PAVmed Announces Successful First-in-Human Implantations of its PortIO™ Intraosseous Infusion System
17 März 2022 - 1:25PM
Business Wire
First three patients underwent successful
implantation and clinical utilization of PortIO as part of a
first-in-human study in Colombia, South America
PAVmed Inc. (Nasdaq: PAVM, PAVMZ) (the “Company” or
“PAVmed”), a diversified commercial-stage medical technology
company, today announced that physicians at the Clinica Porto Azul
in Barranquilla, Colombia successfully implanted the Company’s
PortIO™ Intraosseous Infusion System in three patients—the first
human implants of the device—as part of its IRB-approved
first-in-human (FIH) clinical study of up to 40 patients. All
patients have also undergone successful infusion of fluids
consistent with the study protocol and the device’s intended use.
No complications have occurred.
“This is a groundbreaking milestone in the field of vascular
access,” said implanting interventional radiologist Dr. David
Sabbag of Sabbag Radiology at Clinica Porta Azul. “PortIO
implantation was technically straightforward and well-tolerated by
the patients, as was its use for infusions of fluid. We are very
excited about what we believe is the first maintenance-free,
long-term vascular access device. All other vascular access devices
require costly, labor-intensive regular maintenance with flushes to
prevent clotting and obstruction. Repeated flushing can also result
in serious or even deadly complications such as infection and
clotting disorders. PortIO has the potential to provide significant
benefits to many patients – particularly those with poor venous
access, those on, or expected to need, kidney dialysis, and those
with known adverse reactions to the commonly used anticoagulant
heparin.”
“Simply stated, PortIO worked exactly as designed and intended,”
said Brian J. deGuzman, M.D., PAVmed’s Chief Medical Officer, who
led the PAVmed team in Colombia, trained the physicians, and was
present in the interventional suite during the first implantations.
“Although, for many decades, physicians have used the intraosseous
route to infuse fluids, medications, and other substances into
patients through the bone marrow instead of through a vein,
existing intraosseous devices protrude through the skin and can
only be used for 48 hours or less, generally for emergency
situations. PortIO is the first implantable intraosseous vascular
access device designed for long-term use and to eliminate many of
the shortcomings of existing vascular access devices, including the
need for regular flushing to maintain patency. We look forward to
completing this FIH study to demonstrate that PortIO can serve as
such a maintenance-free long-term vascular access device.”
The PortIO Intraosseous Infusion System consists of an
implantable intraosseous vascular access device and insertion kit.
Instead of a catheter located in a vein, it has a short extension
from the device, which a physician inserts into a bone, leaving the
device to reside completely beneath the skin. This allows direct
access to the bone marrow, which is a well-established route for
the delivery of medications, fluids, and other substances. PortIO
can be inserted and removed near-percutaneously without requiring a
surgical pocket or significant dissection and does not require
confirmation of the position of the tip by x-ray or other means.
Once in place, the device can be accessed by the nurse through the
skin using the same techniques as existing implantable ports.
PortIO addresses known limitations of existing long-term vascular
access devices which, in addition to the need for regular
maintenance with flushes, include occlusion from blood clots, blood
stream infections, and difficult or impossible insertion in
patients with poor veins. PortIO’s addressable market opportunity
is estimated to be at least $500 million based on short-term
patients with poor veins and medium- or long-term patients who
would benefit from its advantages over existing devices. This
estimate does not include a separate, possibly larger, market
opportunity in patients with renal failure whose veins must be
carefully preserved for future hemodialysis.
About PAVmed
PAVmed Inc. is a diversified commercial-stage medical technology
company operating in the medical device, diagnostics and digital
health sectors. Its major subsidiary, Lucid Diagnostics Inc.
(Nasdaq: LUCD), markets the EsoGuard® Esophageal DNA Test and
EsoCheck® Esophageal Cell Collection Device—the first and only
commercial tools for widespread early detection of esophageal
precancer to prevent esophageal cancer deaths. Another major
subsidiary, Veris Health Inc., is a digital health company
developing the first intelligent implantable vascular access port
with biologic sensors and wireless communication to improve
personalized cancer care through remote patient monitoring.
PAVmed’s CarpX® Minimally Invasive Device for Carpal Tunnel
Syndrome is currently in limited commercial release. The product
pipeline also includes the EsoCure™ Esophageal Ablation Device with
Caldus™ Technology, which complements EsoGuard and EsoCheck, the
NextFlo™ Intravenous Infusion Set, the PortIO™ Implantable
Intraosseous Vascular Access Device, novel pediatric ear tubes,
mechanical circulatory support technology and glucose monitoring.
For more information, please visit www.pavmed.com, follow us on
Twitter, connect with us on LinkedIn, and watch our videos on
YouTube. For more information on our majority owned subsidiary,
Lucid Diagnostics Inc., please visit www.luciddx.com, follow Lucid
on Twitter, and connect with Lucid on LinkedIn. For detailed
information on EsoGuard, please visit www.EsoGuard.com and follow
us on Twitter, Facebook and Instagram.
Forward-Looking Statements
This press release includes forward-looking statements that
involve risk and uncertainties. Forward-looking statements are any
statements that are not historical facts. Such forward-looking
statements, which are based upon the current beliefs and
expectations of PAVmed’s management, are subject to risks and
uncertainties, which could cause actual results to differ from the
forward-looking statements. Risks and uncertainties that may cause
such differences include, among other things, volatility in the
price of PAVmed’s common stock; PAVmed’s Series W and Series Z
warrants; general economic and market conditions; the uncertainties
inherent in research and development, including the cost and time
required to advance PAVmed’s products to regulatory submission;
whether regulatory authorities will be satisfied with the design of
and results from PAVmed’s clinical and preclinical studies; whether
and when PAVmed’s products are cleared by regulatory authorities;
market acceptance of PAVmed’s and products once cleared and
commercialized; PAVmed’s ability to raise additional funding as
needed; and other competitive developments. In addition, PAVmed has
been monitoring the COVID-19 pandemic and the pandemic’s impact on
PAVmed’s businesses. PAVmed expects the significance of the
COVID-19 pandemic, including the extent of its effect on its
financial and operational results, to be dictated by, among other
things, the success of efforts to contain the pandemic and the
impact of such efforts on PAVmed’s businesses. These factors are
difficult or impossible to predict accurately and many of them are
beyond PAVmed’s control. In addition, new risks and uncertainties
may arise from time to time and are difficult to predict. For a
further list and description of these and other important risks and
uncertainties that may affect PAVmed’s future operations, see Part
I, Item IA, “Risk Factors,” in PAVmed’s most recent Annual Report
on Form 10-K filed with the Securities and Exchange Commission, as
the same may be updated in Part II, Item 1A, “Risk Factors” in any
Quarterly Report on Form 10-Q filed by PAVmed after its most recent
Annual Report filed with the Securities and Exchange Commission.
PAVmed disclaims any intention or obligation to publicly update or
revise any forward-looking statement to reflect any change in its
expectations or in events, conditions, or circumstances on which
those expectations may be based, or that may affect the likelihood
that actual results will differ from those contained in the
forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220317005414/en/
Investors Adrian K. Miller PAVmed
Inc. AKM@PAVmed.com
Media Shani Lewis
LaVoieHealthScience (609) 516-5761
PAVmed@lavoiehealthscience.com
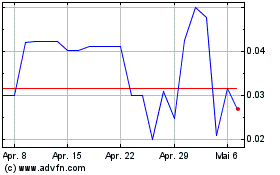
PAVmed (NASDAQ:PAVMZ)
Historical Stock Chart
Von Mär 2024 bis Apr 2024
PAVmed (NASDAQ:PAVMZ)
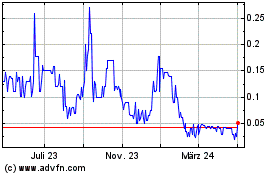
Historical Stock Chart
Von Apr 2023 bis Apr 2024