Press release Biocartis Group NV: Biocartis announces the US FDA 510(k) clearance for the Idylla™ MSI Test
02 März 2023 - 7:00AM

Press release Biocartis Group NV: Biocartis announces the US FDA
510(k) clearance for the Idylla™ MSI Test
PRESS RELEASE
2 March 2023, 7:00 CET
BIOCARTIS
ANNOUNCES THE US FDA
510(k) CLEARANCE FOR THE
IDYLLA™ MSI TEST
Mechelen,
Belgium, 2 March
2023 – Biocartis Group NV (the ‘Company’ or
‘Biocartis’), an innovative molecular diagnostics company (Euronext
Brussels: BCART), today announces the U.S. Food and Drug
Administration (FDA) 510(k) clearance1 for its fully automated
Idylla™ MSI Test. This 510(k) clearance reinforces Biocartis’
commitment to enable clinical molecular diagnostics in the U.S.
Now, labs of all sizes can benefit from Idylla™’s high sensitivity,
unmatched ease-of-use, and rapid turnaround times.
MSI is the result of inactivation of the body’s
so-called DNA mismatch repair (MMR) system, which normally
spontaneously corrects errors that occur during DNA replication. In
case this MMR system does not function properly, microsatellite
instability occurs. MSI-High (MSI-H) is detected in approximately
15% of all colorectal cancers and 3% are associated with Lynch
syndrome, whereas the other 12% have sporadic disease2. Lynch
syndrome is the most common cause of hereditary colorectal cancer
and is caused by inherited changes (mutations) in genes that affect
DNA mismatch repair3.
The Idylla™ MSI Test is cleared for in-vitro
diagnostic use on the Biocartis Idylla™ System only. The
Idylla™ MSI Test, for use on the Idylla™ System, uses
formalin-fixed, paraffin-embedded (FFPE) tissue sections of human
CRC tumor, from which nucleic acids are liberated, then analyzed
using PCR amplification of seven monomorphic biomarkers (ACVR2A,
BTBD7, DIDO1, MRE11, RYR3, SEC31A and SULF2) and subsequent
melt-curve analysis. The Idylla™ MSI Test reports results as
either microsatellite stable (MSS), or microsatellite instability
high (MSI-H) or invalid. Idylla™ MSI Test is indicated for use by
healthcare professionals for the qualitative identification of
microsatellite instability (MSI) in colorectal cancer (CRC) tumors,
indicative of mismatch repair deficiency, as an aid in the
identification of potential Lynch syndrome to help identify
patients that would benefit from additional genetic testing to
diagnose Lynch syndrome. The results from the Idylla™ MSI
Test should be interpreted by healthcare professionals in
conjunction with other clinical findings, family history, and other
laboratory data. The Idylla™ MSI Test should not be used for
diagnosis of CRC. The clinical performance of this device to guide
treatment decision for MSI high patients has not been
established.
The Idylla™ MSI Test is a fully automated test,
that provides information on the MSI status of CRC tumors within
approximately 150 minutes from just one section of formalin-fixed,
paraffin-embedded (FFPE) tumor tissue, without the need for paired
normal tissue sample.
Commenting on the U.S. Food and Drug
Administration (FDA) 510(k) clearance Herman Verrelst, Chief
Executive Officer of
Biocartis said:
“This first US FDA 510(k) clearance of an oncology assay is a major
milestone for the Company. Both large and small US labs are
expected to benefit from this fast and easy to use Idylla™ MSI
testing thanks to the fully automated sample-to-result nature of
our platform. We can now start to commercialize our in-vitro
diagnostic solution for clinical use, which will unlock significant
additional market potential and pave the way for continued strong
growth of our oncology business in the U.S. We continue to build
momentum in our regulatory program and plan to submit more products
to the U.S. FDA, also supported by our pharma partners.”
--- END ---
More information:
e-mail: ir@biocartis.com
@Biocartis_
www.linkedin.com/Biocartis
About Biocartis
With its revolutionary and proprietary Idylla™
platform, Biocartis (Euronext Brussels: BCART) aspires to enable
personalized medicine for patients around the world through
universal access to molecular testing, by making molecular testing
actionable, convenient, fast and suitable for any lab. The Idylla™
platform is a fully automated sample-to-result, real-time PCR
(Polymerase Chain Reaction) based system designed to offer in-house
access to accurate molecular information in a minimum amount of
time for faster, informed treatment decisions. Idylla™'s
continuously expanding menu of molecular diagnostic tests address
key unmet clinical needs, with a focus in oncology. This is the
fastest growing segment of the molecular diagnostics market
worldwide. Today, Biocartis offers tests supporting melanoma,
colorectal, lung and liver cancer, as well as for COVID-19, Flu,
RSV and sepsis. For more information, visit www.biocartis.com
or follow Biocartis on Twitter @Biocartis_ , Facebook or
LinkedIn. Biocartis and Idylla™ are registered trademarks in
Europe, the United States and other countries. The Biocartis and
Idylla™ trademark and logo are used trademarks owned by Biocartis.
Please refer to the product labeling for applicable intended uses
for each individual Biocartis product. This press release is not
for distribution, directly or indirectly, in any jurisdiction where
to do so would be unlawful. Any persons reading this press release
should inform themselves of and observe any such restrictions.
Biocartis takes no responsibility for any violation of any such
restrictions by any person. This press release does not constitute
an offer or invitation for the sale or purchase of securities in
any jurisdiction. No securities of Biocartis may be offered or sold
in the United States of America absent registration with the United
States Securities and Exchange Commission or an exemption from
registration under the U.S. Securities Act of 1933, as amended.
Forward-looking
statementsCertain statements, beliefs and opinions in this
press release are forward-looking, which reflect the Company's or,
as appropriate, the Company directors' or managements' current
expectations and projections concerning future events such as the
Company's results of operations, financial condition, liquidity,
performance, prospects, growth, strategies and the industry in
which the Company operates. By their nature, forward-looking
statements involve a number of risks, uncertainties, assumptions
and other factors that could cause actual results or events to
differ materially from those expressed or implied by the
forward-looking statements. These risks, uncertainties, assumptions
and factors could adversely affect the outcome and financial
effects of the plans and events described herein. A multitude of
factors including, but not limited to, changes in demand,
competition and technology, can cause actual events, performance or
results to differ significantly from any anticipated development.
Forward-looking statements contained in this press release
regarding past trends or activities are not guarantees of future
performance and should not be taken as a representation that such
trends or activities will continue in the future. In addition, even
if actual results or developments are consistent with the
forward-looking statements contained in this press release, those
results or developments may not be indicative of results or
developments in future periods. No representations and warranties
are made as to the accuracy or fairness of such forward-looking
statements. As a result, the Company expressly disclaims any
obligation or undertaking to release any updates or revisions to
any forward-looking statements in this press release as a result of
any change in expectations or any change in events, conditions,
assumptions or circumstances on which these forward-looking
statements are based, except if specifically required to do so by
law or regulation. Neither the Company nor its advisers or
representatives nor any of its subsidiary undertakings or any such
person's officers or employees guarantees that the assumptions
underlying such forward-looking statements are free from errors nor
does either accept any responsibility for the future accuracy of
the forward-looking statements contained in this press release or
the actual occurrence of the forecasted developments. You should
not place undue reliance on forward-looking statements, which speak
only as of the date of this press release.
1 A 510(k) is a premarketing submission made to FDA to
demonstrate that the device to be marketed is as safe and
effective, that is, substantially equivalent (SE), to a legally
marketed device that is not subject to premarket approval (PMA).
510(k) (premarket notification) to FDA is required at least 90 days
before marketing unless the device is exempt from 510(k)
requirements. Source:
https://www.fda.gov/medical-devices/products-and-medical-procedures/device-approvals-denials-and-clearances,
last consulted on 28 February 20232 Dudley JC et al. (2016)
Microsatellite instability as a biomarker for PD-1 blockade. Clin
Cancer Res. 22(4):813–8203 Source: CDC, last consulted online here
on 27 February 2023
Biocartis Group NV (EU:BCART)
Historical Stock Chart
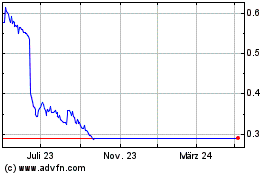
Von Mär 2024 bis Apr 2024
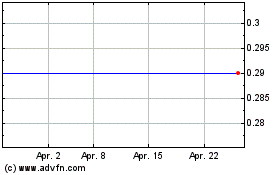
Biocartis Group NV (EU:BCART)
Historical Stock Chart
Von Apr 2023 bis Apr 2024