- Actively enrolling participants in our Phase 2/3 clinical trial
for the potential treatment of progressive NF2-mutated
meningiomas
- On track to initiate our Phase 2 clinical trial for the
potential treatment of FAP in the third quarter of 2022
- U.S. FDA granted Recursion Fast Track designation and the
European Commission granted Recursion Orphan Drug Designation for
REC-4881 for the potential treatment of FAP
- On track to initiate our Phase 1 clinical trial for the
potential treatment of Clostridium difficile colitis in the
second half of 2022
SALT
LAKE CITY, Aug. 9, 2022 /PRNewswire/ -- Recursion
(Nasdaq: RXRX), the clinical-stage biotechnology company
industrializing drug discovery by decoding biology, today reported
business updates and financial results for its second quarter
ending June 30, 2022.
"Recursion continues to make progress in advancing its clinical
programs, including initiating a Phase 2 trial for NF2-mutated
meningiomas and receiving U.S. FDA Fast Track and European
Commission Orphan Drug designations for REC-4881 for the potential
treatment of FAP," said Chris
Gibson, Ph.D., Co-Founder & CEO at Recursion. "In the
context of continued capital markets friction we are increasingly
focusing our pipeline around rapidly deliverable oncology programs.
We also continue to lead the digital transformation of drug
discovery by building additional capabilities into our Recursion OS
platform, including scaling our transcriptomics hit validation
platform to up to 13,000 near-whole exomes per week and advancing
our ChemOS systems by preparing to install our scalable and
automated drug metabolism and pharmacokinetics platform. Across our
diverse platform that spans target and hit discovery through
optimization and translation, we have now generated and control
over 16 petabytes of proprietary biological and chemical data and
2.4 trillion predicted biological and chemical relationships,
helping us turn the bespoke, artisanal, and serial process of drug
discovery into a search and validation problem."
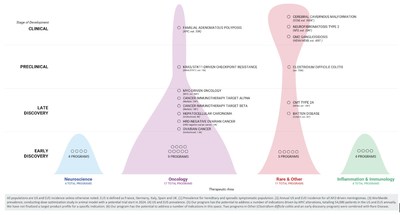
Summary of Business Highlights
- Internal Pipeline
-
- Cerebral cavernous malformation (CCM) (REC-994):
In March 2022, we announced the
initiation of our Phase 2 SYCAMORE clinical trial, which is a
double-blind, placebo-controlled safety, tolerability and
exploratory efficacy study of this drug candidate in 60
participants with CCM. At this time, we continue to
actively enroll participants.
- Neurofibromatosis type 2 (NF2) (REC-2282): In
June 2022 at the Children's Tumor
Foundation NF Conference, we announced the initiation
of our Phase 2/3 POPLAR clinical trial, which is a parallel group,
two stage, randomized, multicenter study of this drug
candidate in approximately 90 participants with progressive
NF2-mutated meningiomas. At this time, we
continue to actively enroll participants.
- Familial adenomatous polyposis (FAP) (REC-4881):
We are on track to initiate a Phase 2, randomized,
double-blind, placebo-controlled study to evaluate safety,
pharmacokinetics and exploratory efficacy of this drug
candidate in FAP in the third quarter of 2022.
Recently, the U.S. Food and Drug Administration (FDA) granted
Recursion Fast Track designation and the European Commission
granted Recursion Orphan Drug Designation for REC-4881 for the
potential treatment of FAP.
- Clostridium difficile colitis (REC-3964): We made
progress in IND-enabling studies for REC-3964 and are on track to
initiate a Phase 1 study in the second half of 2022.
- Oncology pipeline: We continue to focus our pipeline on
oncology and oncology-like programs while advancing numerous
programs discovered using our next generation mapping and
navigating technology, including programs focused on novel targets
and polypharmacology.
- Transformational Collaborations
-
- We continue to advance efforts to discover new potential
therapeutics with our strategic partners in the areas of fibrotic
disease (Bayer) as well as neuroscience and a
single indication in gastrointestinal oncology (Roche
and Genentech).
- Recursion OS
-
- ChemOS: We are preparing to install our automated
and scalable drug metabolism and pharmacokinetics
(DMPK) platform which will allow for the
processing and evaluation of compounds for protein plasma
binding, microsomal stability, and cell permeability.
Such continuous chemical data generation will help enable us to
build machine learning approaches that predict properties for our
and our partners' growing libraries of chemical compounds.
Furthermore, we are implementing a unified workflow for medicinal
and computational chemists to seamlessly access assay data, design
molecules, and perform predictive analyses to support our internal
and partnership programs.
- Machine Learning: We continue to improve the ease,
scale, and biological relevance of our machine learning models and
added new benchmarking flows to evaluate how well our models
recapitulate known biological relationships associated with protein
complexes and pathways. We improved our machine learning models to
achieve state-of-the-art results in 9 of 22 absorption,
distribution, metabolism, excretion, and toxicity
(ADMET) benchmark tasks from Therapeutic Data Commons
and we are leveraging them for our oncology programs.
- Transcriptomics: We continued building out our
scaled transcriptomics platform, with infrastructure,
automation, and operational processes that will enable the robust
validation of inferences from our maps of biology and chemistry. We
have now been able to carry out and analyze up to 13,000
near-whole exomes per week, creating another growing
relatable dataset that we can integrate in our
Recursion OS for continued improvement of our inferences across
compounds and biology at scale.
- Compounding Cycles of Discovery: The visualization
below frames how additional datasets and capabilities
compound cycles of discovery to potentially translate novel
insights into clinical candidates. In this technology stack
graphic, bold text signifies capabilities that
have been built to some meaningful scale already, italic
text signifies capabilities that are in the process of
being built, and standard text signifies capabilities that
Recursion intends to incorporate in the future.
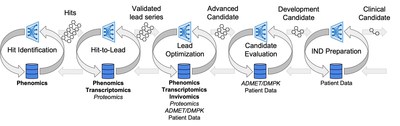
Second Quarter 2022 Financial Results
- Cash Position: Cash, cash equivalents, and investments
were $515.4 million as of
June 30, 2022.
- Revenue: Total revenue, consisting primarily of revenue
from collaborative agreements, was $7.7
million for the second quarter of 2022, compared to
$2.5 million for the second quarter
of 2021. The increase was due to revenue recognized from our
Roche-Genentech collaboration.
- Research and Development Expenses: Research and
development expenses were $38.4
million for the second quarter of 2022, compared to
$29.6 million for the second quarter
of 2021. The increase in research and development expenses was
primarily due to an increased number of pre-clinical assets being
validated and increased clinical costs as studies progressed.
- General and Administrative Expenses: General and
administrative expenses were $21.2
million for the second quarter of 2022, compared to
$13.9 million for the second quarter
of 2021. The increase in general and administrative expenses was
due to the growth in size of the company's operations, including an
increase in salaries and wages of $1.8
million, a fixed asset write-down of $2.8 million, increased rent expense of
$1.0 million, and other
administrative costs associated with operating a public
company.
- Net Loss: Net loss was $65.6
million for the second quarter of 2022, compared to a net
loss of $43.4 million for the second
quarter of 2021.
About Recursion
Recursion is the clinical-stage
biotechnology company industrializing drug discovery by decoding
biology. Enabling its mission is the Recursion OS, a platform built
across diverse technologies that continuously expands one of the
world's largest proprietary biological and chemical datasets.
Recursion leverages sophisticated machine-learning algorithms to
distill from its dataset a collection of trillions of searchable
relationships across biology and chemistry unconstrained by human
bias. By commanding massive experimental scale — up to millions of
wet lab experiments weekly — and massive computational scale —
owning and operating one of the most powerful supercomputers in the
world, Recursion is uniting technology, biology and chemistry to
advance the future of medicine.
Recursion is headquartered in Salt
Lake City, where it is a founding member of BioHive, the
Utah life sciences industry
collective. Recursion also has offices in Toronto, Montreal and the San
Francisco Bay Area. Learn more at www.Recursion.com, or
connect on Twitter and LinkedIn.
Media Contact
Media@Recursion.com
Investor Contact
Investor@Recursion.com
Consolidated Statements of Operations
|
Recursion
Pharmaceuticals, Inc.
|
|
Condensed
Consolidated Statements of Operations (unaudited)
|
|
(in thousands,
except share and per share amounts)
|
|
|
|
Three months
ended
|
|
Six months
ended
|
|
|
June
30,
|
|
June
30,
|
|
Revenue
|
2022
|
2021
|
|
2022
|
2021
|
|
Operating
revenue
|
$
7,653
|
$
2,500
|
|
$
12,952
|
$
5,000
|
|
Grant
revenue
|
21
|
49
|
|
55
|
111
|
|
Total
revenue
|
7,674
|
2,549
|
|
13,007
|
5,111
|
|
|
|
|
|
|
|
|
Operating costs and
expenses
|
|
|
|
|
|
|
Cost of
revenue
|
14,227
|
-
|
|
22,026
|
-
|
|
Research and
development
|
38,439
|
29,624
|
|
70,880
|
53,733
|
|
General and
administrative
|
21,199
|
13,854
|
|
42,273
|
22,791
|
|
Total operating
costs and expenses
|
73,865
|
43,478
|
|
135,179
|
76,524
|
|
|
|
|
|
|
|
|
Loss from
operations
|
(66,191)
|
(40,929)
|
|
(122,172)
|
(71,413)
|
|
Other income (loss),
net
|
631
|
(2,472)
|
|
633
|
(2,705)
|
|
Net
loss
|
$
(65,560)
|
$
(43,401)
|
|
$
(121,539)
|
$
(74,118)
|
|
|
|
|
|
|
|
|
Per share
data
|
|
|
|
|
|
|
Net loss per share
of Class A and B
common stock, basic and diluted
|
$
(0.38)
|
$
(0.31)
|
|
$
(0.71)
|
$
(0.91)
|
|
Weighted-average
shares (Class A and B)
outstanding, basic and diluted
|
172,212,390
|
138,360,646
|
|
171,455,595
|
81,022,240
|
Consolidated Balance Sheets
|
Recursion
Pharmaceuticals, Inc.
|
|
Condensed
Consolidated Balance Sheets (unaudited)
|
|
(in
thousands)
|
|
|
|
|
|
|
June
30,
|
December
31,
|
|
|
2022
|
2021
|
|
Assets
|
|
|
|
Current
assets
|
|
|
|
Cash and cash
equivalents
|
$
453,875
|
$
285,116
|
|
Restricted
cash
|
1,901
|
1,552
|
|
Accounts
receivable
|
21
|
34
|
|
Other
receivables
|
11,659
|
9,056
|
|
Investments
|
61,561
|
231,446
|
|
Other current
assets
|
16,979
|
7,514
|
|
Total current
assets
|
545,996
|
534,718
|
|
|
|
|
|
Restricted cash,
non-current
|
8,334
|
8,681
|
|
Property and equipment,
net
|
81,508
|
64,725
|
|
Operating lease
right-of-use assets
|
34,643
|
-
|
|
Intangible assets,
net
|
1,233
|
1,385
|
|
Goodwill
|
801
|
801
|
|
Other non-current
assets
|
-
|
35
|
|
Total
assets
|
$
672,515
|
$
610,345
|
|
|
|
|
|
Liabilities and
stockholders' equity
|
|
|
|
Current
liabilities
|
|
|
|
Accounts
payable
|
$
3,176
|
$
2,819
|
|
Accrued expenses and
other liabilities
|
24,361
|
32,333
|
|
Unearned
revenue
|
63,781
|
10,000
|
|
Notes
payable
|
93
|
90
|
|
Operating lease
liabilities
|
5,242
|
-
|
|
Lease incentive
obligation
|
-
|
1,416
|
|
Total current
liabilities
|
96,653
|
46,658
|
|
|
|
|
|
Deferred
rent
|
-
|
4,110
|
|
Unearned revenue,
non-current
|
89,934
|
6,667
|
|
Notes payable,
non-current
|
586
|
633
|
|
Operating lease
liabilities, non-current
|
47,763
|
-
|
|
Lease incentive
obligation, non-current
|
-
|
9,339
|
|
Total
liabilities
|
234,936
|
67,407
|
|
|
|
|
|
Commitments and
contingencies
|
|
|
|
|
|
|
|
Stockholders'
equity
|
|
|
|
Common stock (Class A
and B)
|
2
|
2
|
|
Additional paid-in
capital
|
959,393
|
943,142
|
|
Accumulated
deficit
|
(521,619)
|
(400,080)
|
|
Accumulated other
comprehensive loss
|
(197)
|
(126)
|
|
Total stockholder's
equity
|
437,579
|
542,938
|
|
|
|
|
|
Total liabilities
and stockholders' equity
|
$
672,515
|
$
610,345
|
Forward-Looking Statements
This document contains
information that includes or is based upon "forward-looking
statements" within the meaning of the Securities Litigation Reform
Act of 1995, including, without limitation, those regarding early
and late stage discovery, preclinical, and clinical programs;
licenses and collaborations; prospective products and their
potential future indications and market opportunities; Recursion OS
and other technologies; business and financial plans and
performance; and all other statements that are not historical
facts. Forward-looking statements may or may not include
identifying words such as "plan," "will," "expect," "anticipate,"
"intend," "believe," "potential," "continue," and similar terms.
These statements are subject to known or unknown risks and
uncertainties that could cause actual results to differ materially
from those expressed or implied in such statements, including but
not limited to: challenges inherent in pharmaceutical research and
development, including the timing and results of preclinical and
clinical programs, where the risk of failure is high and failure
can occur at any stage prior to or after regulatory approval due to
lack of sufficient efficacy, safety considerations, or other
factors; our ability to leverage and enhance our drug discovery
platform; our ability to obtain financing for development
activities and other corporate purposes; the success of our
collaboration activities; our ability to obtain regulatory approval
of, and ultimately commercialize, drug candidates; the impact of
the COVID-19 pandemic and force majeure events; our ability to
obtain, maintain, and enforce intellectual property protections;
cyberattacks or other disruptions to our technology systems; our
ability to attract, motivate, and retain key employees and manage
our growth; inflation and other macroeconomic issues; and other
risks and uncertainties such as those described under the heading
"Risk Factors" in our filings with the U.S. Securities and Exchange
Commission, including our most recent Quarterly Report on Form 10-Q
and our Annual Report on Form 10-K. All forward-looking statements
are based on management's current estimates, projections, and
assumptions, and Recursion undertakes no obligation to correct or
update any such statements, whether as a result of new information,
future developments, or otherwise, except to the extent required by
applicable law.

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/recursion-provides-business-updates-and-reports-second-quarter-2022-financial-results-301602924.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/recursion-provides-business-updates-and-reports-second-quarter-2022-financial-results-301602924.html
SOURCE Recursion