3), and 97% successful
revascularization after final angiogram through site reported
findings. Final mTICI≥2b-3 scores were reported in 98.1% of
patients with firm-red clots and in 100% of patients with other
clot types.
“Expanding access to our leading RED catheters in Europe
enhances Penumbra’s ability to support more physicians and patients
around the world,” said Sandra Lesenfants, president of
interventional at Penumbra, Inc. “As we continue to pioneer
interventional technology, our commitment is to improve care so
patients, especially those with serious conditions such as stroke,
can return home quickly and live full lives.”
About Penumbra
Penumbra, Inc., headquartered in Alameda, California, is a
global healthcare company focused on innovative therapies. Penumbra
designs, develops, manufactures and markets novel products and has
a broad portfolio that addresses challenging medical conditions in
markets with significant unmet need. Penumbra supports healthcare
providers, hospitals and clinics in more than 100 countries. For
more information, visit www.penumbrainc.com and connect on Twitter
and LinkedIn.
Important Safety Information
Additional information about Penumbra’s products can be located
on Penumbra’s website at
http://www.penumbrainc.com/healthcare-professionals. Prior to use,
please refer to Instructions for Use for complete product
indications, contraindications, warnings, precautions, potential
adverse events and detailed instructions for use. Risk information
can be found here.
Forward-Looking Statements
Except for historical information, certain statements in this
press release are forward-looking in nature and are subject to
risks, uncertainties and assumptions about us. Our business and
operations are subject to a variety of risks and uncertainties and,
consequently, actual results may differ materially from those
projected by any forward-looking statements. Factors that could
cause actual results to differ from those projected include, but
are not limited to: the impact of the COVID-19 pandemic on our
business, results of operations and financial condition; failure to
sustain or grow profitability or generate positive cash flows;
failure to effectively introduce and market new products; delays in
product introductions; significant competition; inability to
further penetrate our current customer base, expand our user base
and increase the frequency of use of our products by our customers;
inability to achieve or maintain satisfactory pricing and margins;
manufacturing difficulties; permanent write-downs or write-offs of
our inventory; product defects or failures; unfavorable outcomes in
clinical trials; inability to maintain our culture as we grow;
fluctuations in foreign currency exchange rates; potential adverse
regulatory actions; and the potential impact of any acquisitions,
mergers, dispositions, joint ventures or investments we may make.
These risks and uncertainties, as well as others, are discussed in
greater detail in our filings with the Securities and Exchange
Commission (SEC), including our Annual Report on Form 10-K for the
year ended December 31, 2021 filed with the SEC on February 22,
2022. There may be additional risks of which we are not presently
aware or that we currently believe are immaterial which could have
an adverse impact on our business. Any forward-looking statements
are based on our current expectations, estimates and assumptions
regarding future events and are applicable only as of the dates of
such statements. We make no commitment to revise or update any
forward-looking statements in order to reflect events or
circumstances that may change.
Source: Penumbra, Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220907005565/en/
Jennifer Heth Penumbra, Inc jheth@penumbrainc.com
510-995-9791
Joni Ramirez Merryman Communications
joni@merrymancommunications.com 323.532.0746
Penumbra (NYSE:PEN)
Historical Stock Chart
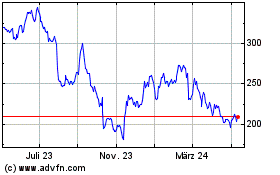
Von Mär 2024 bis Apr 2024
Penumbra (NYSE:PEN)
Historical Stock Chart
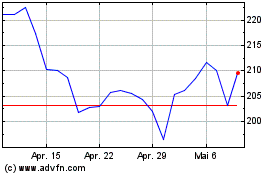
Von Apr 2023 bis Apr 2024